Mini Review
Critical limb ischemia (CLI) is the advanced stage of peripheral arterial occlusive disease (PAOD). This stage can be considered as a step prior to impending gangrene and amputation. Recently, it has been demonstrated that the use of stem cells and gene therapy, either alone or in combination in cases of no-option CLI, can provide therapeutic benefits in terms of relieving the rest-pain and healing the ischaemic ulcers. It seems, that although such combinations of treatment are safe, a grey area still exists to whether they are effective in reducing the risk of amputation and improving the quality of life to a reasonable degree. More studies are necessary.
Considerations
Critical limb ischemia is characterized by severe obstruction of
the arteries, reduction of blood flow to the extremities, rest pain and
tissue loss. A step farther stands the no-option CLI, where neither
the use of vasoactive drugs nor the use of open or endovascular
procedures respond well. This implies that a significant percentage
of such patients will have a below or above knee amputation
in the foreseeable future. For such patients, only some “exotic”
interventional and some new non-interventional remedies exist,
which are based on angiogenesis [1-5]. It has been demonstrated
that the use of stem cells alone or in combination with gene therapy
can provide therapeutic benefits in terms of relieving rest-pain and
healing the ischemic ulcers. Main steps when using stem cells are:
a) Harvesting
b) Separation
c) Activation
d) Administration of stem cells to the patient.
The use of stem cells for angiogenesis is based on the principles
of holistic medicine. Various studies have shown that cellular
mediators and tissue-specific chemokines may facilitate selective
recruitment of bone marrow stem and progenitor cells [5-8],
improving blood flow in the limbs, retina and the myocardium,
via astrogenesis. Consequently, the principle for therapeutic
application of stem cells in CLI, is aiming to the restoration of
vascular cellularity and control of the newly formed vessels. End
results are characterized by less pain, better healing of ulcerations
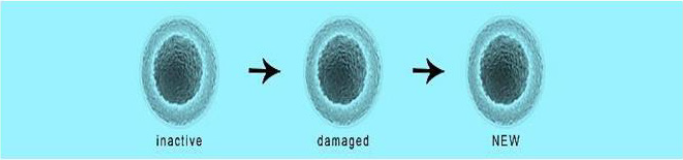
and improved sitting, standing and walking (Figure 1). Aerogenesis
can be considered as the transformation of nonfunctional collateral
arterioles into functional. Evidence showed that stem cells deriving
from human bone marrow can promote arteriogenesis. Two major
factors were used to mobilize these stem cells:
a. Granulocyte colony stimulating factor (G-CSF)
b. Granulocyte-macrophage colony-stimulating factor (GM-CSF).
Figure 1: The aim of the procedure is to activate the non-active mesenchymal stem cells and replace the damaged cells with activated identical new cells.
Therapeutic administration of stem cells does not imply that
their origin is from bone marrow only, but also from adipose
tissues and umbilical cord. Nevertheless, injection of bone marrow
mononuclear cells (BM-MNCs) has significantly improved painfree
walking time, rest pain and tissue oxygen pressure 6 months
after treatment in no-option CLI patients, whereas injection of
mononuclear cells from the peripheral blood, did not lead to similar
effect. These findings suggested that angiogenic-cell-therapy is a
valid choice and is not inferior to the conventional revascularization
therapies in patients with no-option CLI. Stem cells have been used
for therapeutic purposes the last 40 years [9-11]. For example,
bone marrow transplantation is a life-saving procedure for patients
with certain types of bone marrow diseases and malignancies. In
the past, the usefulness of stem-cell-transplantation, was limited
by the fact that some organs (like brain, spinal cord, heart, and
kidney) were thought to lack detectable stem cells. Several recent
discoveries have revolutionized stem cell biology. What are these
discoveries?
(a) Stem-cells have been detected in organs, such as brain and
muscles that were thought to lack regenerative potential.
(b) Similarly, it has been shown that skeletal muscle stem cells
(myoblasts) can be cultured in vitro and then transplanted into
a recipient’s muscle, where they can re-populate the damaged
muscle.
(c) Specific stem cells appeared to display much more plasticity
than originally thought. This implies, that isolated stem-cells
from one tissue can differentiate into a variety of nonrelated
cell-types and tissues.
(d) It has been demonstrated that under appropriate culture
conditions, embryonic stem cells have the capacity for
replication in vitro.
(e) Recently, “hypoxia inducing factors” have been shown to
activate specific pathways, that may control stem-cell selfrenewal.
These factors may prove to be of therapeutic value
since many cancers were considered to be developed from
“self-renewing cancer stem cells”. All these observations have
expanded therapeutic utility of cell-based treatments.
Furthermore, stem-cell therapies, in combination with gene
therapies, may prove to be useful in the treatment of a variety of
human diseases. In particular, certain vascular groups of patients
with very poor outflow and other comorbidities, cannot be
subjected to any revascularization procedure. Similarly, patients
who had several failed revascularization attempts, cannot be
considered as good candidates for a new open or endovascular
surgical procedure [11-13]. These subgroups of patients, represent
a population with the highest rate of limb loss and death. In such
patients, angiogenesis through the administration of stem cells
alone or in combination with new gene therapies, may become
a standard strategy. Gene therapies involve a vast new field for
treating inherited and acquired diseases. This kind of therapy
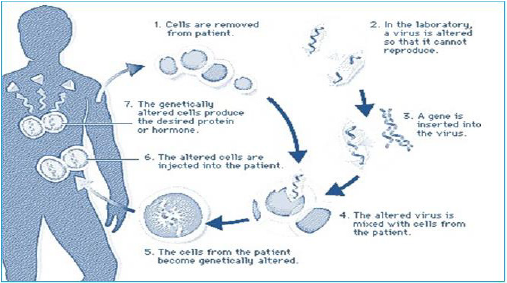
implies the genetic modification of cells to produce a therapeutic
effect (Figure 2). Such genetic modification can be carried out either
in cultured cells in vitro, which then are administered to the patient,
or may involve modification of cells, in vivo. Most approaches to
gene therapy involve 3 interacting components:
a) The presence of a therapeutic gene
b) A vector that allows delivery of the gene to the appropriate
cell, and
c) A device (i.e. a catheter or stent) for delivering the gene/vector
complex to the appropriate tissue in vivo.
