Abstract
In forensic practice the unavailability of traditional biological samples for toxicological examinations, such as in decomposed body, is not rare. The continuous scientific research of possible alternative matrices has identified the bone tissue as a substrate usable for the toxicological purpose. Particularly, rib bone marrow (BM) seems to be an optimal specimen for xenobiotics recovery. The aim of the present study was a suitability assessment of rib BM to detect acute and previous xenobiotics consumption. Rib segments of about 5 g were spiked with antidepressant (Amitriptyline, Citalopram, Venlafaxine and Sertraline) and antipsychotic (Haloperidol, Chlorpromazine, Clotiapine, Methotrimeprazine, Promazine) at the concentration levels of 200 and 500 ng/g. Pig ribs were examined at time 0 and after 15, 30, 60 and 90 days to verify the persistency of the substances. Histological study to verify any structural alterations in pig rib morphology after extraction procedure were carried out both fresh and following incubation at 25°C for 3 months. All the substances were still measurable after three months aging. Only haloperidol was no longer detectable at T90. No significant recovery differences were noted between ribs fortified with 200 or 500 ng/g, both for antidepressants and antipsychotics, at the other sampling times. No structural alterations in pig rib morphology neither after leaching nor after the aging process (25°C for 3 months) were found. There is limited scientific evidence to show that bone/BM drug concentrations reflect those of blood at time of death, yet. However, bone/BM drug detection is helpful in forensic investigation to give information on xenobiotics intake.
Keywords: Bone Marrow Ribs Pig; Toxicological Investigations; Aging Bone Experimental Model; Antidepressants; Antipsychotics
Abbreviations: BM: Bone Marrow; T0:After Fortification (fresh); T15: After 15 Days From The Fortification;T30: After 30 Days From The Fortification;T60: After 60 Days From The Fortification;T90: After 60 Days From The Fortification ;MSD: Mass Selective Detector ;SIM: Select Ion Monitoring; EDTA: Ethylene-Diamin-Tetra-Acetate; n.d.: No Detected
Introduction
Viscera, biological liquids, and hair represent the common samples for toxicological analyses. However, it is possible that these tissues are not available. This situation can occur when corpses are already skeletonized, burned or exhumed. In these cases, it is necessary to collect alternative matrices. It has been reported that other organs, such as bone marrow (BM), can be used for toxicological investigations [1,2]. Particularly, several authors have described how bone tissue is useful to detect many drugs [2-16], such as barbiturates [17,18], opioids [2-4,19-23], antidepressants [2,24-27], antipsychotics [2,24], benzodiazepines [2-4,28,29]. Particularly, rib BM seems to be an optimal specimen for xenobiotics recovery, because of the by high blood supply [2, 13,14,25,26,30,31].
The aim of the present study was a suitability assessment of rib BM to detect acute and previous xenobiotics consumption. To this end, we performed analyses on pig ribs fortified with antidepressant and antipsychotic mix concentrations. The pig ribs were examined by leaching both while fresh and following incubation at 25°C (to mimic a postmortem corruption). Specifically, pig ribs were examined at time 0 and after 15, 30, 60 and 90 days of aging to verify the recovery of the substances. The pig ribs, both fresh and following incubation at 25°C for 3 months, underwent histological study to evaluate their conditions after leaching and aging. the promising results suggest that rib bone tissue could be a usable substrate for toxicological detection of xenobiotics when conventional matrices are not available.
Materials and Methods
Samples
The study was performed on pig ribs fortified with antidepressant (Amitriptyline, Citalopram, Venlafaxine and Sertraline) and antipsychotic (Haloperidol, Chlorpromazine, Clotiapine, Methotrimeprazine, Promazine). Two different mix concentrations (200 and 500 ng/g) were used. Groups of seven pig ribs for each experimental concentration were examined both fresh and following incubation at 25°C for 3 months (to mimic a post mortem corruption).
