Abstract
The oil seed crops are the important sources of phenolic compounds in the human diet. Important oil seed crops include olive and soybean. In Virgin olive oil (VOO) Phenolic compounds is present which are important in regard to dietary and breeding aspects. Study of Olive oil composition shows that it contains two fractions i.e. major and minor fractions with most of its fundamental medical advantages related to its minor fraction. The minor fraction contains phenols, alcohols, tocopherols and some other compounds. The Phenolic compounds is determined by different some genetic and environmental factors and there is a direct connection of phenolic profile of the olive fruit with phenolic profile of VOO. Modern analytical tools are utilized to find a connection between fatty acids, sterols, tocopherols, or squalene with the composition of olive fruit and oil. A class of important secondary metabolites, Secoiridods are essentially important part of VOO. The composition of secondary metabolites shows that different parts of olive contains diverse quantities of secondary metabolites e.g. flavonoids quercetin, rutin and luteolin7-glucoside are present only in the fruit peel. salidroside and nuzhenide are only found in the seed of olive tree. The investigation of early steps of the flavonols biosynthesis pathway shows that CHI (Chalcone--flavanone isomerase) is a crucial gene for flavonol production. The role of phenol toward ecological aspects is of vital importance because they assume a critical job in the plant reaction to ecological signs. Genes which regulate and degrade various secondary compounds in olive fruits are still not defined due to unavailability of sequencing data of olive.
Keywords: Metabolic Phenolic compound; QTLs; Oil Seed Crops
Abbreviations: FLS: Flavonol Synthase; VOO: Virgin olive oil; TAG: Triacylglycerol; MUFA: Mono Unsaturated Fatty Acid; DHPAE: 3,4-dihydroxyphenylethanol; F3H: Flavanone 3-hydroxylase; CHS: Chalcone Synthase; MAS: Marker Assisted Selection; PAL: Phenylalanine Ammonia-lyase; FDS: Farnesyl Diphosphate Synthase; GGPP: Geranylgeranyl diphosphate; SQE: Squalene Epoxide; SQS: Squalene Synthase; DHPAC: 3,4-dihydroxyphenylacetic Acid; DMAPP: Dimethylallyl Pyrophosphate
Introduction
Background
The olive, known by the botanical name Olea europaea is the most imperative tree of the Mediterranean region, which accounts for 95% olive trees that are developed globally Loumou and Giourga [1]; Khan et al. [2]. Olive oil is the 6th abundant vegetable oil manufactured globally so we can say that prime objective behind olive plant production is oil extraction Conde et al. [3]; Peragon et al. [4]. Olea europaea is a specie that contains 46 chromosomes in each cell (diploid chromosome 2n=46). Seed development is an important process in olive and seeds are produced via out crossing. Khadari et al. [5]; Lee et al. [6]; Busconi et al. [7]. These seeds have the capacity to stay feasible for long term. It is a glycophytic specie and is more resistant towards stresses like salinity and dry season when contrasted with other fruit trees that are susceptible to these stresses. Oil which is solely taken out from fruits is generally called Olive oil. However, the term “virgin olive oil (VOO)” represents the oil that is separated from fruits by utilizing physical or mechanical approaches. Gila et al. [8] So, for the purpose of virgin olive oil production we can apply different techniques like cleaning with water, process of decantation, separation by the process of centrifugation and removing impurities by the process of filtration. However, such conditions should be necessarily provided, that supports adjustments (IOOC, 2014).
The importance of Virgin olive oil (VOO) inside the Mediterranean eating regimen is due to the fact, that it is surely an advantageous everyday nutrition for human wellbeing and is considered as an important part of daily diet Estruch et al. [9], Sahyoun et al. [10]. Europe is producing 78% of olive oil in the world production. While “Picual” and “Arbequina” are the two cultivars which isessential Spanish varieties regarding oil generation and Spain being the its biggest maker has produced average of 1.3 million tons’ oil in the course of recent years. Most of those cultivars are grown frequently, precise breeding methods are now undertaking to create new varieties. Bellini et al. [11]; Lavee et al. [12]. The phenolic components which were separated from the seeds of soyabean had effective anti-cancer activities which include gastric, breast, ovarian, prostate and colorectal cancers Belaj et al. [13]; Dong et al. [14]; Niedzwiecki et al. [15].
It is predicted that there is some kind of relationship between flavonoid contents, phenolic compound components in developed soybean. We can conclude that the goals of latest breeding projects are not just agronomic.
In this scenario, there is a need to fulfill two distinctive market methodologies
(i) Production of surplus virgin olive oil with good quality and its should be affordable for consumers as well,
(ii) Offering shoppers, a variety of extra VOO with fantastic norms and diverse tactile characterize or profiles.
These two approaches are associated with the Mediterranean daily nutrition. Shah et al. [16] The first perspective is normally identified with super-escalated development and exceedingly automated harvesting techniques. The second perspective indicates to ensure the protection of biodiversity of olive tree and conventional strategies as a component of the exceptional nutrition customs related with the eating routine of Mediterranean nations Ilarioni and Proietti [17].
The main goal of this review is to understand that phenolic profiling and its composition in oil seed crops. Also understand the different phenolic pathways in oil biosynthesis in oil seed crops. Study the what kind of enzyme involve in active the Virgin olive oil. Further perspective of the genetic factors involved in the advancement of olive fruit in regard to its phenolic composition.
Phenol Biochemical Composition Related to Nutrition:
Olive breeding programs have been negatively influenced because there essential agronomic and oil quality attributes which have not been given interest. Learning should be expanded to identify real variability of olive germplasm for a large number of useful traits. Few examinations on olive oil have revealed two fractions in its composition. The first one being the major fraction is saponifiable. This fraction constitutes 98 to 99% of the total weight of oil and called as the glyceride fraction. Kiritsakis et al. [18]; Romero et al. [19] Primary composition of major fraction consists of the lipid compound triacylglycerol (TAG). Further, there are some mono-glycerol and diglycerols found accumulated to this lipid compound. An investigation on virgin olive oil composition has revealed that oleic acid, linoleic acid, palmitic acid and some other acids are quintessential fatty-acid components of VOO (Table 1). Oleic acid is the most prevalent (68-81. 5%) of them and it also characterizes it amongst monounsaturated fatty acids oils (MUFA oils) Ramírez et al. [20].
Table 1: In this table describe techniques some are natural traits e.g. glycophytic which enhanced resistant, others are accomplished by artificially as shown in table.