(Figure 3). Finally, combined clinical trials of stem and gene
therapy have demonstrated that vascular endothelial growth factor
(VEGF) and fibroblast growth factor (FGF) can be delivered as
proteins or DNA. The results still are under consideration, because
some gray areas appeared (i.e., insufficient duration of the exposure
of the ischemic tissue to the growth factors in order to promote the
production of stable vessels) [14,15]. The development of more
efficient gene transfer systems will probably make these novel
therapies more effective in improving the symptoms of CLI. Future
plans for using such therapies
a) Combination of stem cells and gene therapy in no-option CLI.
b) Combination of embryonic stem-cell and gene therapy in
ischaemic heart disease
c) Combination of stem cells and gene therapy in chronic
ischaemic colitis.
d) Prevention of acute mesenteric ischemia, by using
combinations of stem-cell and gene therapy.
We have to keep in mind, that gene and stem-cell therapies raise a number of important ethical issues and concerns. Some of them, are common to any new therapy involving human experimentation. Others are more unique to specific methods used in gene and stem cell therapeutics. Nevertheless, such ethical debates will continue to be an important determinant of the progress of similar therapies in the future.
References
- Chochola M, Pytlik R, Kobylka P, L Skalická, L Kideryová, et al. (2008) Autologous intra- arterial infusion of bone marrow mononuclear cells in patients with critical leg ischemia. Int Angiol 27(4): 281-290.
- Matoba S, Tatsumi T, Murohara T, Tsutomu Imaizumi, Yousuke Katsuda, et al. (2008) Long-term clinical outcome after intramuscular implantation of bone marrow mononuclear cells (Therapeutic Angiogenesis by Cell Transplantation [TACT] trial) in patients with chronic limb ischemia. Am Heart J. 156(5): 1010-1018.
- Amann B, Luedemann C, Ratei R, J André Schmidt-Lucke (2009) Autologous bone marrow cell transplantation increases leg perfusion and reduces amputations in patients with advanced critical limb ischemia due to peripheral artery disease. Cell Transplant 18(3): 371-380.
- Fadini GP, Agostini C, Avogaro A (2010) Autologous stem cell therapy for peripheral arterial disease met analysis and systematic review of the literature. Atherosclerosis 209(1): 10-17.
- Rigato M, Monami M, Fadini GP (2017) Autologous Cell Therapy for Peripheral Arterial Disease: Systematic Review and Meta- Analysis of Randomized, Nonrandomized, and uncontrolled Studies. Circ Res 120(8): 1326-1340.
- Bura A, Planat-Benard V, Bourin P, Jean-Sebastien Silvestre, Fabian Gross, et al. (2014) Phase I trial: the use of autologous cultured adipose-derived stroma/stem cells to treat patients with non-revascularizable critical limb ischemia. Cytotherapy 16(2): 245-257.
- Makarevich P, Rubina KA, Diykanov DT, V A Tkachuk, Ye V Parfyonova, et al. (2015) Therapeutic Angiogenesis Using Growth Factors: Current State and Prospects for Development 55(9): P: 59-71.
- Bompais H, Chagraoui J, Canron X, Mihaela Crisan, Xu Hui Liu, et al. (2004) Human endothelial cells derived from circulating progenitors display specific functional properties compared with mature vessel wall endothelial cells. Blood 103(7): 2577-2584.
- Wu X, Zhao Y, Tang C, Tieying Yin, Ruolin Du, et al. (2016) Re-endothelialization Study on Endovascular Stents Seeded by Endothelial Cells through Up- or Downregulation of VEGF. ACS Appl Mater Interfaces 8(11): 7578-7589.
- Lu XW, Idu MM, Ubbink DT (2006) Mata-analysis of the clinical effectiveness of venous arterialization for salvage of ischaemic limbs. Eur J Vasc Endovasc Surg 31(5): 493-499.
- Alexandrescu V, Ngongang C, Vincent G (2011) Deep calf veins arterialization for inferior limb preservation in patients with extended ischaemic wounds, unfit for direct arterial reconstruction (preliminary results) Cardiovasc Revasc Med 12(1): 10-19.
- Schreve MA, Vos CG, Vahl AC (2017) Venous arterialization for salvage of ischemic limbs: a systematic review and meta-analysis. Eur J Vasc Endovasc Surg 53(3): 387-402.
- Kum S, Tan YK, Schevre MA, Ferraresi R (2017) Midterm outcomes from a pilot study of percutaneous deep veinarterialization for the treatment of no-option ischemia limb ischemia. J Endovasc Ther 24(5): 619-626.
- Gandini R, Merolla S, Scaggiante J, Marco Meloni, Laura Giurato, et al. (2018) Endovascular distal plantar vein arterialization in dialysis patients with no-option critical limb ischemia and posterior tibial artery occlusion. J Endovasc Ther 25(1): 127-132.
- Migliara B, Cappellari TF (2018) A novel technique to create an arteriovenous fistula during total percutaneous deep foot venous arterialization using an IVUS guided catheter. Eur J Vasc Endovasc Surg 55(5): 735.

 Mini Review
Mini Review