Extraction Procedure
The extraction was carried out on rib segments of about 5 g. Each rib was cleaned of any remains of muscles and connective tissues by sharp dissection. The ribs were then washed with deionized water to remove external contaminations and left to dry in the air. Each sample was spiked with 500 ng/g of bupivacaine as internal standard. The extraction was performed by leaching process using absolute ethanol + 1% ammonia solution. The eluate was acidified with HCl (37%) to pH 5-6 and then evaporated to dryness by rotavapor. The residue was reconstituted in 8 mL of chlorobutane, and sodium borate buffer (pH 11.5) until pH 9 was achieved. After centrifugation at 3000 rpm for 5 min, the organic phase was transferred in a vial, while the aqueous phase was alkalinized to pH 12 with NaOH 1 M and extracted with 4 mL of chlorobutane. After centrifugation at 3000 rpm for 5 min, the organic phase was reunited to the previous extract and acidified with 4 mL of H2SO4 0.1 M. After centrifugation at 3000 rpm for 5 min, the aqueous phase was transferred in a vial, and extracted twice, at pH 8-8.5 an at pH 12, with 2.5 mL of chlorobutane. The organic phases were reunited to the previous extract and added with 500 ng of lidocaine (as external standard) per gram of rib sample. The extract was evaporated to dryness under a steam of nitrogen. The residue was reconstituted in 30 mcl of acetone and transferred into autosampler vial for analysis.
GC-MS Analysis
A Hewlett-Packard (HP) gas-chromatograph (Agilent Technology Inc., Santa Clara, CA) coupled to a HP 5973 Mass Selective Detector (Agilent) equipped with AT 7683 Series autosampler was used. An HP5 cross-linked fused-silica capillary column (30 m, 0.25 mm i.d.) with a 0.25 ml film thickness (Agilent Technology Inc.) was linked to the Mass Selective Detector (MSD) through a direct capillary interface. The temperatures of the injection port and interface were 220 °C and 210 °C, respectively. The oven temperature was maintained at 100 °C for 2 min, then programmed to 220 °C at 30 °C/min. Helium was used as a carrier gas with a flow rate of 1.0 mL/min. A splitless injection mode was selected. Each rib extract was analyzed by select ion monitoring (SIM), in order to resolve the substances with the same retention time, by monitoring the following ions:
Bupivacaine (S.I.): 140; 141.
Amitriptyline: 58; 202; 215.
Sertraline: 274; 276; 304.
Citalopram: 58; 208; 238.
Venlafaxine: 58; 134; 179.
Haloperidol:224; 237; 206.
Chlorpromazine: 58; 272; 318.
Clotiapine: 83; 244; 273.
Methotrimeprazine: 58; 228; 328.
Promazine: 58; 238; 284.
Histological Evaluation
Collected bone fragments, both fresh and following incubation at 25°C for 3 months, were fixed in 4% formalin and then decalcified in EDTA (ethylene-diamin-tetra-acetate) for 36 hours. After decalcification samples were processed for routine paraffin inclusion. Transverse sections, 6μm thick, were cut and stained with haematoxylin-eosin staining.
