Abstract
Objective: To study the parameters of respiration of mitochondria of rat brain
homogenates with its total and subtotal ischemia.
Methods: The studies used models of total and subtotal cerebral ischemia. Cerebral
ischemia was modeled under conditions of intravenous thiopental anesthesia (40-
50mg/kg). Total cerebral ischemia was modeled by decapitation of animals. Subtotal
cerebral ischemia was modeled by simultaneous ligation of both common carotid
arteries. The sampling of material for the study of tissue respiration of mitochondria was
carried out 1 hour and 24 hours after decapitation or ligation. To study mitochondrial
respiration, the brain was removed in the cold (0-4°C), dried with filter paper, weighed
and homogenized in an isolation medium containing 0.32 M sucrose, 10 mM Tris-HCl,
1 mM EDTA, pH 7.4 (in the ratio 1:10) using a Potter-Evelheim homogenizer with a
Teflon pestle according to the modified method.
Results: An increase in V1 and V2 and a decrease in the phosphorylation coefficient
(ADP/O) indicates proton transfer bypassing the ATP synthase complex. Enzymes
of the mitochondrial matrix and cytochrome in this model of cerebral ischemia do
not yet have pronounced damage, as evidenced by the high rates of V1 and V2. More
pronounced disturbances with the use of succinate than with the use of malate/
glutamate indicate a greater damage to the succinate dehydrogenase complex of the
electron transport chain, as in the case of total cerebral ischemia.
Conclusion: The most pronounced decrease in the respiration indices of the
mitochondrial fraction of brain homogenates occurs in total cerebral ischemia due to
the complete cessation of the blood supply to the brain neurons. With this method of
modeling cerebral ischemia, the appearance of hyperchromic shriveled neurons with
pericellular edema is characteristic.
Keywords: Mitochondrial Respiration; Neurons; Cerebral Ischemia
Introduction
Energy exchange in the cell is associated with mitochondria, which play an important role in vital processes, participating not only in the formation of ATP, but also in the storage and transmission of hereditary information, apoptosis and plastic processes [1-3]. Mitochondria are very mobile and plastic organelles that constantly change their shape, have the ability to fusion and subsequent separation. The movement of mitochondria in the cytoplasm is associated with microtubules, which determines their orientation and distribution in the cell. In some cells, mitochondria form long mobile filaments or chains, while in others they are fixed near the places of consumption of ATP [4-6]. Neurons need a constant supply of ATP for their stability and maintaining the level of potassium ions K + inside the cell, and sodium and calcium ions outside. At rest, the brain consumes up to 20% of the oxygen received by the body. Under normal conditions, effective biological oxidation is the main source of energy-rich phosphate compounds required for the renewal of structures corresponding to the functional activity of cells [5-7]. Elucidation of the mechanisms of development of energy deficiency in ischemic damage is advisable for detailing the pathogenesis, the ratio of damage and compensation processes in this pathology.
Materials and Research Methods
The experiments were carried out on 88 male outbred white
rats weighing 260 ± 20 g in compliance with the Directive of
the European Parliament and of the Council No. 2010/63/EU
of 22.09.2010 on the protection of animals used for scientific
purposes. The studies used models of total (TCI) and subtotal
(SCI) cerebral ischemia. Cerebral ischemia (CI) was modeled under
conditions of intravenous thiopental anesthesia (40-50 mg/kg).
Total cerebral ischemia was modeled by decapitation of animals.
Subtotal cerebral ischemia was modeled by simultaneous ligation
of both common carotid arteries (CCA). The sampling of material
for the study of tissue respiration of mitochondria was carried
out 1 hour and 24 hours after decapitation or ligation of the CCA.
The control group consisted of sham-operated rats of the same
sex and weight. To study mitochondrial respiration, the brain was
removed in the cold (0-4°C), dried with filter paper, weighed and
homogenized in an isolation medium containing 0.32 M sucrose,
10 mM Tris-HCl, 1 mM EDTA, pH 7.4 (in the ratio 1:10) using a
Potter-Evelheim homogenizer with a Teflon pestle according to the
modified method [8]. Mitochondria were isolated by differential
centrifugation.
