Abstract
Introduction: Oxidative stress (OS) is associated with significant damage to cell structures leading to multiple disease patterns. Dietary supplementation or topical administrations of non-enzymatic antioxidants are described to prevent OS-related disease rates. Many of the most powerful and most discussed natural non-enzymatic antioxidants can be found in the plant kingdom. In this study, the antioxidant effect of a commercially available plant extract remedy was evaluated.
Methods: Primary human cells (peripheral blood mononuclear cells [PBMC], erythrocytes and polymorphonuclear cells [PMN]) were pre-incubated with Inflamyar™ (PEKANA Naturheimittel GmbH, Germany). Cell viability (MTT-assay), cellular antioxidant protection (CAP-e assay) and reactive oxygen species (ROS)-formation assays were performed.
Results: The MTT test showed a predominantly consistent to slightly increased cellular viability. A mild protection of oxidative damage in human erythrocytes at higher doses of the test substance was seen in the CAP-e assay. In the ROS assay, slightly reduced levels of ROS formation for all tested concentrations of the test substance were documented in PMN cells from two of three donors.
Conclusion: This study documented stable vitality and protective capacities against cellular oxidative stress of cells treated with the test substance.
Keywords: Inflamyar™; Flamyar™; Reactive Oxygen Species (ROS); Homeopathy; Spagyric
Abbreviations: OS: Oxidative Stress; PMN: Polymorphonuclear Cells; PBMC: Peripheral Blood Mononuclear Cells; ROS: Reactive Oxygen Species
Introduction
Oxygen is one of the most important elements for life, but in certain circumstances oxygen can also harm the organism. Influences of the outside world such as redox chemicals, ionizing radiation, UV light, heavy metal ions, ozone exposure and cigarette smoke can readily generate reactive oxygen species (ROS), which have an oxidizing effect on other substances [1,2]. A large part of these reactive species are represented by free radicals, for example hydroxyl radicals, superoxide radicals, nitric oxide radicals and the lipid peroxyl radical, and have an excess of free electrons. These substances are unstable and have a highly reactive potential on other substances [2]. In addition to the environmental formation of ROS, these can also be found as by-products in the oxygen metabolism of aerobic organisms. ROS are generated in connection with the electron transport in mitochondrial energy generation, immune reactions against microorganisms and viruses [3,4] as well as processes of the cytochrome P-450 system [5,6].
The human antioxidant defense system consists out of two components, an enzyme component on the one hand, comprising enzymes such as glutathione peroxidase, catalase and superoxide dismutase and the small molecule antioxidant component on the other, which comprises vitamin A and E, ascorbate, glutathione and thioredoxin. These substances are the natural protection against endogenous ROS and other free radicals. Oxidative stress occurs when the production of ROS exceeds the natural antioxidant defense mechanisms of the body and damages biomolecules such as lipids, proteins and nucleic acids [1].
Oxidative stress has been thought to contribute to the general decline in cellular functions that are associated with many human diseases including amyotrophic lateral sclerosis [7,8], Alzheimer’s disease [9-11], Parkinson’s disease [12,13], ischemia/ reperfusion neuronal injuries, degenerative disease of the human temporomandibular-joint [14], atherosclerosis [15], macular degeneration [7,16], degenerative retinal damage [17], cataract formation [18], multiple sclerosis [19], muscular dystrophy [20,21], rheumatoid arthritis [22], human cancers [23,24] the aging process itself [25,26] as well as inflammatory disorders and tissue injury [27-29].
Several studies associated an additional supply of antioxidants with lower disease rates and preventive protection by dietary supplementation or topical administration [30-32]. Thus, the supplementation of non-enzymatic antioxidants could be a feasible way of restoring redox homeostasis and reduce ROS-associated diseases. Many of the most powerful and most discussed natural non-enzymatic antioxidants, for example the polyphenols, can be found in the plant kingdom. Polyphenols are naturally occurring compounds found in multiple plants, exhibiting largest antioxidant potential of secondary plant metabolites [33]. Five major groups of phytophenolics are classified, flavonoids, phenolic acids, stilbenes, lignans and tannins [34-36]. A special antioxidative activity has been described for substances from the flavonoid and phenolic acid classes both in vitro and in vivo. They also show a synergistic enhancement when interacting with other antioxidants such as tocopherol and ascorbate [33,37].