Result
Antidepressants and Antipsychotics Recovery
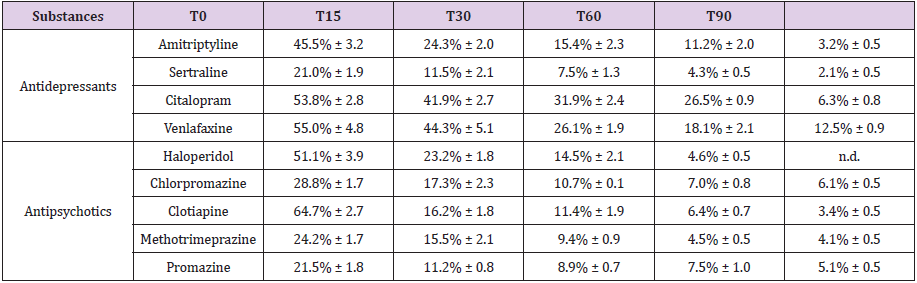
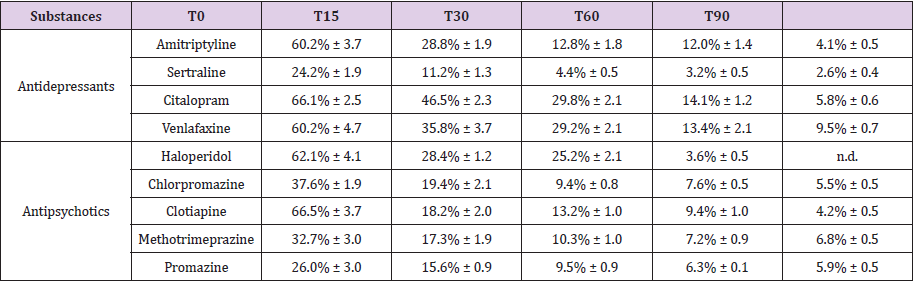
The recovery of antidepressants and antipsychotics was calculated comparing the peak area of external standard with that of each analyte tested. The recovery of the substances at 200 ng/g and 500 ng/g fortification (average of seven pig ribs for each experimental concentration) found at time 0 and after 15, 30, 60 and 90 days of aging is reported in Tables 1 & 2, respectively.
Table 1: Fortification: 200 ng/g. Average percentage found in the pig ribs at time 0 and after 15, 30, 60 and 90 days of aging.
Table 2: Fortification: 500 ng/g. Average percentage found in the pig ribs at time 0 and after 15, 30, 60 and 90 days of aging.
Histological Evaluation
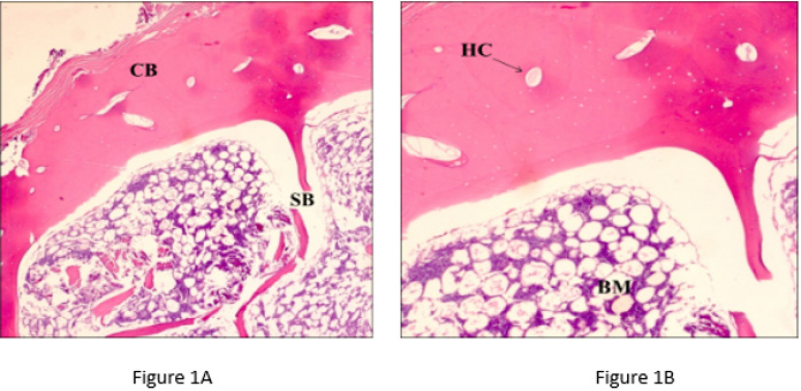
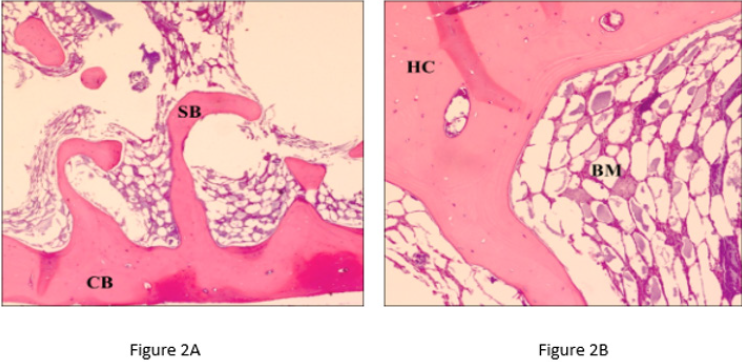
The results of the histological evaluation are illustrated in Figures 1A & 1B (fortified and leached; T0) and Figures 2A & 2B (fortified, leached and aged; T90).