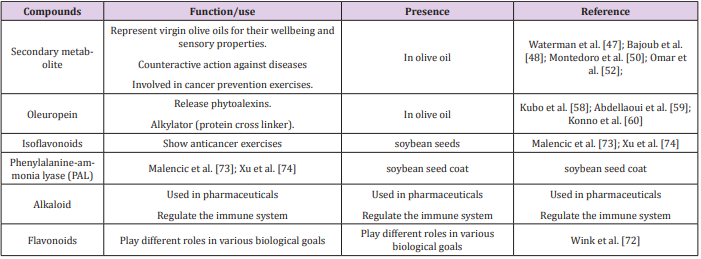
It was found that proportion among n-6 and n-3 unsaturated fats isn’t of most elevated, for example 16 (distributed extents for vegetable oils: 0-738) and subsequently its fundamental presumed medical advantages are normally connected with its minor division Dubois et al. [21]. Be that as it may, minor fraction comprises in excess of 230 distinct parts, which usually comprises 1-2% of total weight of oil. Moreover, A few classes can be incorporated into these compounds, e.g., non-glyceride esters (for example waxes, hydrocarbons (for example squalene),sterols (Figure 1) (for example campestral), few alcohols of aliphatic and triterpenic type, some polar stains(for example chlorophylls),tocopherols and volatiles (such as the aromatic aldehyde i.e. benzaldehyde) and a class of secondary metabolites called phenolic compounds (like hydro-xytyrosol), Ramírez-Tortosa et al. [20].However, it was found that not many of them were recognized as bioactive compounds Covas et al. [22] further explored the importance and advantages of those which were recognized as biologically active and significant. Olive-breeding projects with new dietetic targets have been driven to meet the increasing demands of the consumers with respect to olive oil quality Rugini et al. [23]; Velasco et al. [24]. It is generally accepted that the main health related characteristics of VOO are attributed by phenolic compound seven though VOO contains various minor compound fractions with fascinating organic exercises.
Hence, regarding both dietary and breeding aspects the phenolic compounds present in VOO are of great importance Bernardini and Visioli et al. [25]. Further, it is known that these phenolic compounds in VOO are the primary supporters of bitter and pungent sharp tangible descriptors which implies that these phenolic contents in VOO also have essential organoleptic connotations. In every single olive item, secoiridoid compounds are plentiful in which consist of phenolic alcohol tyrosol (p-HPEA) or its hydroxyl subordinate hydro xytyrosol (3,4-DHPEA) in their structure. Secoiridoids oleuropein, (Figure 1) ligstroside and dimethyl oleuropein are found to be the primary phenolic glucosides present in the olive. Their hydrolytic subsidiaries, the di aldehydic types of decarboxy-methyl oleuropein and decarboxy methyl ligstroside aglucones (3,4-DHPEA-EDA and p-HPEA-EDA, respectively) and the aldehydic types of oleuropein and ligstroside aglucones (3,4-DHPEA-EA and p-HPEA-EA, individually) are phenolic parts in olive oils which are known to be most essential components. It was reported by further examinations that levels of 3,4-DHPEA in plasma is connected to the capacity of VOO phenolics to the lessen chronic inflammation and oxidative harm Redondo et al. [26]; Bernardini et al. [13]. These results were helpful for the European Union to support a wellbeing guarantee on polyphenols of olive oil that contain less than 250 mg/kg of hydroxy tyrosol and its subordinates (European Commission, 2012).
Metabolic Profiling of Oil Seed Crops:
The complex phenomenon of phenol metabolism in the olive tree are regulated by different ecological Romero and Motilva [27] and genetic factors Talhaoui et al. [28] and these are the main determinants of the final composition phenolic contents in olive fruit. Likewise, the impact of other influential factors has been explained which include important farming practices, for example, restricted water system Gennai et al. [29], the advancement of pruning to expand light accessibility Famiani et al. [30] or determination of accurate harvest date Proietti et al. [31] etc. Further examinations revealed that during oil extraction process the activation of few oxidative enzymes and some endogenous hydrolytic is involved with the modification of phenolic glycosides which are present in the fruits of olive tree (Table 2). In such a way, it was found that there is a direct connection of phenolic profile of the olive fruit with phenolic characterization of VOO. It was also revealed that during milling and malaxation, action of hydrolytic and oxidative which is also related to profile of virgin olive oil. Purpose of improvement of VOO’s phenolic composition, a well-designed experimental structure should be considered for any program of breeding. There are many factors which affects the phenolic composition of VOO.
Table 2: In this table the classes of phenolic compounds and other compounds present in the oil seed crops, identification, role and for which it is used are described.
For this purpose, precise scientific tools are required to find the effect of each of these particular factors on the ultimate phenolic composition of VOO. It was revealed that there are ongoing projects of olive breeding which mainly focus on the quality of olive, but these programs have extra restrictions to those recently referenced Velasco et al. [24]. The reason of these limitations is a very little amount of oil production from a very vast number of genotypes in the beginning periods of the breeding. Thus, it is fundamental to carefully examine the fruit to predict total oil composition using solid logical analytical tools. Fatty acids, tocopherols, squalene, or sterols are important components of the composition of olive fruit and oil. For this reason, examinations were carried out which portrayed a huge connection between the profile of olive fruit and oil for above mentioned components Velasco et al. [32]; Rosa et al. [28]. Olive breeding programs till now do not provide any evidence about the utilization of phenolic fruit profiling as selection criteria in these programs.
Secondary Metabolites Distribution in Olive Fruit
Eocardiids is a class of fundamental secondary metabolites which are widely found in the Mesocarp tissue of olive fruit. These Secoiridods are actually gatherings of monoterpenoids with a cleave dimethyl cyclopentane skeleton and are common in few dicotyledonous families and few flowering plants of Oleaceae family. These secondary metabolites or Secoiridods are phenol-conjugated aggravates with a glycoside element and are bounteous in olive tree. Oleuropein (Figure 1), dimethyl oleuropein, oleuroside, ligstroside, nuzhenide are considered as most important Secoiridods in olive fruit are VOO. Also, their structure forms, such as the dialdehydic form of decarboxymethyl elenolic acid linked to either 3,4-DHPEA or p-HPEA (3,4-DHPEA-EDA and p-HPEA-EDA, respectively) the ligstroside aglycon (p-HPEA-EA) and an isomer of oleuropein aglycon (3,4-DHPEA-EA), are essentially important Obied et al. [33]. In extra-virgin olive oil, a secoiridoid compound called Oleocanthal (p-HPEA-EDA) was observed which was missing from the fruits Beauchamp et al. [34]. Flavonoids and lignans, phenolic acids and some phenolic alcohols like hydro xytyrosol (3,4-DHPEA) and tyrosol (p-HPEA) are some important phenolics of olive Oliveras et al. [35] which were seen in the fruits with the pulp being richest region of these phenolics Owen et al. [36].
