ABSTRACT
Background: Hypophosphatemia causes a decrease in bone strength, fractures and pain in adulthood. However, it is not associated with the onset or progression of osteoarthritis (OA) of the knee.
Case Presentation: A 47-year-old man was diagnosed with hypophosphatemia and started receiving treatment five years previously. The patient had developed progressive pain in the right knee after four and half years of treatment and underwent an opening-wedge high-tibial osteotomy (HTO) for OA of the knee. At 18 months postoperatively, he had almost no symptoms in the right knee.
Conclusion: Open-wedge HTO with successful bone fusion was performed in a relatively young patient with OA of the knee and hypophosphatemia.
Keywords: Osteoarthritis; Hypophosphatemia; Opening-Wedge High-Tibial Osteotomy
Abbreviations: XLH: X-Linked Hypophosphatemia; OA: Osteoarthritis; HTO: High Tibial Osteotomy; HSS: Knee-Rating Scale of Hospital for Special Surgery
Introduction
Phosphorus is an essential element in all living cells and has various functions [1-3]. The phosphate balance is a complex interplay between phosphate uptake and excretion. The extracellular to intracellular shift of phosphate and decreased reabsorption of phosphate in the intestinal tract and kidney are considered mechanism that cause hypophosphatemia [1,2,4]. Mild hypophosphatemia is relatively rare, and several causes may often overlap in patients with severe hypophosphatemia [5]. Familial tumoral calcinosis, X-linked hypophosphatemia (XLH), tumor-induced osteomalacia and chronic kidney disease are typical diseases that cause hypophosphatemia [6-10]. Although hypophosphatemia causes decreased bone strength in adulthood, it is not associated with the onset or progression of osteoarthritis (OA) of the knee. OA of the knee in relatively young patients is often secondary to previous trauma. We present the case of a relatively young patient with hypophosphatemia who underwent openingwedge high-tibial osteotomy (HTO) for OA of the knee.
Case
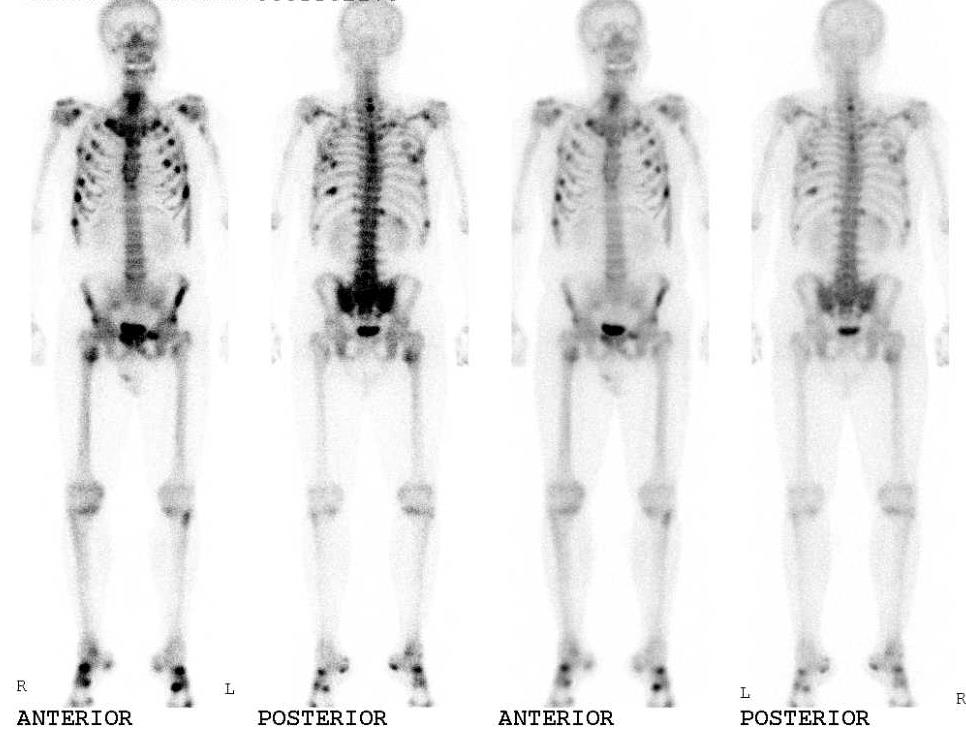
A 47-year-old man presented with progressive pain in the right knee and gait disturbance. The patient had developed progressive pain in his right hip five years previously, and laboratory test results had revealed a phosphate level of 0.58mmol/L. Bone scintigraphy had demonstrated high uptake of technetium-99m in many bones, including the proximal tibia (Figure 1). He was diagnosed with hypophosphatemia and was prescribed oral supplements of phosphate and vitamin D for the treatment of hypophosphatemia. Thereafter, he experienced relief in the hip joint symptoms. However, he developed progressive pain in the right knee four and a half years later. He was diagnosed with OA of the knee and received hyaluronic acid injections; however, the treatment was ineffective, and his right knee pain worsened. Therefore, the patient was referred to our department for surgery. He had a history of mild medial collateral ligament (MCL) injury in the right knee in adolescence. However, he had no symptoms in the knee until six months previously. The patient’s family history was unremarkable. Physical examination revealed swelling and tenderness of medial joint line in the right knee. The range of motion was from10° to 140°. The McMurray test for the medial meniscus showed negative results. The Hospital for Special Surgery (HSS) knee rating scale score was 74.