In this study, the antioxidant effect of a commercially available plant extract, consisting of extracts from Arnica montana, Bryonia cretica, Guajacum, Toxicodendron quercifolium, Bellis perennis, Ledum palustre, Ruta graveolens and Viscum album was evaluated.
Material and Methods
All experiments were conducted by NIS Labs, Klamath Falls, USA.
Test Substance
The test substance, Flamyar™, is a natural plant extract remedy manufactured according to the german homeopathic pharmacopoeia by PEKANA Naturheilmittel GmbH (Kißlegg, Germany) and distributed in the USA under the name Inflamyar™. The test substance was developed for the treatment of sports injuries, sprains, joint problems, bruises, and muscle strains. Active ingredients are Arnica montana spag. Peka Dil. D12, Bryonia cretica spag. Peka Dil. D4, Guajacum Dil. D4, Toxicodendron quercifolium Dil. D12, Bellis perennis spag. Peka Dil. D8, Ledum palustre Dil. D4, Ruta graveolens spag. Peka Dil. D6, Viscum album spag. Peka Dil. D4.
Isolation of Peripheral Blood Mononuclear and Polymorphonuclear Cells and Erythrocytes
Upon written informed consent from healthy human donors, and as approved by the Sky Lakes Medical Center Institutional Review Board (Federalwide Assurance 2603) for ethical standards, peripheral blood was drawn into heparinized vacutainer tubes. Blood was layered onto Lympholyte-Poly (Thermo Fisher Scientific, Waltham, MA, USA) and centrifuged at 1800 rpm for 35 minutes. Peripheral blood mononuclear cells (PBMC), polymorphonuclear (PMN) cells, and erythrocytes were harvested separately and each cell fraction washed in PBS (Sigma-Aldrich, Saint Louis, MO, USA) by centrifugation at 2400 rpm for 10 minutes. PBMC and PMN cells were washed twice and erythrocytes four times prior to use in assays.
MTT Assay
Freshly purified PBMCs cells were plated in flat-bottom 96- well microtiter plates [NUNC, Roskilde, Denmark] at 2x106 cells/ ml in 90 μl. 10 μl PBS was added to untreated control wells (n=6), and 10μl of serial dilutions of the test substance in PBS added to assay wells (n=3 per group). Cells were incubated at 37°C, 5% CO2 for 24 hours. Then 10μl of Component A from Vybrant® MTT Cell Proliferation Assay Kit (Thermo Fisher Scientific, Waltham, MA, USA) was added, and allowed to incubate for 4 hours. Component B (SDS-HCl) was added, and the plate returned to the incubator for an additional 18 hours. At the end of the incubation, the optical density was measured as the absorbance at 570 nm, using a colorimetric plate reader (BioTek PowerWave, Winooski, VT, USA).
Cellular Antioxidant Protection (CAP-e) Assay
For the cellular CAP-e assay (antioxidant protection of erythrocytes assay), inert human erythrocytes (RBC) were transferred in V-bottom 96-well microtiter plates (NUNC, Roskilde, Denmark), and incubated in a serial dilution of the test substance for 20 minutes (n=2 per group) [38]. During this period, antioxidant compounds could cross the cell membrane and get inside the cells. In order to remove any compounds that were not absorbed by the cells, the erythrocytes were then washed twice with PBS and subsequently loaded with 0.4 μg/ml of the indicator dye 2’, 7’-dichlorofluorescin diacetate (DCF-DA, Thermo Fisher Scientific, Waltham, MA, USA) and 1mg/ml of α,α′-Azodiisobutyramidine dihydrochloride (AAPH, Wako Chemicals, Richmond, VA, USA). DCF-DA becomes fluorescent when oxidized and AAPH is a free radical generator which induces intracellular oxidative stress. The fluorescence was quantified via fluorescence plate reader at 488nm (Tecan Spectrafluor, Tecan, Durham, NC, USA). The fluorescence intensity of untreated cells was defined as baseline, only AAPHtreated cells served as positive control (n=6). A lower fluorescence intensity in cells that were previously exposed to the test product indicates the availability of bioavailable antioxidants in the test substance.