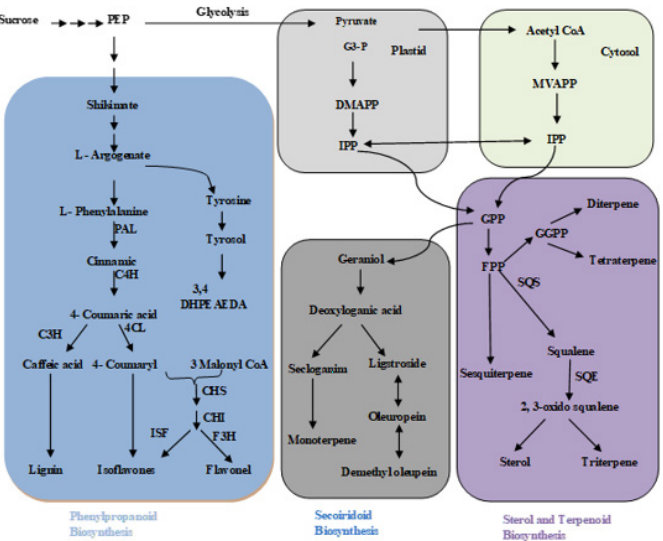
In oil seed crops different pathways are involved MEP, MVA, Phenylpropanoid, secoridoid, sterol and terpenoid pathways.
Each pathway depends upon different enzyme that can work to active these phenolic compounds which is helpful for our health. In oilseed crops sucrose (Figure 1) is convert into PEP (Figure 1) by different process that is indeterminate until PEP is convert by different enzyme are involve shikimate (Figure 1) is a primary step to produce phenylpropanoid compound in oil seed crops. Shikimate are convert by different enzyme to produce the Phenylpropanoid pathway (Figure 1). PEP covert into Pyruvate (Figure 1) in the presence of glycolysis in plastid then covert into IPP (Figure 1) with the help of different enzyme. Pyruvate are converting Acetyl CoA (Figure 1) and acetyl CoA is convert in IPP (Figure 1) with the help of different enzymein cytosol (Figure 1). In oilseed crops Secoiridoid Biosynthesis (Figure 1) and Sterol and Terpenoid Biosynthesis (Figure 1) are start from IPP (Figure 1). If IPP (Figure 1) are active than both of these pathways is produced. In Secoiridoid Biosynthesis Geraniol (Figure 1) are convert into Deoxyloganic acid (Figure 1) by different enzyme work it. In Sterol and Terpenoid Biosynthesis GPP (Geranyl diphosphate) (Figure 1) are convert into FPP (Figure 1) produce Tetraterpene, Diterpene, Sesquiterpene and Squalene (Figure 1). Squalene is conert into Sterol and Triterpene (Figure 1). In all pathways different enzyme are involved to produce Phenolic compound (Figure 1).
At maturity there has been observed A high measure of glycoside Verbasco side Servili et al. [37] was observed in products of a few cultivars of olive. A small concentration of the compound comselogoside, vanillic acid Boskou et al. [38] and Jermanet al. [39], caffeic acid, p-coumaric acid, 3,4-dihydroxyphenylacetic acid (DHPAC) and homo vanillic alcohol are some other phenolics present in the fruit at maturity. Tocopherols Dabbou et al. [40], oleanolic acids and maslinic acid are few important triterpenic acids (Figure 1) found in olive fruit and are of great significance. Specific compounds which are present in various tissues of the fruit like stone, leaf, mesocarp and exocarp have been discovered by the investigation of phenolic profiles in these tissues. For example, flavonoids (Figure 1) quercetin, rutin and luteolin-7-glucoside were found to be present only in the fruit peel Servili et al. [41]. Similarly, only olive seeds contain phenolic compounds salidroside and nuzhenide Ryan et al. [42]; Mansour et al. [43]. A crucial gene, CHI (Figure 1) was observed for enhancing flavonol production. It is a protein which is involved in the primary stages of the flavonols biosynthesis pathway Muir et al. [44]. Petunia CHI (Figure 1) and CHS (Figure 1) (Chalcone synthase) genes in tomatoes which were overexpressed and increase insufficient amount of rutin and naringenin substance, separately. Be that as it may, RNAi hindrance of the tomato CHS1(Figure 1) genebrought about a solid decrease of both naringenin and quercetin levels Martens et al. [45].
It was found that F3H (Figure 1) (flavanone 3-hydroxylase) and FLS (Flavonol synthase)over expression had no consequences for flavonoid levels when contrasted with non-transgenic controls, however F3H (Figure 1) RNAi tomatoes brought about a 20% decline in wild-type rutin intensities Muir et al. [44] and Bovy et al. [46]. Further, the oil which is extracts from seed overexpression with multigene (CHS, CHI, and (DFR)(Figure 1) dihydroflavonol 4-reductase) from petunia into flax displayed larger amounts of quercetin (46– 90%), kaempferol (70– 83%), and anthocyanin (198%) than the control Zuk et al. [47]. The precursor compound of some triterpenic diols (for example, uvaol and erythrodiol) and amyrins (for example, α-and β-amyrins) is known as squalene (Figure 1) which is also an intermediate product of the pathways of sterol synthesis (Figure 1). Only olive and some other vegetable oils contain squalene (Figure 1) in a steady sum. Due to its perceived consequences for human wellbeing this compound is considered very significant phenolic compound Waterman et al. [48]; Bajoub et al. [49].