The nuclear fraction was separated by centrifugation at 600g
for 10min (4°C). The resulting supernatant was centrifuged at
8500g for 10min (4°C), the mitochondrial pellet was washed
twice in the isolation medium and resuspended to a protein
concentration of 35-40mg/ml in the isolation medium and stored
in a short tube on ice. Protein concentration was determined
by the Lowry method. To study mitochondrial respiration, a
concentrated suspension of mitochondria was introduced into a
thermostated sealed polarographic cell with an incubation medium
in an amount providing a final protein concentration in the cell
of 1mg/ml. The incubation medium for recording mitochondrial
respiration includes 0.17 M sucrose, 40 mM KCl, 10 mM Tris-HCl, 5
mM KH2PO4, 8 mM KHCO3, 0.1 mM EDTA, pH 7.4. The principle of
operation of a 3.0 ml polarographic cell is based on the registration
of oxygen uptake by mitochondria using a built-in Clarke electrode
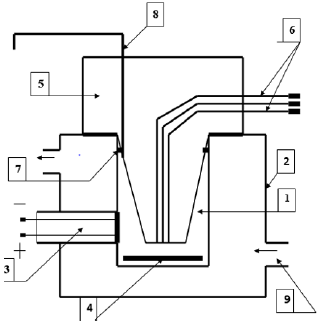
at a temperature of 25°C (Figure 1).
Figure 1: Polarographic cell for studying the respiratory activity of mitochondria
1. Cell;
2. Thermostatically controlled chamber;
3. Clarke electrode;
4. Mmagnetic stirrer;
5. Sealing plug;
6. Channels for dosed anaerobic administration of substrates and ADP;
7. A sealing ring;
8. Channel for removing air and excess liquid;
9. Fitting for connection to an ultrathermostat.
Registration of changes in oxygen tension (pO2) in the
mitochondrial suspension was carried out using an electronic
recorder KSP-4. The Clarke electrode was calibrated by successively
blowing air (pO2 of air) and gaseous nitrogen (pO2 = 0 mmHg)
through the cell. After recording the rate of basal (endogenous)
respiration in the absence of a substrate (V1), respiration substrates
(malate - 2 mM/glutamate - 5 mM or succinate - 5 mM) are alternately
introduced into the mitochondrial suspension, and then ADP in an
amount of 200 nmol/ml. The obtained polarograms are used to
calculate the respiration rate of mitochondria in different metabolic
states and the coefficients characterizing the conjugation of
oxidation and phosphorylation processes. The following indicators
of mitochondrial respiration were recorded: V1 - the rate of basal
respiration, V2 - the rate of substrate-dependent respiration, V3-
- the rate of respiration coupled with phosphorylation (after the
addition of ADP), V4 - the rate of respiration after the completion of
phosphorylation of the added ADP.
The indicators characterizing the conjugation of oxidation and
phosphorylation processes in mitochondria were determined: the
acceptor control coefficient (AK = V3/V2), the respiratory control
coefficient (DC = V3/V4) and the phosphorylation coefficient -
ADP/O. The use of solutions of substrates of the complex “malate/
glutamate” and succinate makes it possible to assess the degree
of functional activity of the electron transfer chain (ETC) in
mitochondria in general, in particular I and II of the ETC complex
[9,10]. To prevent systematic measurement errors, brain samples
from the compared control and experimental groups of animals
were studied under the same conditions. As a result of research,
quantitative continuous data were obtained. Since the experiment
used small samples that had an abnormal distribution, the analysis
was carried out by methods of nonparametric statistics using the
licensed computer program Statistica 10.0 for Windows (StatSoft,
Inc., USA). Data are presented as Me (LQ; UQ), where Me is the
median, LQ is the value of the lower quartile; UQ is the upper quartile
value. Differences between groups were considered significant at
p<0.05 (Kruskell-Wallis test with Bonferoni’s correction).