Reactive Oxygen Species Assay
Since they are known to produce high levels of ROS, freshly purified PMN cells were used for the testing of effects of the test substance on reactive oxygen species (ROS) formation [39]. PMN cells (2x105 cells in 200μl per well) were exposed to serial dilutions of the test substance during an incubation period of 20 minutes. The PMN cells were then washed and loaded with the indicator dye DCF-DA (0.5 μg/ml), which turns fluorescent upon exposure to ROS. The ROS formation was triggered by addition of 2mM H2O2 (Sigma-Aldrich, Saint Louis, MO, USA) for 45 minutes. The PMN cells were washed, transferred to cold RPMI-1640 medium (Sigma-Aldrich, Saint Louis, MO, USA), and stored in the dark on ice. Fluorescence intensity was evaluated by flow cytometry using an Attune® acoustic focusing cytometer (Thermo Fisher Scientific, Waltham, MA, USA). The low fluorescence intensity of untreated control cells served as a baseline and PMN cells treated with H2O2 alone served as a positive control. The testing was performed on PMN cells from three different healthy donors, where each dose of the test substance was tested in triplicate.
Statistical Analysis
Averages and standard deviations for each data set were calculated using Microsoft Excel. Statistical analysis was performed using the two-tailed, dependent t-test. Statistical significance was designated as p<0.05, and a high level of significance was designated as p<0.01.
Results
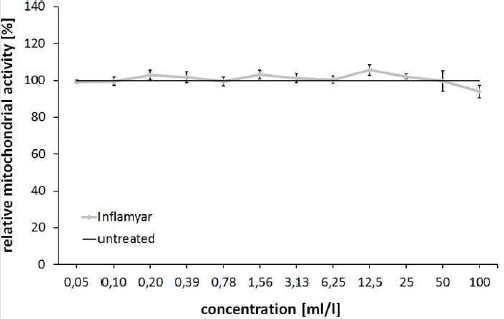
In order to exclude any cytotoxic or cytostatic effects of the test substance, the impact of serial dilutions of the test substance on cell viability of PBMCs were evaluated using MTT Cell-Viability Assay. The data obtained showed no negative effects on cellular viability or mitochondrial function at any of the doses of the test substance; only at the highest concentration of 100ml/l a slightly, but not significantly reduced cell viability was seen (93.95±3.42%, p=0.0756). Highest cell viability was observed after incubation with 12.5ml/l (105.58±2.90%, p=0.1903). All other samples possessed viability data between 99.31±0.86% (0.05ml/l) and 103.18±2.39% (1.56ml/l) compared to untreated controls.
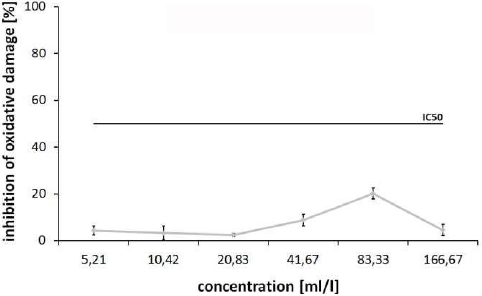
For investigation of the ability for cellular oxidative damage, a Cellular Antioxidant Protection (CAP-e) assay was performed on human red blood cells (RBCs) after incubation with serial dilutions of the test substance. The treatment of cells with the test substance led to an inhibition of oxidative damage at the 41.67ml/l (8.88±2.42%) and 83.33ml/l (20.22±2.39%) doses. Concentrations of 5.21ml/l to 20.83ml/l showed a comparable moderate protection from 2.53±0.59% (20.83ml/l) to 4.38±1.91% (5.21ml/l), and the highest concentration of the test substance used in this experiment (166.67ml/l) led to an inhibition of 4.64±2.36%.