It was revealed that when the final size of olive fruit is achieved and process of ripening starts, progressive accumulation of 24-methylenecycloartanol, cycloartenol and β-sitosterol Sterolsbegins in olive fruit Stiti et al. [50]. Secoiridoids (Figure 1) usually represents the most perilous microconstituents present in virgin olive oils important for their sensory properties and wellbeing because Secondary metabolites are not soluble in oil. However, only a little amount of Secoiridoid compounds is recouped in the oil after the practice of mechanical extraction is done Montedoro et al. [51]; Martins et al. [52]. The secoiridoids compunds of olive are important agents against diseases like atherosclerosis and these are also involved in restraining peroxidation of lipoprotein conjugates of low-density Waterman et al. [48]. Further, their cancer prevention exercises have also been demonstrated Omar et al. [53]. Additionally, diseases like osteoporosis are prevented by the activities of these Secoiridoid compounds. Regarding well-being of human oleocanthal Beauchamp et al. [34]., hydro xytyrosol Omar et al. [53]. and Oleuropein are known to specifically affect human health in great ways. Furthermore, secoiridoids (Figure 1) are known to be engaged with oil oxidative security by acting as primary antioxidants. Secoiridoids (Figure 1) also impact oil taste Andar accountable for pungency and bitterness sensory notes hence it is adding to the quality of olive oil in different ways Servili et al. [37].
Role of Phenol Toward Ecological Aspects
The study of Phenolics is of great importance because they assume a critical job in the plant reaction to ecological signs and they are most essential resistance components against defoliating bugs Mumm et al. [54]. They likewise influence shoot spreading Umehara et al. [55] and have been guessed to be involved in the cell security and counteract fungal infiltration into the cambial region Beckman and Franceschi et al. [56]. Lately, A few information bolsters the possibility that the protection from explicit pathogens may likewise be identified with specific sorts of phenolics which acts as cell guards [57,58]. It was demonstrated that Oleuropein (Figure 1) displays the most grounded movement noted for a plant metabolite and it is thought to be in charge of the release of phytoalexins which an anti-parasitic substance Kubo et al. [59]; Abdellaoui et al. [60]. It behaves as a multifunctional alkylator that acts as aperfect protein cross linker. The strongest activities shown by this plant metabolite diminishes nutritive estimation of proteins which are involved in daily nutrition. Thus, it badly affects herbivores Konno et al. [61]. Till now, only some pathways of secoiridoid (Figure 1) metabolism in Oleaceae species have been suggested Butthe complete process of secoiridoid (Figure 1) metabolism is still not clarified Damtoft et al. [62]; Alagna et al. [63].
The composition and expression of secoiridoids varies considerably among varieties, formative stages and, tissues. Various ecological factors also influence the expression and composition of secoiridoids (Figure 1) Malik et al. [64]. Only few genes have been described which are engaged with biosynthetic processes of triterpene while there are some crucial genes, which regulate and degrade various secondary compounds in olive fruits, are still not defined Shibuya et al. [65] and Saimaru et al. [66]. The reason behind this failure is the absence of data for the olive genome sequencing. Till now, only few goals like transcriptome data of olive fruit, are achieved regarding olive sequencing Galla and Alagna et al. [67]. As the process of fruit metabolism is yet to be demonstrated and the genes related to the process are still to be identified so this transcriptome data of olive fruit could be a useful asset for the scientists to identify genes related with the process. Perennial woody species are long lasting and practical genetic investigations are hard to perform with them because productive protocols for transformation, invitro reculture and mutagenesis are absent. Therefore, it would be a valuable tool to understand regular variations for genes of great importance. The latest work done is to investigate various complex metabolic pathways by the combination of gene expression and metabolic informational indexes Saito et al. [68].
QTLs Regulating the Different Phenolic Compound in Oil Crops
In order to understand intricate nature of these pathways we can utilize modern genetic techniques. Additionally, advanced genetic tools like Marker Assisted Selection (MAS) and other genomic apparatuses could be introduced in olive breeding projects to produce useful olive cultivars with attractive phenotypes in brief time. Moreover, research is progressing on its sequencing because genomic investigations could be accelerated if sequence information of olive is generated Torchia et al. [69]; Rosa et al. [70]. Different crop has different size of genome the size of olive genome is expected to be around 1,800 Mb and high amount of intra-specific genetic variations are found within Loureiro et al. [71]. Further advancements in olive like genome mapping, and identifying the sequence is still ongoing. These are cooperating in expanding further information about olive genome and it could be a one big step towards great achievements Collins et al. [72]. Work is still ongoing on sequencing of olive, though whole plastome sequencing of an Italian cultivar “Frantoio” was reported by Marriotti et al. [23]. An attempt on olive genome sequencing was endeavored recently by project entitled OLEA in Italy. There is a need to understand different essential biochemical and physiological elements of olive plant.
To understand these essential dynamics of the olive tree, latestgenomics studies should be employed which include omics studies like transcriptomic, proteomics and metabolic. Also, by understanding the role of proteins and their respective genes in Arabidopsis different pathways of lipid arrangement and oxidation have been identified for olive plant. Alkaloids, terpenoids (Figure 1) and phenolics are the three noteworthy groups of plant secondary metabolites This categorization is done on the basis of their biosynthetic origin Wink et al. [73]. Flavonoids (Figure 1) and non-flavonoids are two major types of phenolic metabolites which are originated from aromatic amino acids via phenylpropanoid (Figure 1) pathway. Flavonoids (Figure 1) and Iso-flavonoids are compounds with backbone of 2-phenylchromen4-one and 3-phenylchromen-4-one respectively. The purpose of this investigation is to include iso flavonoids to the term flavonoids. These iso flavonoids are found abundantly in soyabean seeds which are also contain other phenolic compounds in abundant Malencic et al. [74]. It was depicted that the phenolic components which were separated from the seeds of soyabean had effective anticancer activities which include gastric, breast, ovarian, prostate and colorectal cancers Xu and Chang [75,76]; Dong et al. [14]. It is predicted that there is some kind of relationship between flavonoid contents, phenolic compound components in developed soya bean. Endeavors have likewise been made to investigate this relationship and to understand the phenolic aggregation of the soybean seed.
Soybean with black seed coat is the plant under examination to identify these however, there was no specific relationship found between seed coat color and antioxidant activities of soybean seed (Qi et al. [77]). Some investigations on crop improvement have revealed that some oil crop like wild soybeans are important genetic assets for improvement after provided with a strong proof from Next-generation sequencing-based studies. So wild soybean can be introduced into crop improvement programs to modify other cultivars to achieve desired products Kim et al. [78]; Zhou et al. [79]. Few investigations have declared that QTL that regulates antioxidant contents lies on chromosome 19. This QTL was identified in a recombinant population that was developed by a cross between cultivated and wild soybean parents Qi et al. [77]. It was then confirmed that this QTL does not coincide with the QTL that controls anthocyanin contains a seed coat color Qi et al. [77], that provide a fact that high anthocyanin substance in seed coats has no direct relationship with high seed antioxidant exercises in oil crops like wild soybeans [80-90]. Besides, the identified QTL was found recognizably different from the other of QTLs genistein and total isoflavone which is present on chromosome 19. phenylalanine-ammonia lyase (PAL) (Figure 1)-encoding gene is found in this region Gutierrez et al. [80]. The committing step in the phenolic and flavonoid synthesizing via phenylpropanoid pathway is catalyzed by PAL (Figure 1) [91-95].