Figure 1A & 1B: Ribs analyzed after fortification and leaching (T0). Magnification respectively ×20 and ×40 for left and right panel; Scale bars = 100 μm. CB (Compact Bone), SB (Spongy Bone), HC (Haversian Canal), BM (Bone Marrow).
Figure 2A & 2B: Ribs analyzed after fortification, leaching and three months aging (T90). Magnification respectively ×20 and ×40 for left and right panel; Scale bars = 100 μm. CB (Compact Bone), SB (Spongy Bone), HC (Haversian Canal), BM (Bone Marrow).
Discussion
In forensic practice the unavailability of traditional biological samples for toxicological examinations is not rare. In these cases, it is necessary to use the so-called alternative biological matrices in order to verify whether xenobiotics were present in the body of the deceased at the time of death; and if so, determine if they can be linked to the cause of death. In the last decade, there have been a number of reports describing extraction methods for the analysis of drugs in bone. Several studies have shown the possibility of using bone or bone marrow for post-mortem detection of xenobiotics [1- 32], even though the extraction of drugs from bones tissue is much more difficult compared to conventional extraction and recovery of analyte(s) from biological fluids or other types of tissue [12]. In fact, bone tissue provides a natural physical barrier that can prevent exposure to exogenous contaminating factors such as substances contained in the soil or products of soft tissue putrefaction, as well as protect against the destructive postmortem activity of fungi, bacteria, animals and plants [2]. , However, the marrow also has an extensive blood supply and its great vascularization and lipid-rich matrix allow the xenobiotics to enter it.
The nutritive artery provides the supply of blood, and its capillaries make a system of sinusoids inside the medullary cavity where the hematopoiesis takes place; such sinusoids converge into the central venous system of the bone, and the medullary blood return from there to the systemic venous circulation. In fact, the vessel walls are extremely thin and covered by a single layer of adventitial cells. The endothelial cells lining the vessels lumen form a barrier that limits the absorption of xenobiotics and regulates the passage of cells both in and out of the marrow through specific receptors. The nutrient artery-derived capillaries extend into the Haversian canals and return to the marrow cavity by opening into the venous sinuses. Thus, there is a circular pattern to blood flow within the marrow cavity, from the center of the marrow cavity toward the periphery of the marrow cavity then back toward the center [33]. The described properties of bone marrow are very advantageous for its use as a specimen for analyses in the field of forensic toxicology. However, it is not commonly used, due to insufficient data about the pharmacokinetics, as well as the absence of general knowledge on whether the drug concentrations found in marrow represent blood levels at the time of death [1,34]. According with other authors, our data show that ribs BM is an excellent analytical sample for xenobiotics recovery [2, 13,14,25,26,30,31]. In fact, a good recovery of all drugs has been obtained, although with small individual differences. A higher recovery rate was observed in the ribs treated with 500 ng/g, as showed in Tables 1 & 2, soon after fortification (T0). No significant recovery differences were noted between ribs fortified with 200 or 500 ng/g, both for antidepressants and antipsychotics, at the other sampling times. Among antidepressants, the best recovery was observed for Venlaxfaxine, followed by Citalopram and Amitriptyline; Sertraline showed the lowest recovery.
Among antipsychotics, the best recovery was observed for Clotiapine and Haloperidol; for Chlorpromazine, Methotrimeprazine and Promazine a lower recovery was achieved. All the substances were still measurable after three months aging. Only haloperidol was no longer detectable at T90. The extraction method used in the study reported here did not damage bone tissues as demonstrated by histology. In fact, no structural alterations in pig rib morphology neither after leaching nor after the aging process (25°C for 3 months) were found.
Conclusion
At the current state of knowledge, there is limited scientific evidence to show that bone/BM drug concentrations reflect those of blood at time of death. However, bone/BM drug detection is helpful in forensic investigation because it may reveal intake before death. Our study has been able to quantify, in ribs, nine substances of toxicological interest three months after aging. Further experimental data and validated analytical assays are necessary to make qualitative and quantitative determination in human BM samples.