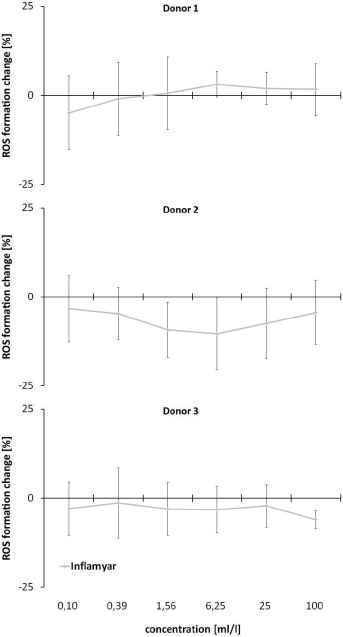
PMN cells from donor 1 showed a reduction of ROS formation at lower concentrations of 0.1ml/l (4,94±10,38%; p=0.542) and 0.39ml/l (0.92±10.29%; p=0.907). Treatment of the cells with test substance concentrations of 1.56 ml/l to 100ml/l led to a ROS formation range between 0.62±10.22% (1.56ml/l; p=0.5480) and 3.15±3.61% (6.25ml/l; p=0.9366). In contrast, reduced levels of ROS formation for all tested concentrations of the test substance were assessed in PMN cells from donor 2 and 3. Cells from donor 2 showed a range of -3.33±9.40% (0.1ml/l; p=0.6950) to -10.38±10.01% (6.25ml/l; p=0.2608). Data from donor 3 were evaluated from -1.30±9.95% (0.39ml/l; p=0.8578) to -6.00±2.45% (100ml/l; p=0.1811).
Discussion
The application of plant extract remedies is widespread among the people. However, the effect of these products is controversially discussed in the scientific community. In the past years, plant extract agents have come more and more into the focus of research and a rising number of studies, showing a positive effect of these remedies can be found in the databases. In contrast to this, different studies conclude that these preparations are not significantly different from placebo treatment [40]. In this study, a plant extract remedy from A. montana, B. cretica, Guajacum, T. quercifolium, B. perennis, L. palustre, R. graveolens and V. album was evaluated in vitro regarding its anti-oxidative capacity on human cells. At the beginning of this study, a MTT cell viability assay was performed on PBMCs in order to get an insight in effects of the test substance on cellular mitochondrial activity, also serving as a baseline for the concentrations used in further experiments. Given the dilution (D4 - D12) of the ingredients, we expected to see little or no cellular stress, even at higher doses, as we typically do with many traditional botanical extracts [41-44]. As expected, the test substance showed no significant reduction of cell viability or mitochondrial function (Figure 1).
Figure 1: MTT Cell-Viability Assay. Viability (relative mitochondrial activity) of PBMCs after 24 hours incubation in serial dilutions of the test substance compared to untreated control cells.
In the next step, the antioxidant potential of the test product was assessed by a Cellular Antioxidant Protection (CAP-e) assay [45,46]. This test allows assessment of antioxidant potential in a method that is comparable to the Oxygen Radical Absorbance Capacity (ORAC) test, but only allows measurement of antioxidants that are able to cross the lipid bilayer cell membrane, enter the cells, and provide biologically meaningful antioxidant protection under conditions of oxidative stress. A mild protection of up to 20% inhibition of oxidative damage in human RBCs at doses of 41.6 to 83.3 ml/l reflects the antioxidant capacity of this product at higher concentrations (Figure 2). Combining data from CAP-e assay with results from ROS assay, some interesting conclusions for the test substance could be made. A large number of antioxidative natural substances are also able to reduce the ROS release in immune cells [27,33]. In contrast, ROS formation is increased in part despite the initial antioxidative effect of a substance through the interaction with the inflammatory signaling pathways of cells. The following three effects of natural substances on the ROS formation of PMN cells are therefore possible:
Figure 2: Cellular Antioxidant Protection (CAP-e) Assay. Inhibition [%] of oxidative damage for the test substance in human RBC after addition of α,α′-Azodiisobutyramidine dihydrochloride (AAPH) to induce intracellular oxidative stress (IC50: half maximal inhibitory concentration).
(1) Direct inhibitory effect on the ROS formation;
(2) Indirect inhibitory effect through activation of an antiinflammatory signaling pathway or
(3) Activation of an inflammatory cascade, which results in an increased release of ROS [27,47].
In this study, three patients were analysed, since due to the current health status of the patients, experiments on immune cells (PMN cells in the ROS assay) of a single patient are not very meaningful. Interestingly, two out of three donors showed a slight but non-significant decrease in reactive oxygen species formation by PMN cells under oxidative stress conditions after pretreatment of cells with all doses of the test substance (Figure 3). This, in combination with the results from CAP-e assay, indicates a triggering of an anti-inflammatory mechanism leading to a reduced ROS-formation at low concentrations of the test substance, whereas the antioxidant capacities at these doses are low. This might be an interesting issue for further analysis of the test substance. In comparison to previous published data, only a few literatures on applications of the active substances contained in the test substance are available. Many of the published data show the effect of undiluted or not in potencies diluted extracts from the herbs. In this context, Gaspar, et al. described no cytotoxicity for isolates from A. montana at low concentrations [42].