Figure 1: A proposed diagram showing the biosynthetic pathways of different phenolic compounds in oil seed crops.
Conclusion
Different studies are aiming to understand the composition, profiling and biosynthesis of phenolic compounds or secondary metabolites in oil seeds [96-101]. Although, addition of genomic data in databases has accelerated genomic studies but still there is a lot of scope available to researchers for mining the genes related to the breakdown of secondary metabolite in oil seeds. To understand these essential dynamics of the oil seeds, latest integrated genomics studies should be employed which include omics studies like transcriptomic, proteomics and metabolic. That will explore the functional characterization of proteins and genes involved in biosynthesis.
References
- Loumou A, Giourga C (2003) Olive groves: The life and identity of the Mediterranean. Agric Hum Values 20(1): 87-95.
- Khan T, Abbasi BH, Khan MA, Shinwari ZK (2016) Differential effects of thidiazuron on production of anticancer phenolic compounds in callus cultures of Fagonia indica. Appl Biochem Biotechnol 179: 46-58.
- Conde C, Delrot S, Geros H (2008) Physiological, biochemical and molecular changes occurring during olive development and ripening. J Plant Physiol 165(15): 1545-1562.
- Gomez de la Cruz Fj, PJC Palaez, JMP Carnicero, FC Peragon (2015) Modelling of olive oil mill waste rotary dryer: Green energy recovery system. J Appl Therm eng 80: 362-373.
- Besnard G, Khadari B, Villemur P, A. Bervillé (2000) Cytoplasmic male sterility in the olive (Olea europaea L.). Theor Appl Genet 100(7): 1018- 1024.
- Doveri S, Donal M, Lee D (2006) Non concordance between genetics profiles of olive oil and fruit: a cautionary note to the use of DNA markers for provenance testing. J Agric Food Chem 54(24): 9221-9226.
- Sebastiani L, Busconi M (2017) Recent developments in olive (Olea europaea L.) genetics and genomics: Applications in taxonomy, varietal identification, traceability and breeding. Plant Cell Rep 36: 1345-1360.
- Gila A, A Jimenez, G Beltran, A Romero (2015) Correlation of fatty acid composition of virgin olive oil with thermal and physical properties. Eur J Lipid Sci Technol 117(3): 366–376.
- Estruch R, Ros E, Salas Salvado J, Covas MI, Corella D, et al. (2013) Primary prevention of cardiovascular disease with a Mediterranean diet. N Engl J Med 368: 1279–1290.
- Sahyoun NR, Sankavaram K (2016) Historical origins of the mediterranean diet, regional dietary profiles and the development of the dietary guidelines. In Mediterranean Diet, p. 43-56.
- Bellini E, Giordani E, Rosati A (2002) Genetic improvement of olive from clonal selection to crossbreeding programs. Adv Hortic Sci 22: 73–86.
- Lavee S (2013) Evaluation of the need and present potential of olive breeding indicating the nature of the available genetic resources involved. Sci Hort 161: 333–339.
- Belaj A, Dominguez Garcia, Gustavo Atienza, S Martin Urdiroz N, De La Rosa, et al. (2012) Developing a core collection of olive (Olea europaea L.) based on molecular markers (DArTs, SSRs, SNPs) and agronomic traits. Tree Genet Genomes 8: 365–378.
- Dong X, Xu WQ, Sikes RA, Wu CQ (2012) Apoptotic effects of cook edandin vitro digested soy on human prostate cancer cells. Food Chem 135(3): 1643-1652.
- Niedzwiecki A, MW Roomi, T Kalinovsky, M Rath (2016) Anticancer Efficacy of Polyphenols and Their Combinations. J Nutrients 8(9): 552- 568.
- Shah ZH, Hamooh BT, Daur I, Rehman HM, Alghabari F (2017) Transcriptomics and biochemical profiling: Current dynamics in elucidating the potential attributes of olive. Curr Issues Mol Biol 21: 73- 98.
- Ilarioni L, Proietti P (2014) “Olive tree cultivars,” in The Extra-Virgin Olive Oil Handbook, ed C Peri (West Sussex: John Wiley & Sons, Ltd.), p. 59–67.
- Kiritsakis A, Christie W, Harwood JL, Aparicio R (2000) Handbook of Olive Oil Aspen Publishers, Gaithersburg (Maryland), pp. 129–151.
- Ramírez Tortosa MC, Granados S, Quiles JL (2006) Chemical composition. Types and characteristics of olive oil. Olive Oil and Health. CAB International, Oxfordshire, England, UK.
- Dubois V, Breton S, Linder M, Fanni M, Parmentier M (2007) Fatty acid profiles of 80 vegetable oils with regard to their nutritional potential. Eur J Lipid Sci Tech 109: 710-732.
- Covas MI, Ruiz Gutiérrez V, De la Torre R, Kafatos A, Lamuela Raventós RM, et al. (2006) Minor components of olive oil: Evidence to date of health benefits in humans. Nutri Rev 64: 20-30.
- Mariotti R, Cultrera NGM, Munoz Diez C, Baldoni L, Rubini A (2010) Identification of new polymorphic regions and differentiation of cultivated olives (Olea europaea L.) through platome sequence comparison. BMC Plant Biol, p. 10.
- De la Rosa R, Arias Calderón R, Velasco L, León L (2016) Early selection for oil quality components in olive breeding progenies. Eur J Lipid Sci Technol 118: 1160–1167.
- Bernardini E, Visioli F (2017) High quality, good health: the case for olive oil. Eur J Lipid Sci Technol.
- Mateos R, Pereira Caro G, Saha S, Cert R, Redondo Horcajo M, et al. (2011) Acetylation of hydroxytyrosol enhances its transport across differentiated Caco-2 cell monolayers. Food Chem 125(3): 865-872.