Results
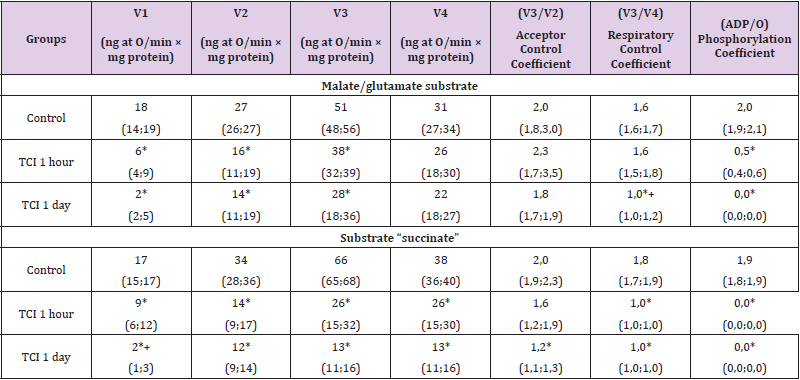
Compared with the control, at 1-hour TCI in the presence of the substrate “malate/glutamate”, which characterizes the state of the first (NADH-dehydrogenase) complex, the electron transport chain V1 decreased by 65 (58; 67)%, p<0.05, V2 - by 41 (38; 48)%, p<0.05, V3 - by 25 (22; 38)%, p<0.05, and the phosphorylation coefficient - by 78 (71; 84)%, p<0,05. The rest of the indicators (V4, the coefficient of acceptor control and the coefficient of respiratory control) did not change (p>0.05), (Table 1). In the presence of succinate substrate, which reflects the work of complex II (succinate dehydrogenase) of the electron transport chain, a decrease in the parameters of energy exchange was established: the rate of basal respiration V1 – by 44 (38; 52)%, p<0.05, the rate of substratedependent respiration V2 – by 60 (48; 64)%, p<0.05, respiration rate associated with V3 phosphorylation - by 59 (38; 65)%, p<0.05, respiration rate after completion of phosphorylation of added V4 ADP – by 32 (28; 46)%, p<0.05 [9].
Table 1: Respiration indices of the mitochondrial fraction of rat brain homogenates with total cerebral ischemia when using the substrates “malate/glutamate” and “succinate”, Me (LQ; UQ).
Note: p<0.05 compared with the control group, + – p<0.05 compared with 1-hour TCI, TCI - total cerebral ischemia.
The respiratory control coefficient (V3/V4) decreased by 45
(42; 48) % (p<0.05), the phosphorylation coefficient (ADP/O) at
1-hour CTI was zero. The acceptor control coefficient (V3/V2) did
not change (p>0.05). The decrease in the rate of basal respiration
V1 was more pronounced when using the substrate succinate (by
21%, p<0.05), which indicates a greater damage to the II complex
(succinate dehydrogenase) of the electron transport chain (ETC)
during TCI. There were no differences between other indicators
(p>0.05). Under conditions of 1-day TCI in the presence of the
substrate “malate/glutamate” at 1-day TCI, V1 decreased by 90 (82;
96) %, p<0.05, V2 – by 46 (42; 55) %, p<0,05, and V3 – by 45 (39;
56) % (p<0.05), the respiratory control coefficient – by 35 (31; 42)
% (p<0.05). Indicators V4 and the coefficient of acceptor control
did not change (p>0.05). The phosphorylation coefficient (ADP/O)
when using both succinate and malate/glutamate with 1-day TCI,
as with 1-hour TCI, was equal to zero. Compared to 1-hour TCI, the
respiratory control coefficient with 1-day TCI was 41 (35; 47) %
less (p<0.05).