Figure 1: Bone scintigraphy had demonstrated high uptake of technetium-99m in many bones, including the proximal tibia.
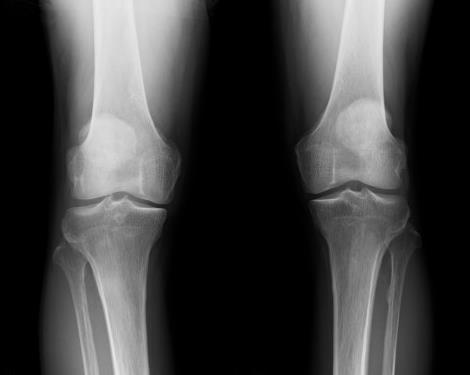
Figure 2: Weight-bearing long leg plain film radiography showed Kellgren-Lawrence OA grade 2 and the anatomical lateral distal femoro-tibial angle (FTA) was 181°.
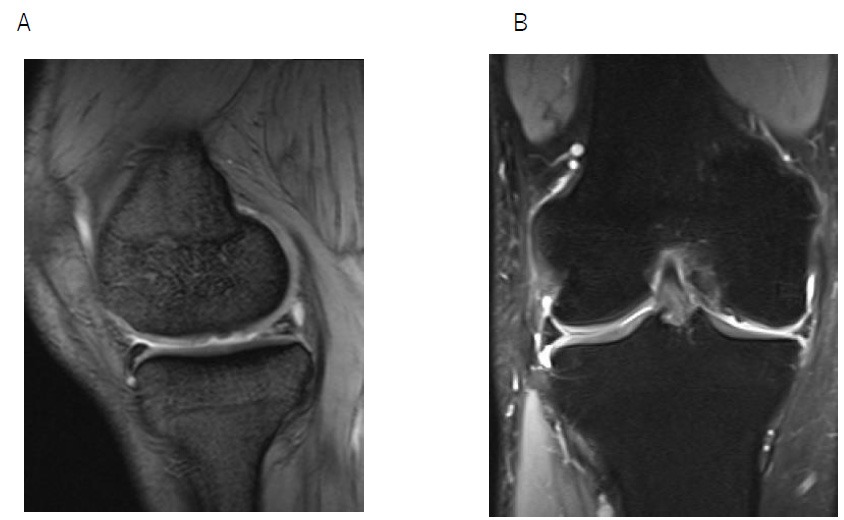
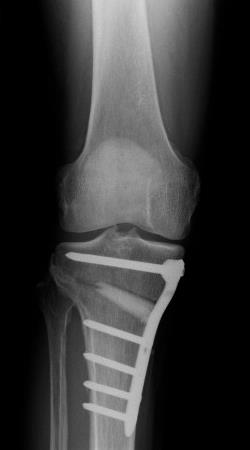
The Rosenberg view showed grade 2 OA according to the Kellgren-Lawrence classification, and the anatomical lateral distal femorotibial angle was 181° (Figure2). Magnetic resonance imaging revealed degeneration of the medial meniscus, slight joint effusion, an edematous area, and articular cartilage defect in the medial femoral cartilage (Figures 3A & B). The patient was diagnosed with OA of the right knee and opening wedge HTO was performed using TomoFix Japanese and a synthetic bone substitute (Figure 4). A knee brace was placed on the right leg for one week. In addition, the patient was instructed to avoid weight bearing for 1 week. Range of motion (ROM) exercises were started 1 week postoperatively. Full weight-bearing was allowed at 4 weeks. The bone fusion had been successful. At 18 months postoperatively, he had almost no symptoms in the right knee. The ROM was from 0° to 145°, and the HSS scale score was 97.