- Romero MP, Motilva MJ (2010) Effect of climatic conditions on quality of virgin olive oil,” in Olives and Olive Oil in Health and Disease Prevention, eds VR Preedy, RR Watson (San Diego, CA: Elsevier Inc), p. 43–50.
- Talhaoui N, Gómez-Caravaca AM, León L, De la Rosa R, FernándezGutiérrez A, et al. (2016) From olive fruits to olive oil: Phenolic compound transfer in six different olive cultivars grown under the same agronomical conditions. Int J Mol Sci 17: 337.
- Cirilli M, Caruso G, Gennai C, Urbani S, Frioni E, et al. (2017) The role of polyphenoloxidase, peroxidase, and b-glucosidase in phenolics accumulation in Olea europea L. Fruits under different water regimes. Front Plant Sci 8: 717.
- Proietti P, Nasini L, Famiani F, Guelfi P, Standardi A (2012) Influence of light availability on fruit and oil characteristics in Olea europaea L. Acta Hortic 949: 243–250.
- Famiani F, Proietti P, Farinelli D, Tombesi A (2000) Oil quality in relation to olive ripening. Acta Hortic 586: 671–674.
- Velasco L, Fernandez-Cuesta A, De la Rosa R, Ruiz-Mendez M V, León L (2014) Selection for some olive oil quality components through the analysis of fruit flesh. J Am Oil Chem 91: 1731-1736.
- Obied HK, Prenzler PD, Ryan D, Servili M, Taticchi A, et al. (2008) Biosynthesis and biotransformations of phenol-conjugated oleosidic secoiridoids from Olea europaea L. Nat Prod Rep 25(6): 1167–1179.
- Beauchamp GK, Keast RS, Morel D, Lin J, Pika J, et al. (2005) Phytochemistry: ibuprofen-like activity in extra-virgin olive oil. Nature 437(7055): 45–46.
- Oliveras López MJ, Innocenti M, Ieri F, Giaccherini C, Romani A, et al. (2008) HPLC/DAD/ESI/MS detection of lignans from Spanish and Italian Olea europaea L. fruits. Journal of Food Composition and Analysis 21(1): 62–70.
- Owen RW, Haubner R, Mier W, Giacosa A, Hull WE, et al. (2003) Isolation, structure elucidation and antioxidant potential of the major phenolic and flavonoid compounds in brined olive drupes. Food Chem Toxicol 41(5): 703–717.
- Servili M, Selvaggini R, Esposto S, Taticchi A, Montedoro G, et al. (2004) Health and sensory properties of virgin olive oil hydrophilic phenols: Agronomic and technological aspects of production that affect their occurrence in the oil. J Chromatogr A 1054(1–2): 113-127.
- Boskou G, Salta FN, Chrysostomou S, Mylona A, Chiou A, et al. (2006) Antioxidant capacity and phenolic profile of table olives from the Greek market. Food Chem 94(4): 558–564.
- Jerman T, Trebše P, Mozeticˇ Vodopivec B (2010) Ultrasound-assisted solid liquid extraction (USLE) of olive fruit (Olea europaea) phenolic compounds. Food Chem 123(123): 175–182.
- Dabbou S, Issaoui M, Esposto S, Sifi S, Taticchi A, et al. (2009) Cultivar and growing area effects on minor compounds of olive oil from autochthonous and European introduced cultivars in Tunisia. J Sci Food Agric 89(8): 1314–1325.
- Servili M, Baldioli M, Selvaggini R, Macchioni A, Montedoro G (1999) Phenolic compounds of olive fruit: One- and two-dimensional nuclear magnetic resonance characterization of nuzhenide and its distribution in the constitutive parts of fruit. J Agric Food Chem 47(1): 12-18.
- Ryan D, Antolovich M, Herlt T, Prenzler PD, Lavee S, et al. (2002) Identification of phenolic compounds in tissues of the novel olive cultivar hardy’s mammoth. J Agric Food Chem 50(23): 6716-6724.
- Mansour AB, EA Porter, GC Kite, MSJ Simmonds, R Abdelhedi, et al. (2015) Phenolic profile characterization of chemlali olive stones by liquid Chromatography-Ion trap mass spectrometry. J Agric Food chem 63(7): 1990-1995.
- Muir SR. Collins GJ, Robinson S (2001) Overexpression of petunia chalcone isomerase in tomato results in fruit containing increased levels of flavonols. Nature Biotechnology 19(5): 470-474.
- GWM Schijlen, CHR De Vos, S Martens et al. (2007) RNA interference silencing of chalcone synthase, the first step in the flavonoid biosynthesis pathway, leads to parthenocarpic tomato fruits. Plant Physiology 144(3): 1520–1530.
- Bovy E Schijlen, RD Hall (2007) Metabolic engineering of flavonoids in tomato (Solanum lycopersicum): The potential for metabolomics. Metabolomics 3(3): 399–412.
- M Zuk, A Kulma, L Dymi´nska et al. (2011) Flavonoid engineering of flax potentiate its biotechnological application. BMC Biotechnology 11: 10.
- Waterman E, Lockwood B (2007) Active components and clinical applications of olive oil. Altern Med Rev 12(4): 331-342.
- Bajoub A, EH Fernandez, EA Ajal, N Ouazzani, A Gutierrez, et al. (2015) Comprehensive 3-year study of the phenolic profile of Moroccan monovarietal virgin olive oil from the meknes region. J Agric Food Chem 63(17): 4376-4385.
- Stiti N, Triki S, Hartmann MA (2007) Formation of triterpenoids throughout olea europaea fruit ontogeny. Lipids 42(1): 55-67.
- Montedoro G, Baldioli M, Selvaggini R, Begliomini AL, Taticchi A (2002) Relationship between phenolic compounds of olive fruit and olive oil: importance of the endogenous enzymes. Acta Hort 586: 551–556.
- Martins FP, A Silva, V Almeida, M carvalheira, C Serra, et al. (2013) Protective activity of hydroxytyrosol metabolites on erythrocyte Oxidative-Induce hemolysis. J Agric Food chem 61(27): 6636-6642.
- Omar SH (2010) Oleuropein in olive and its pharmacological effects. Sci Pharm 78(2): 133–154.
- Mumm R, Hilker M (2006) Direct and indirect chemical defence of pine against folivorous insects. Trends Plant Sci 11(7): 351–358.
- Umehara M, Hanada A, Yoshida S, Akiyama K, Arite T, et al. (2008) Inhibition of shoot branching by new terpenoid plant hormones. Nature 455(7210): 195-200.