There were no differences in other indicators (V2, V3¬, V4,
acceptor coefficient and phosphorylation coefficient) (p>0.05),
(Table 2). In the presence of succinate, there was a more significant
decrease in V1 than with 1-hour TCI - by 90 (84; 95) %, p<0.05,
V2 - by 65 (54; 70) %, p<0.05, V3 - by 78 (54; 87) %, p<0.05, V4
- by 67 (59; 81) %, p<0.05. The respiratory control coefficient
decreased by 45 (41; 49) %, p<0.05. Compared with 1-hour TCI, the
V1 index was 80 (75; 86) % less (p<0.05) Changes in mitochondrial
respiration in relation to the control level when using both
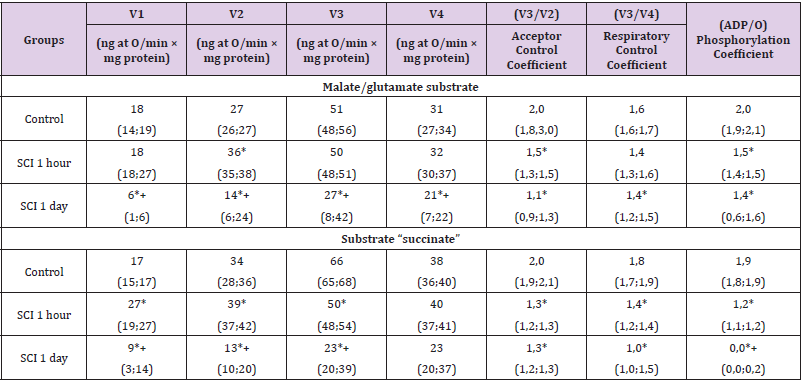
substrates were equivalent (p>0.05). In the SCI group lasting 1
hour, compared with the control group, in the presence of malate/
glutamate, V2 increased by 24 (18; 27) %, p<0.05, and the acceptor
control coefficient and phosphorylation coefficient decreased by
25 (17; 29) %, p<0.05. Other indicators (V1, V3, V4, respiratory
control coefficient) did not change (p>0.05). In the presence of
the substrate “malate/glutamate” at 1-hour SCI, the mitochondrial
respiration indices V1, V2, V3 and V4 were 89 (82; 93) % higher
than at 1-hour TCI, p<0.05, 58 (55; 63) %, p<0.05, 24 (21; 29) %,
p<0.05 and 32 (27; 37) %, p<0.05, respectively [10-19].
Table 2: Respiration indices of the mitochondrial fraction of rat brain homogenates in subtotal cerebral ischemia when using the substrates “malate/glutamate” and “succinate”, Me (LQ; UQ).
Note: p<0.05 - in relation to the control level, + – p<0.05 compared to 1-hour SCI, SCI - subtotal cerebral ischemia
The respiratory control coefficient did not change (p>0.05),
while the acceptor control coefficient was 34 (24; 43) % less,
p<0.05, and the phosphorylation coefficient was 66 (58; 73) %
more, p<0.05. The decrease in the phosphorylation coefficient
with SCI was less significant by 53% (p<0.05). In the presence of
succinate substrate, an increase in the rate of basal respiration
V1 was noted - by 38 (34; 42) %, p<0.05, the rate of substratedependent
respiration V2 - by 13 (9; 18) %, p<0.05, rate respiration
associated with V3 phosphorylation - by 26 (21; 32) %, p<0.05.
These changes indicate a significant decoupling of oxidation and
phosphorylation. The respiration rate after the completion of
phosphorylation of the added ADP (V4) did not change (p>0.05).
At the same time, the acceptor control coefficient, the respiratory
control coefficient and the phosphorylation coefficient decreased by 35 (31; 39) %, p<0.05, 20 (18; 28) %, p<0.05 and by 36 (30;
41) %, p<0.05, respectively, which indicates a decrease in energy
production.
Compared with 1-hour TCI, with 1-hour SCI in the presence
of the substrate “succinate” the rates V1, V2¬, V3 and V4 were 67
(62; 71) % higher, p<0.05, 64 (58; 68) %, p<0.05, 46 (39; 52) %,
p<0.05 and 35 (31; 41) %, p<0.05, respectively. The respiratory
control coefficient increased by 30 (24; 36) %, p<0.05. The
phosphorylation coefficient at 1-hour TCI was zero. When using
succinate, the decrease in the respiratory control coefficient was
less pronounced with SCI (by 10%, p<0.05). When using succinate,
when using malate/glutamate, in relation to the control level, the
phosphorylation coefficient was lower by 11% (p<0.05). Other
indicators (V1, V3¬, V4, respiratory control coefficient, acceptor
control coefficient) did not differ (p>0.05). An increase in V1 and V2
and a decrease in the phosphorylation coefficient (ADP/O) indicates
proton transfer bypassing the ATP synthase complex. Enzymes of
the mitochondrial matrix and cytochrome in this model of CI do not
yet have pronounced damage, as evidenced by the high rates of V1
and V2, however, a decrease in the coefficients of acceptor control,
respiratory control and phosphorylation indicates the separation
of oxidation and phosphorylation processes and a decrease in the
production of ATP during SCI.