Figure 3: Reactive Oxygen Species (ROS) Assay. Evaluation of effects of the test substance under oxidative stress. PMN cells were incubated with serial dilutions of the test substance before adding H2O2 to induce ROS formation.
Other studies also described no or only low cytotoxicity for Arnica-species [43,44]. For other plant species enclosed in the test substance, the information of cytotoxicity is very poor. No data regarding mitochondrial activity was found. Interestingly, an antioxidant capacity was described for each plant enclosed in the test substance: A. montana [43,48], B. cretica [49], Guajacum [50,51], T. quercifolium [52], B. perennis [53,54], L. palustre [55], R. graveolens [56,57] and V. album [58,59]. Moreover, A. montana, members of Bryonia species, Guajacum and R. graveolens were also described to have anti-inflammatory [56,60-63] and immunomodulatory [52,63] capacities. This might be an interesting issue for further analysis, since data from this study also suggest an antiinflammatory effect of the test substance (ROS-inhibition together with low anti-oxidative capacity at low doses). Interestingly, some of the ingredients possess oppositional actions on human cells. For example, T. quercifolium exhibits genotoxic capabilities at higher concentrations, whereas lower concentrations of the same isolate showed anti-genotoxic capacities [64], suggesting a beneficial impact of the dilutions used for the production of the test substance.
Conclusion
This study documented stable vitality and protective capacities against cellular oxidative stress of cells treated with the test substance.
Acknowledgements
Not applicable.
Conflict of Interest
The authors declare that they have no competing interests.
Funding
This work was financially supported by PEKANA Naturheilmittel GmbH (Kißlegg, Germany).
Ethics approval and consent to participate.
All the procedures performed on human cells are approved by the Sky Lakes Medical Center Institutional Review Board (Federalwide Assurance 2603) for ethical standards.
Authors´ Contributions
All experiments were conducted by NIS Labs, Klamath Falls, USA. MS, VS, MH and LH have performed data analysis and interpretation, MS prepared the manuscript. VS, MH, UK and LH critically reviewed the manuscript and contributed intellectual content. All authors read and approved the final version of the manuscript.
References
- Birben E, UM Sahiner, C Sackesen, S Erzurum, O Kalayci (2012) Oxidative stress and antioxidant defense. World Allergy Organ J 5(1): 9-19.
- Krumova, K. and G. Cosa (2016) Chapter 1 Overview of Reactive Oxygen Species, in Singlet Oxygen: Applications in Biosciences and Nanosciences. The Royal Society of Chemistry 1: 1- 21.
- Weiss SJ, AF LoBuglio (1982) Phagocyte-generated oxygen metabolites and cellular injury. Lab Invest 47(1): 5-18.
- Griot C, T Burge, M Vandevelde, E Peterhans (1989) Antibody-induced generation of reactive oxygen radicals by brain macrophages in canine distemper encephalitis: a mechanism for bystander demyelination. Acta Neuropathol 78(4): 396-403.
- Scholz W, K Schutze, W Kunz, M Schwarz (1990) Phenobarbital enhances the formation of reactive oxygen in neoplastic rat liver nodules. Cancer Res 50(21): 7015-7022.
- Cederbaum AI (2001) Introduction-serial review: alcohol, oxidative stress and cell injury. Free Radic Biol Med 31(12): 1524-1526.
- Christen WG, RJ Glynn, CH Hennekens (1996) Antioxidants and age-related eye disease. Current and future perspectives. Ann Epidemiol 6(1): 60-66.
- Jackson CE, WW Bryan (1998) Amyotrophic lateral sclerosis. Semin Neurol 18(1): 27-39.
- Anneren G, A Gardner, T Lundin (1986) Increased glutathione peroxidase activity in erythrocytes in patients with Alzheimer's disease/senile dementia of Alzheimer's type. Acta Neurol Scand 73(6): 586-589.