- Franceschi VR, Krokene P, Christiansen E, Krekling T (2005) Anatomical and chemical defenses of conifer bark against bark beetles and other pests. New Phytol 167(2): 353–375.
- Ockels FS, Eyles A, McPherson BA, Wood DL, Bonello P (2007) Phenolic chemistry of coast live oak response to Phytophthora ramorum infection. J Chem Ecol 33(9): 1721–1732.
- Wallis C, Eyles A, Chorbadjian R, McSpadden Gardener B, Hansen R, et al. (2008) Systemic induction of phloem secondary metabolism and its relationship to resistance to a canker pathogen in Austrian pine. New Phytol 177(3): 767-778.
- Kubo I, Matsumoto A, Takase I (1985) A multichemical defense mechanism of bitter olive Olea europaea (Oleaceae). Is oleuropein a phytoalexin precursor? J Chem Ecol 11(3): 251–263.
- Abdellaoui K, O Boussadia, M Miladi, I Boughattas, G Omri, et al. (2019) Olive Leaf Extracts Toxicity to the Migratory Locust, Locusta migratoria: Histopathological Effects on the Alimentary Canal and Acetylcholinesterase and Glutathione S-Transferases Activity. J Neotrop Entomol 48(2): 246–259.
- Konno K, Hirayama C, Yasui H, Nakamura M (1999) Enzymatic activation of oleuropein: a protein crosslinker used as a chemical defense in the privet tree. Proc Natl Acad Sci 96(16): 9159–9164.
- Damtoft S, Franzyk H, Jensen SR (1995) Biosynthesis of eocardiids in Fontanesia. Phytochemistry 38(3): 615–621.
- Alagna F, F Geu Flores, H Kries, F Panara, L Baldoni, et al. (2016) Identification and Characterization of the Iridoid Synthase Involved in Oleuropein Biosynthesis in Olive (Olea europaea) Fruits. J BIO CHEM 291(11): 5542–5554.
- Malik NSA, Bradford JM (2008) Recovery and stability of oleuropein and other phenolic compounds during extraction and processing of olive (Olea europaea L.) leaves. Journal of Food Agriculture and Environment 6(2): 8-13.
- Shibuya M, Zhang H, Endo A, Shishikura K, Kushiro T, et al. (1999) Two branches of the lupeol synthase gene in the molecular evolution of plant oxidosqualene cyclases. Eur J Biochem 266(1): 302-307.
- Saimaru H, Orihara Y, Tansakul P, Kang YH, Shibuya M, et al. (2007) Production of triterpene acids by cell suspension cultures of Olea europaea. Chem Pharm Bull (Tokyo) 55(5): 784-788.
- Galla G, Barcaccia G, Ramina A, Collani S, Alagna F, et al. (2009) Computational annotation of genes differentially expressed along olive fruit development. BMC Plant Biol 9: 128.
- Saito K, Hirai MY, Yonekura-Sakakibara K (2008) Decoding genes with co expression networks and metabolomics - ‘majority report by precogs’. Trends Plant Sci 13(1): 36-43.
- Alagna F, D Agostino N, Torchia L, Servili M, Rao R, et al. (2009) Comparative 454 pyrosequencing of transcripts from two olive genotypes during fruit development. BMC Genomics 10: 399.
- Donaire L, Pedrola L, De la Rosa R, L lave C (2011). High-throughput sequencing of RNA silencing-associated small RNAs in olive (Olea europaea L.). PLoS ONE 6: e27916.
- Loureiro J, Rodriguez E, Costa A, Santos C (2007) Nuclear DNA content estimations in wild olive (Olea europaea L. ssp. Europaea var. Sylvestris Brot.) and Portuguese cultivars of O. europaea using flow cytometry. Genet Resour Crop Evol 54(1): 21-25.
- Wu S, Collins G, Sedgley M (2004) A molecular linkage map of olive (Olea europaea L) based on RAPD, microsatellite, and SCAR markers. Genome 47: 26-35.
- Wink M (2010) Introduction: biochemistry, physiology and ecological functions of secondary metabolites. Annual plant reviews: biochemistry of plant secondary metabolism 40: 1-19.
- Malencic D, Popovic M, Miladinovic J (2007) Phenolic content and antioxidant properties of soybean (Glycinemax (L.) Merr.) seeds. Molecules 12(3): 576–581.
- Xu B, Chang SK (2011) Reduction of antiproliferative capacities, cellbased antioxidant capacities and phytochemical contents of common beans and soybeans upon thermal processing. Food chem 129: 974-981.
- Xu B, Chang SK (2012) Comparative study on antiproliferation properties and cellular antioxidant activities of commonly consumed food legumes against nine human cancer cell lines. Food chem 134: 1287-1296.
- Zhang Y, M Wang, FC Zhao, Liang Mian Chen, Qi Wei Zhang et al. (2014) Phenolic metabolites from the stems and leaves of Sophora flavescens. Helvetica Chimica Acta 97(11): 1516–1525.
- Kim MY, Lee S, Van K, Kim TH, Jeong SC, et al. (2010) Whole-genome sequencing and intensive analysis of the undomesticated soybean (Glycine soja Sieb and Zucc) genome. Proc Natl Acad Sci USA 107(51): 22032-22037.
- Zhou Z, Jiang Y, Wang Z, Gou Z, Lyu J, et al. (2015) Resequencing 302 wild and cultivated accession side notifies genes related to domestication and improvement in soybean. Nat Biotechnol 33: 408-414.
- Gutierrez Gonzalez JJ, Wu XL, Gillman JD, Lee JD, Zhong R, et al. (2010b) Intricate environment-modulated genetic networks control isoflavone accumulation in soybean seeds. BMC Plant Biol 10: 105.
- Abdessemeda S, I Muzzalupob, H Benbouza (2015) Assessment of genetic diversity among Algerian olive (Olea europaea L.) cultivars using SSR marker. J Sci Horti 192: 10–20.
- Ghasemzadeh, N Ghasemzadeh (2011) Flavonoids and phenolic acids: role and biochemical activity in plants and human. Journal of Medicinal Plant Research 5(31): 6697–6703.
- Bartolini G, Prevost G, Messeri C, Carignani C (2005) Olive Germplasm: Cultivars and World-Wide Collections. Rome: FAO/Plant Production and Protection.
- Beckman CH (2000) Phenolic-storing cells: keys to programmed cell death and periderm formation in wilt disease resistance and in general defence responses in plants? Physiological and Molecular Plant Pathology 57: 101–110.