More pronounced disturbances with the use of succinate than
with the use of malate/glutamate indicate a greater damage to the
succinate dehydrogenase complex of ETC, as in TCI. In the presence
of the substrate “malate/glutamate” at 1-day SCI, compared with
1-day TCI, V1 decreased by 66 (60; 71)%, p<0.05, V2 - by 45 (41;
50)%, p<0.05, V3 - by 47 (39; 52)%, p<0.05, V4 - by 34 (27; 39)%,
p<0.05, which is more significant than with 1-hour SCI by 87 (72;
94) %, p<0.05, 61 (58; 73)%, p<0.05 and by 46 (41; 52)%, p<0.05,
respectively, except for the value of the V4 indicator, which did not
change, p>0.05. The coefficients of acceptor control, respiratory
control and phosphorylation decreased by 42 (37; 51) %, p<0.0;
by 12 (9; 18) %, p<0.05 and by 25 (21; 32) %, p<0.05, respectively.
In the presence of the “malate/glutamate” substrate, the basal
respiration rate V1 was 67% higher (p<0.05), the acceptor control
coefficient was 39% lower (p<0.05), and the phosphorylation
coefficient at 1-day TCI was zero. Under the conditions of daily SCI
in the presence of succinate substrate, a decrease in V1 was noted
- by 47 (39; 51)%, p<0.05, V2 - by 62 (54; 66)%, p<0.05, V3 - by 64
(59 ; 68)%, p<0.05, which is more pronounced than with 1-hour SCI
by 67 (62; 72)%, p<0.05; by 66 (63; 74)%, p<0.05 and by 55 (49;
59)%, p<0.05, respectively.
The acceptor control coefficient and the respiratory control
coefficient decreased by 35 (29; 41) %, p<0.05 and by 44 (38;
49) %, p<0.05, respectively. The phosphorylation coefficient with
1-day SCI, as well as with 1-day TCI, was equal to zero. Compared
with TCI lasting 1 day, with 1-day SCI in the presence of succinate
substrate, the V1 rate was 78% higher (p<0.05), and the V3 and V4
rates were higher by 43% (p<0.05). while other parameters did not
change (p>0.05). The use of a mixture of malate with glutamate as
a substrate for 1-day SCI showed similar changes in mitochondrial
respiration parameters as with succinate, with the exception
of a higher phosphorylation coefficient - 1.4 (0.6; 1.6), p<0,05.
The decrease in the V1, V2, and V3 indices with 1-day SCI is a
consequence of a decrease in the oxygen content for mitochondrial
respiration.
The suppression of energy processes was more pronounced than
with 1-hour SCI, which reflects an extremely low phosphorylation
coefficient. Changes in V1, V2 and V3 indicators at 1-hour SCI
and 1-hour TCI were multidirectional. Their increase in SCI is
associated with the uncoupling of oxidation and phosphorylation,
while a decrease in TCI is associated with a lack of substrates
for mitochondrial respiration. The decrease in the rate of basal
respiration V1 with 1-day SCI was less pronounced than with TCI:
in the presence of succinate - by 43% (p<0.05), and in the presence
of malate/glutamate - by 24% (p<0.05).
Conclusion
During cerebral ischemia, damage to the inner mitochondrial
membrane occurs due to the activation of free radical oxidation
processes [5]. Damage to the inner mitochondrial membrane, in
turn, leads to an increase in its permeability and a decrease in the
level of the proton gradient due to the transition of protons along
the concentration gradient through the formed nonspecific pores
into the mitochondrial matrix [13,19,18]. As a result, the efficiency
of ATP synthesis decreases, and to maintain the intermembrane
potential under these conditions, more substrates and oxygen
are required [14-18]. Thus, the most pronounced decrease in
the respiration indices of the mitochondrial fraction of brain
homogenates occurs in total cerebral ischemia due to the complete
cessation of the blood supply to the brain neurons.