Disclosures/Conflict of interest
The authors declare that there is no conflict of interest.
Funding
This research was funded by the Department of Medical and Surgical Sciences and Advanced Technologies “GF Ingrassia,” University of Catania, Italy (Piano Triennale di Sviluppo delle Attività di Ricerca Scientifica del Dipartimento 2016–18).
References
- Cartiser N, Bévalot F, Fanton L, Gaillard Y, Guitton J (2011) State-of-the-art of bone marrow analysis in forensic toxicology: a review. Int J Legal Med 125(2): 181-198.
- McIntyre LM, King CV, Boratto M, Drummer OH (2000) Post-mortem drug analyses in bone and bone marrow. Ther Drug Monit 22(1): 79-83.
- Mc Grath KK, Jenkins AJ (2009) Detection of drugs of forensic importance in postmortem bone. Am J Forensic Med Pathol 30(1): 40-44.
- Watterson JH, Desrosiers NA (2011) Microwave‐assisted extraction in the study of the effect of dose‐death interval on meperidine detection in skeletal tissue. Forensic Sci Int 207: 40-45.
- Watterson JH, Vandenboer TC (2008) Effects of tissue type and the dose‐death interval on the detection of acute ketamine exposure in bone and marrow with solid‐phase extraction and ELISA with liquid chromatography‐tandem mass spectrometry confirmation. J Anal Toxicol 3(8): 631-639.
- Österman T, Virtamo T, Kippo K, Laurén L, Pasanen I, et al. (1997) Distribution of clodronate in the bone of adult rats and its effects on trabecular and cortical bone. J Pharmacol Exp Ther 280(2): 1051-1056.
- Lien EA, Solheim E, Ueland PM (1991) Distribution of tamoxifen and its metabolites in rat and human tissues during steady‐state treatment. Cancer Res 51: 4837-4844.
- Ji AJ, Saunders JP, Wadgaonkar ND, Petersen PJ, O’Leary K, et al. (2007) A novel antibiotic bone assay by liquid chromatography/tandem mass spectrometry for quantitation of tigecycline in rat bone. J Pharm Biomed Anal 44: 970-979.
- Baeck SK, Lim MA, Park SY, Lee JS, Lee HS, et al. (2000) Blood concentrations of amitriptyline and its metabolite in rats after acute oral administration of amitriptyline. J Anal Toxicol 24: 271-274.
- Iskierka M, Zawadzki M, Szpot P, Jurek T (2019) Comparison of post-mortem ethanol level in blood and bone marrow. J Forensic Leg Med 61: 65-68.
- Watterson JH, Donohue JP, Betit CC (2012) Comparison of relative distribution of ketamine and norketamine in decomposed skeletal tissues following single and repeated exposures. J Anal Toxicol 36(6): 429-433.
- Orfanidis A, Gika H, Mastrogianni O, Krokos A, Theodoridis G, et al. (2018) Determination of drugs of abuse and pharmaceuticals in skeletal tissue by UHPLC-MS/MS. Forensic Sci Int 290: 137-145.
- De Souza Santos E, Spinelli E, Vieira AA, Rodrigues SV (2019) Postmortem analysis of famprofazone and its metabolites, methamphetamine and amphetamine, in porcine bone marrow. Talanta 191: 545-552.
- Wietecha Posluszny R, Lendor S, Garnysz M, Zawadzki M, Kóscielniak P(2017) Human bone marrow as a tissue in post-morten identification and determination of psychoactive Substances-Screening methodology. J. Chrom B 1061-1062: 459-467.
- Tominaga M, Michiue T, Ishikawa T, Kawamoto O, Oritani S, et al. (2013) Postmortem Analyses of Drugs in Pericardial Fluid and Bone Marrow Aspirate. J Anal Toxicol 37: 423-429.
- Kojima T, Okamoto I, Miyazaki T, Chikasue F, Yashiki M, et al. (1986) Detection of methamphetamine and amphetamine in a skeletonized body buried for 5 years. Forensic Sci Int 31: 93-102.