- B Weisshaar, GI Jenkinst (1998) Phenylpropanoid biosynthesis and its regulation. Current Opinion in Plant Biology 1(3): 251–257.
- Winkel Shirley (2001) Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiology 126(2): 485–493.
- Cho KM, Lee YB, Seo WD, Kim JY, Ryu HW, et al. (2013) Soluble phenolics and antioxidant properties of soybean (Glycine max L.) cultivars with varying seed coat colours. J Funct Foods 5: 1065–1076.
- Treutter (2005) Significance of flavonoids in plant resistance and enhancement of their biosynthesis. The Journal of Plant Biology 7(6): 581–591.
- El Riachy M, Priego Capote F, León L, Luque de Castro MD, Rallo L (2012) Virgin olive oil phenolic profile and variability in progenies from olive crosses. J Sci Food Agric 92(12): 2524–2533.
- Elloumi J, Ben Ayed R, Aifa S (2012) An overview of olive oil biomolecules. Curr Biotechnol 1(2): 115-124.
- I Hwang, M Kaneko, Y Ohnishi, S Horinouchi (2003) Production of plant-specific flavanones by Escherichia coli containing an artificial gene cluster. Applied and Environmental Microbiology 69(5): 2699–2706.
- García Gavil JF, M Bullo, S Canudas, MA Martínez Gonzalez, R Estruch, et al. (2018) Extra virgin olive oil consumption reduces the risk of osteoporotic fractures in the PREDIMED trial. J Clinical Nutrition 37(1): 329-335.
- Gomez Rico A, Fregapane G, Salvador MD (2008) Effect of cultivar and ripening on minor components in Spanish olive fruits and their corresponding virgin olive oils. Food Res Int 41(4): 433–440.
- Goulas V, Manganaris GA (2012) Towards an Efficient Protocol for the Determination of Triterpenic Acids in Olive Fruit: A Comparative Study of Drying and Extraction Methods. Phyto chem Anal 23(5): 444–449.
- Kim, MR Lee, GS Lee, WG An, SI Cho (2012) Effect of Sophora flavescens Aiton extract on degranulation of mast cells and contact dermatitis induced by dinitrofluorobenzene in mice. Journal of Ethnopharmacology 142(1): 253-258.
- J Lu, S Ye, R Qin, Y Deng, CP Li (2013) Effect of Chinese herbal medicine extracts on cell-mediated immunity in a rat model of tuberculosis induced by multiple drug-resistant bacilli. Molecular Medicine Reports 8(1): 227–232.
- J Piquemal, S Chamayou, I Nadaud, Beckert M, Barrière Y, et al. (2002) Down-regulation of caffeic acid O-methyltransferase in maize revisited using a transgenic approach. Plant Physiology 130(4): 1675-1685. Republic of Korea.
- K Bae (2000) Medicinal Plants of Korea, K Bae, Ed, Kyo Hak Pub, Seoul, Republic of Korea.
- Lam HM, Xu X, Liu X, Chen WB, Yang GH, et al. (2010) Resequencing of 31wild and cultivated soybean genomes identifies patterns of genetic diversity and selection. Nat Genet 42: 1053-1041.
- Mataix J, Barbancho FJ (2006) Olive oil in Mediterranean food. Olive Oil and Health. CAB International, Oxfordshire, England, UK.
- Owen R, Giacosa A, Hull E, Haubner R, Würtele G, et al. (2000) Oliveoil consumption and health: the possible role of antioxidants. Lancet Oncol 1: 107-121.
- Pérez AG, León L, Pascual M, Romero Segura C, Sánchez Ortiz A, et al. (2014) Variability of virgin olive oil phenolic compounds in a segregating progeny from a single cross in Olea europaea L. and sensory and nutritional quality implications. PLoS ONE 9(3): e92898.
- Rallo L, Barranco D, De la Rosa R, León L (2008) “Chiquitita” olive. Hort Science 43(2): 529–533.
- Riachy ME, Priego Capote F, León L, Rallo L, Luque de Castro MD (2011a) Hydrophilic antioxidants of virgin olive oil. Part 1: Hydrophilic phenols: A key factor for virgin olive oil quality. Eur J Lipid Sci Technol 113(6): 678-691.
- Romero Segura C, García Rodríguez R, Sanz C, Pérez AG (2011) Virgin olive phenolic profile as a result of the anabolic and catabolic enzymes status in the olive fruit. Acta Hort 924: 379–384.
- Rugini E, De Pace C (2016) Olive breeding with classical and modern approaches. The Olive Tree Genome, pp. 163-193.
- Saimaru H, Orihara Y, Tansakul P, Kang YH, Shibuya M, et al. (2007) Production of triterpene acids by cell suspension cultures of Olea europaea. Chem Pharm Bull (Tokyo) 55(5): 784-788.
- Servili M, Montedoro GF (2002) Contribution of phenolic compounds to virgin olive oil quality. European Journal of Lipid Science and Technology 104: 602-613.
- Servili M, Sordini B, Esposto S, Urbani S, Veneziani G, et al. (2014) Biological activities of phenolic compounds of extra virgin olive oil. Antioxidants 3: 1-23.
- Subramanian S, Graham MY, Yu O, Graham T L (2005) RNA interference of soybean isoflavone synthase genes leads to silencing in tissues distal to the transformation site and to enhanced susceptibility to Phytophthora sojae. Plant Physiology 137(4): 1345-1353.
- Wang W, You R L, Qin W J (2015) Anti-tumor activities of active ingredients in compound kushen injection. Acta Pharmacologica sinica 36(6): 676-679.
- He X, Fang J, Huang L, Wang J, Huang X (2015) Sophora flavescens ait.: Traditional usage, phytochemistry and pharmacology of an important traditional Chinese medicine. Journal of Ethnopharmacology 172: 10- 29.
- Piao XL, Piao XS, Kim SW, Park JH, Kim HY, et al. (2006) Identification and characterization of antioxidants from Sophora flavescens. Biological & Pharmaceutical Bulletin 29(9): 1911-1915.
- Xu B, Chang SK, Liu Z, Yuan S, Zou Y, et al. (2010) Comparative studies on the chemical and cell-based antioxidant activities and antitumor cell proliferation properties of soymilk manufactured by conventional and commercial UHT methods. J Agric Food Chem 58: 3558-3566.

 Review Article
Review Article