- Bozner P, V Grishko, SP LeDoux, GL Wilson, YC Chyan, et al. (1997) The amyloid beta protein induces oxidative damage of mitochondrial DNA. J Neuropathol Exp Neurol 56(12): 1356-1362.
- Multhaup G, T Ruppert, A Schlicksupp, L Hesse, D Beher, et al. (1997) Reactive oxygen species and Alzheimer's disease. Biochem Pharmacol 54(5): 533-539.
- Mukherjee SK, JD Adams (1997) The effects of aging and neurodegeneration on apoptosis-associated DNA fragmentation and the benefits of nicotinamide. Mol Chem Neuropathol 32(1-3): 59-74.
- Radunovic A, WG Porto, S Zeman, PN Leigh (1997) Increased mitochondrial superoxide dismutase activity in Parkinson's disease but not amyotrophic lateral sclerosis motor cortex. Neurosci Lett 239(2-3): 105-108.
- Milam SB, G Zardeneta, JP Schmitz (1998) Oxidative stress and degenerative temporomandibular joint disease: a proposed hypothesis. J Oral Maxillofac Surg 56(2): 214-23.
- Stehbens WE (1999) The oxidative stress hypothesis of atherosclerosis: cause or product? Med Hypotheses 53(6): 507-515.
- Nicolas MG, K Fujiki, K Murayama, MT Suzuki, N Shindo, et al. (1996) Studies on the mechanism of early onset macular degeneration in cynomolgus monkeys. II. Suppression of metallothionein synthesis in the retina in oxidative stress. Exp Eye Res 62(4): 399-408.
- Andersson M, J Sjostrand, A Petersen, JO Karlsson (1998) Calcium-dependent proteolysis in rabbit lens epithelium after oxidative stress. Ophthalmic Res 30(3): 157-167.
- Rouhiainen P, H Rouhiainen, JT Salonen (1996) Association between low plasma vitamin E concentration and progression of early cortical lens opacities. Am J Epidemiol 144(5): 496-500.
- Calabrese V, R Raffaele, E Cosentino, V Rizza (1994) Changes in cerebrospinal fluid levels of malondialdehyde and glutathione reductase activity in multiple sclerosis. Int J Clin Pharmacol Res 14(4): 119-23.
- Murphy ME, JP Kehrer (1989) Oxidation state of tissue thiol groups and content of protein carbonyl groups in chickens with inherited muscular dystrophy. Biochem J 260(2): 359-364.
- Rando TA, MH Disatnik, Y Yu, A Franco (1998) Muscle cells from mdx mice have an increased susceptibility to oxidative stress. Neuromuscul Disord 8(1): 14-21.
- Miyata T, N Ishiguro, Y Yasuda, T Ito, M Nangaku, et al. (1998) Increased pentosidine, an advanced glycation end product, in plasma and synovial fluid from patients with rheumatoid arthritis and its relation with inflammatory markers. Biochem Biophys Res Commun 244(1): 45-49.
- DeWeese TL, JM Shipman, NA Larrier, NM Buckley, LR Kidd, et al. (1998) Mouse embryonic stem cells carrying one or two defective Msh2 alleles respond abnormally to oxidative stress inflicted by low-level radiation. Proc Natl Acad Sci U S A 95(20): 11915-11920.
- Meyer TE, HQ Liang, AR Buckley, DJ Buckley, PW Gout, et al. (1998) Changes in glutathione redox cycling and oxidative stress response in the malignant progression of NB2 lymphoma cells. Int J Cancer 77(1): 55-63.
- Tchou J, AP Grollman (1995) The catalytic mechanism of Fpg protein. Evidence for a Schiff base intermediate and amino terminus localization of the catalytic site. J Biol Chem 270(19): 11671-11677.
- Beckman KB, BN Ames (1998) The free radical theory of aging matures. Physiol Rev 78(2): 547-581.
- Mittal M, MR Siddiqui, K Tran, SP Reddy, AB Malik (2014) Reactive oxygen species in inflammation and tissue injury. Antioxid Redox Signal 20(7): 1126-1167.
- Rosanna DP, C Salvatore (2012) Reactive oxygen species, inflammation, and lung diseases. Curr Pharm Des 18(26): 3889-3900.