With this method of modeling cerebral ischemia, the
appearance of hyperchromic shriveled neurons with pericellular
edema is characteristic. In their cytoplasm, the destruction of
organelles occurs, the disintegration of neurofibrils and neuropil,
which indicates their low functional activity. Simultaneous subtotal
ischemia also leads to severe irreversible damage to neurons:
at the morphological level, this is manifested by a significant
increase in the number of hyperchromic shriveled neurons. Their
predominance in the population of neurons in the SCI group
corresponds to the inhibition of respiration of the mitochondrial
fraction of brain homogenates.
References
- Bon LI (2020) Morphology of rat brain neurons in subtotal ischaemia and introduction of L-NAME and omega-3 polyunsaturated fatty acids. Journal of Medical Science, p. 1-8.
- Hoffmann C (2018) The effect of differentiation and TGFß on mitochondrial respiration and mitochondrial enzyme abundance in cultured primary human skeletal muscle cells. Journal of Science Report, pp. 737-740.
- Holvoet P (2017) Low Cytochrome Oxidase 1 Links Mitochondrial Dysfunction to Atherosclerosis in Mice and Pigs. Journal of PLoS One, pp. 307-312.
- Boumans H, Grivell LA, Berden JA (1998) The respiratory chain in yeast behaves as a single functional unit. Journal of Biology Chemistry 273(9): 4872-4877.
- Brand MD, Murphy MP (1987) Control of electron flux through the respiratory chain in mitochondria and cells. Journal of Biological Reviews 62(2): 141-193.
- Hackenbrock CR (1981) Lateral diffusion and electron transfer in the mitochondrial inner membrane . Journal of Trends Biochemistry, pp. 151-154.
- DePierre JW, Ernster L (1988) Enzyme topology of intracellular membranes. Journal of Review Biochemistry 46: 201-261.
- Maksimovich N Ye (2019) Structural and functional features of mitochondria and methods of their study in experiment. Medicus, p. 8-18.
- Casey RP (1984) Membrane reconstruction of the energy-conserving enzymes of oxidative phosphorylation. Journal of Biochemistry Acta, pp. 319-347.
- Chalmers GR (1992) Adaptability of the oxidative capacity of motoneurons / G. R. Chalmers, R. R., Roy, V. R. Edgerton. Journal of Brain Reseaarch 570(1-2): 1-10.
- Baertling F (2018) NDUFA9 point mutations cause a variable mitochondrial complex I assembly defect. Journal of Clinical Genetics 93(1): 111-118.
- Bon LI (2019) Methods of estimation of neurological disturbances in experimental cerebral ischemia. Biomedicine, p. 69-74.
- Bon LI (2021) Histological disorders of neurons of phylogenetically different parts of the cerebral cortex in partial, subtotal, stepwise subtotal, and total cerebral ischemia. Journal of Medical Science 90(1): 108-115.
- DePierre JW, Ernster L (1988) Enzyme topology of intracellular membranes. Journal of Review Biochemistry 46: 201-261.
- Klinyerberg M (1985) Principles of carrier catalysis elucidated by comparing two similar membrane translocators from mitochondria, the ADP/ATP carrier and the uncoupling protein. Journal of New York Academic Science 456: 279-288.
- Leonard K, Haiker H, Weiss H (1987) Three-dimensional structure of NADH: ubiquinone reductase (complex I) from Neurospora mitochondria determined by electron microscopy of membrane crystals. Journal of Моlecular Biology 194: 277-286.
- Prince RC (1988) The proton pump of cytochrome oxidase. Journal of Trends Biochemistry Science, pp. 159-160.
- Slater ЕС (1983) The Q Cycle, a ubiquitous mechanism of electron transfer. Journal of Trends Biochemistry Science 8(7): 239-242.
- Srere PA (1982) The structure of the mitochondrial inner membrane-matrix compartment . Journal of Trends Biochemistry Science, pp. 375-378.

 Research Article
Research Article