- Terazawa K, Takatori T (1982) Determination of aminopyrine and cyclobarbital from a skeleton by radioimmunoassay. J Forensic Sci 27(4): 844-847.
- Desrosiers NA, Betit CC, Watterson JH (2009) Microwave‐assisted extraction in toxicological screening of skeletal tissues. Forensic Sci Int 188(1-3): 23-30.
- Lafrenière NM, Watterson JH (2010) Detection of acute fentanyl exposure in fresh and decomposed skeletal tissues. Part II: the effect of dose‐death interval. Forensic Sci Int 194: 60-66.
- Guillot E, De Mazancourt P, Durigon M, Alvarez JC (2007) Morphine and 6‐acetylmorphine concentrations in blood, brain, spinal cord, bone marrow and bone after lethal acute or chronic diacetylmorphine administration to mice. Forensic Sci Int 166: 139-144.
- Raikos N, Tsoukali H, Njau SN (2001) Determination of opiates in postmortem bone and bone marrow. Forensic Sci Int 123: 140-141.
- Vanden Boer TC, Grummett SA, Watterson JH (2008) Utility of immunoassay in drug screening in skeletal tissues: sampling considerations in detection of ketamine exposure in femoral bone and bone marrow following acute administration using ELISA. J Forensic Sci 53(6): 1474-1482.
- Vandenbosch M, Somers T, Cuypers E (2018) Distribution of Methadone and Metabolites in Skeletal Tissue. J Anal Toxicol 42(6): 400-408.
- Horak EL, Jenkins AJ (2005) Postmortem tissue distribution of olanzapine and citalopram in a drug intoxication. J Forensic Sci 50(3): 1-3.
- Watterson JH, Cornthwaite HM (2013) Discrimination between patterns of drug exposure by toxicological analysis of decomposed skeletal tissues. Part II: Amitriptyline and citalopram. J Anal Toxicol 37(8): 565-572.
- Desrosiers NA, Watterson JH, Dean D, Wyman JF (2012) Detection of Amitriptyline, Citalopram, and Metabolites in Porcine Bones Following Extended Outdoor Decomposition. J Forensic Sci 57: 544-549.
- Vandenbosch M, Somers T, Cuypers E (2019) Distribution of clomipramine, citalopram, midazolam, and metabolites in skeletal tissue after chronic dosing in rats. Drug Test Anal 11(7): 1083-1093.
- Watterson JH, Botman JE (2009) Detection of acute diazepam exposure in bone and bone marrow:influence of tissue type and the dose‐death interval on sensitivity of detection by ELISA and liquid chromatography tandem mass spectrometry confirmation. J Forensic Sci 54(3): 708-714.
- Gorczynski LY, Melbye FJ (2001) Detection of benzodiazepines in different tissues, including bone, using a quantitative ELISA assay. J Forensic Sci 46(4): 916-918.
- Watterson JH, Desrosiers NA, Betit CC, Dean D, Wyman JF (2010) Relative distribution of drugs in decomposed skeletal tissue. J Anal Toxicol 34(8): 510-515.
- Mairs S, Swift B, Rutty GN (2004) Detergent: An alternative approach to traditional bone cleaning methods for forensic practice. Am J Forensic Med Pathol 25: 276-284.
- Mc Grath KK, Jenkins AJ (2009) Detection of drugs of forensic importance in postmortem bone. Am J Forensic Med Pathol 30(1): 40-44.
- Travlos GS (2006) Normal Structure, Function, and Histology of the Bone Marrow. Toxicologic Pathology 34: 548-565.
- Tattoli L, Tsokos M, Sautter J, Anagnostopoulos J, Maselli E, et al. (2014) Postmortem bone marrow analysis in forensic science: study of 73 cases and review of the literature. Forensic Sci Int 234: 72-78.

 Research Article
Research Article