- Hakim J (1993) Reactive oxygen species and inflammation. C R Seances Soc Biol Fil 187(3): 286-295.
- Wagener FA, CE Carels, DM Lundvig (2013) Targeting the redox balance in inflammatory skin conditions. Int J Mol Sci 14(5): 9126-9167.
- Wannamethee SG, GD Lowe, A Rumley, KR Bruckdorfer, PH Whincup (2006) Associations of vitamin C status, fruit and vegetable intakes, and markers of inflammation and hemostasis. Am J Clin Nutr 83(3): 567-574; quiz 726-727.
- Willcox JK, SL Ash, GL Catignani (2004) Antioxidants and prevention of chronic disease. Crit Rev Food Sci Nutr 44(4): 275-295.
- Kasote DM, SS Katyare, MV Hegde, H Bae (2015) Significance of antioxidant potential of plants and its relevance to therapeutic applications. Int J Biol Sci 11(8): 982-991.
- Blokhina O, E Virolainen, KV Fagerstedt (2003) Antioxidants, oxidative damage and oxygen deprivation stress: a review. Ann Bot 91 Spec No(2): 179-194.
- Myburgh KH (2014) Polyphenol supplementation: benefits for exercise performance or oxidative stress? Sports Med 44(Suppl 1): S57-70.
- Duthie GG, SJ Duthie, JA Kyle (2000) Plant polyphenols in cancer and heart disease: implications as nutritional antioxidants. Nutr Res Rev 13(1): 79-106.
- Croft KD (1998) The chemistry and biological effects of flavonoids and phenolic acids. Ann N Y Acad Sci 854: 435-442.
- Honzel D, SG Carter, KA Redman, AG Schauss, JR Endres, et al. (2008) Comparison of chemical and cell-based antioxidant methods for evaluation of foods and natural products: generating multifaceted data by parallel testing using erythrocytes and polymorphonuclear cells. J Agric Food Chem 56(18): 8319-8325.
- Benson KF, JL Beaman, B Ou, A Okubena, O Okubena, et al. (2013) West African Sorghum bicolor leaf sheaths have anti-inflammatory and immune-modulating properties in vitro. J Med Food 16(3): 230-238.
- Ernst E (2002) A systematic review of systematic reviews of homeopathy. Br J Clin Pharmacol 54(6): 577-582.
- Shah N, R Singh, U Sarangi, N Saxena, A Chaudhary, et al. (2015) Combinations of Ashwagandha leaf extracts protect brain-derived cells against oxidative stress and induce differentiation. PloS One 10(3): e0120554.
- Gaspar A, O Craciunescu, M Trif, M Moisei, L Moldovan (2014) Antioxidant and anti- inflammatory properties of active compounds from Arnica Montana L. Romanian Biotechnological Letter 19(3): 9353-9365.
- Ljubuncic P, H Azaizeh, I Portnaya, U Cogan, O Said, et al. (2005) Antioxidant activity and cytotoxicity of eight plants used in traditional Arab medicine in Israel. J Ethnopharmacol 99(1): 43-47.
- Woerdenbag HJ, I Merfort, CM Passreiter, TJ Schmidt, G Willuhn, et al. (1994) Cytotoxicity of flavonoids and sesquiterpene lactones from Arnica species against the GLC4 and the COLO 320 cell lines. Planta Med 60(5): 434-437.
- Jensen GS, X Wu, KM Patterson, J Barnes, SG Carter, et al. (2008) In vitro and in vivo antioxidant and anti-inflammatory capacities of an antioxidant-rich fruit and berry juice blend. Results of a pilot and randomized, double-blinded, placebo-controlled, crossover study. J Agric Food Chem 56(18): 8326-8333.
- George A, CP Ng, M O'Callaghan, GS Jensen, HJ Wong (2014) In vitro and ex-vivo cellular antioxidant protection and cognitive enhancing effects of an extract of Polygonum minus Huds (Lineminus) demonstrated in a Barnes Maze animal model for memory and learning. BMC Complement Altern Med 14: 161.
- Reuter S, SC Gupta, MM Chaturvedi, BB Aggarwal (2010) Oxidative stress, inflammation, and cancer: how are they linked? Free Radic Biol Med 49(11): 1603-1616.
- Craciunescu O, D Constantin, A Gaspar, L Toma, E Utoiu, et al. (2012) Evaluation of antioxidant and cytoprotective activities of Arnica montana L. and Artemisia absinthium L. ethanolic extracts. Chem Cent J 6(1): 97.
- El Hela AA, NM Abdel Hady, GT Dawoud, AM Hamed, TA Morsy (2013) Phenolic content, antioxidant potential and Aedes aegyptii ecological friend larvicidal activity of some selected Egyptian plants. J Egypt Soc Parasitol 43(1): 215-234.
- Higa T, S Sakemi (1988) Guaiazulene derivatives and their methods of use. Google Patents.
- Sarkar A, P Datta, AK Das, A Gomes (2014) Anti-rheumatoid and anti-oxidant activity of homeopathic Guaiacum officinale in an animal model. Homeopathy 103(2): 133-138.
- Lee KJ, MG Yeo (2016) Homeopathic Rhus toxicodendron has dual effects on the inflammatory response in the mouse preosteoblastic cell line MC3T3-e1. Homeopathy 105(1): 42-47.
- Cingöz G, FP Karakas (2016) The effects of nutrient and macronutrient stress on certain secondary metabolite accumulations and redox regulation in callus cultures of Bellis perennis L. Turkish Journal of Biology 40(6): 1328-1335.
- Karakas FP, AU Turker, A Karakas, V Mshvildadze, A Pichette, et al. (2016) In vitro cytotoxic, antibacterial, anti-inflammatory and antioxidant activities and phenolic content in wild-grown flowers of common daisy-A medicinal plant. Journal of Herbal Medicine 8: 31-39.
- Hassan HM, Jiang ZH, Khaper N, Puukila S, Asmussen C, et al. (2014) Antioxidant activity of Northern Ontario medicinal plants and their protective effect on the H9c2 cardiovascular cells against hydrogen peroxide mediated oxidative stress. Journal of Ethnobiology and Traditional Medicine. Photon 122: 820-831.
- Malik S, DFC Moraes, FMM do Amaral, MNS Ribeiro (2017) Ruta graveolens: Phytochemistry, Pharmacology, and Biotechnology, in Transgenesis and Secondary Metabolism. S Jha, (Edi.), Springer International Publishing: Cham, p. 1-28.
- Mohammadi Motamed S, S Shahidi Motlagh, H Bagherzadeh, S Azad Forouz, H Tafazoli (2014) Evaluation of antioxidant activity of Ruta graveolens L. extract on inhibition of lipid peroxidation and DPPH radicals and the effects of some external factors on plant extract's potency. Research Journal of Pharmacognosy 1(1): 45-50.
- Marvibaigi M, N Amini, E Supriyanto, FA Abdul Majid, S Kumar Jaganathan, et al. (2016) Antioxidant Activity and ROS-Dependent Apoptotic Effect of Scurrula ferruginea (Jack) Danser Methanol Extract in Human Breast Cancer Cell MDA-MB-231. PLoS One 11(7): e0158942.
- Onay Ucar E, O Erol, B Kandemir, E Mertoglu, A Karagoz, et al. (2012) Viscum album L. Extracts Protects HeLa Cells against Nuclear and Mitochondrial DNA Damage. Evid Based Complement Alternat Med 2012: 958740.
- Basu BD, KR Kirtikar, K Basu (1991) Indian Medicinal Plants: Bishen Singh Mahendra Pal Singh.
- Chopra RN, SL Nayar, IC Chopra, LV Asolkar, KK Kakkar, et al. (1956) Glossary of Indian medicinal plants. New Delhi: Council of Scientific & Industrial Research.
- Guarrera M, L Turbino, A Rebora (2001) The anti-inflammatory activity of azulene. J Eur Acad Dermatol Venereol 15(5): 486-487.
- Kriplani P, K Guarve, US Baghael (2017) Arnica montana L. - a plant of healing: review. J Pharm Pharmacol 69(8): 925-945.
- Mersch Sundermann V, F Kassie, S Bohmer, WQ Lu, R Wohlfahrth, et al. (2004) Extract of Toxicodendron quercifolium caused genotoxicity and antigenotoxicity in bone marrow cells of CD1 mice. Food Chem Toxicol 42(10): 1611-1617.

 Research Article
Research Article