Figure 3: Preoperative magnetic resonance imaging (right knee)
A. T1-weighted image, sagittal view
B. T2-weighted, fat suppression image, coronal view.
Discussion
In the musculoskeletal system, phosphate is associate with bone structure and striated muscle metabolism [11-13]. Hypophosphatemic osteomalacia causes bone deformities and growth and movement disorders during the growth phase. In adults, decreased bone strength causes fractures and pain [2,14]. Our patient developed mild hypophosphatemia in adulthood. Mild hypophosphatemia is not associated with the onset or progression of knee OA. In contrast, XLH typically causes hypophosphatemia. The most common symptom in adults with XLH is joint pain, which can be due to the development of enthesopathy and osteoarthritis, including thinning of the cartilage and sclerosis of the subchondral bone [3,6]. The Hyo mouse, a murine homolog of XLH, is known to have the same signs as humans with XLH, including hypophosphatemia [15]. Liang et al. reported that degenerative osteoarthropathy characterized by decreased articular cartilage thickness, increased chondrocyte alkaline phosphatase activity, defective mineralization, and vascular invasion of the cartilage, was prominent in these mice [14,15]. Therefore, degenerative osteoarthropathy may occur in patients with mild hypophosphatemia. Our patient had a history of an MCL injury in the right knee.
However, he had not experienced pain in the knee until six months prior to surgery. The past MCL injury might have been involved in the development of knee OA. With regard to the effect of hypophosphatemia to bone structure, bone scintigraphy demonstrated mildly elevated uptake of technetium-99m in both proximal tibiae. However, the patient had no symptoms in both knee until six months prior to surgery. Radiography revealed no deformity of the right proximal tibia. Thus, bone deformities that occur because of hypophosphatemia might not have been involved. Therefore, in our relatively young patient, degenerative medial meniscus changes and decreased articular cartilage thickness might be due to the past MCL injury and mild hypophosphatemia. Phosphate supplementation is indicated for the treatment of hypophosphatemia in symptomatic patients [16]. The safest mode of therapy is the oral route. Overzealous intravenous phosphate therapy for hypophosphatemia causes a precipitous decrease in serum calcium concentration. Liamis, et al. reported that oral phosphate can be administered as tablets of sodium or potassium phosphate at doses of 2.5-3.5g daily [17]. In our patient, hypophosphatemia was corrected with monitoring of serum phosphate levels and renal function, and mild hypophosphatemia did not affect bone fusion after HTO. Currently, 1200mg of phosphate and 3㎍of vitamin D are being administered orally, and the correction is under control.
Conclusion
HTO with successful bone fusion was performed in a relatively young patient with OA of the knee and hypophosphatemia. Past MCL injury and mild hypophosphatemia may be associated with the onset or progression of knee OA.
Ethics Approval and Consent to Participate
Not applicable
Human and Animal Rights
No animals/humans were used for studies that are the basis of this research.
Standard Reporting
CARE guidelines have been used for conducting this research.
Acknowledgement
Declared none.
Consent for Publication
We have obtained consent from the participant to publish this study with comprehensive consent.
Competing Interests
The authors declare that they have no competing interests.
Availability of Data and Material
Not applicable.
References
- Gaasbeek A, Meinders AE (2005) Hypophospatemia: an update on its etiology and treatment. Am J Med 118: 1094-1101.
- Amanzadeh J, Reilly RF Jr (2006) Hypophosphatemia: an evidence-baced approach to its clinical consequences and management. Nat Clin Pract nephrol 2(3): 136-148.
- Weisinger JR, Bellorin Font E (1998) Magnesium and phosphorus. Lancet 352: 391-396.
- Knochel JP (1985) The clinical status of hypophosphatemia: an update. N Engl J Med 313(7): 447-449.
- Surbramanian R, Khardori R (2000) Severe hypophosphatemia. Pathophysiologic implication, clinical presentations, and treatment. Medicine (Baltimore) 79(1): 1-8.
- Carpenter TO, Imel EA, Holm IA, Jan de Beur SM, Insogna KL (2011) A clinician’s guide to X-linked hypophosphatemia. J Bone Miner Res 26(7): 1381-1388.
- Tejeda A, Saffrarian N, Uday K, Dave M (1996) Hypophosphatemia in end stage renal disease. Nephron 73(4): 674-678.
- Ix JH, Anderson CA, Smits G, Persky MS, Block GA (2014) Effect of diatary phosphate concertraction in chronic kidney disease: a crossover study. Am J Clin Nutr 100: 1392-1397.
- Folerenzano P, Hartley IR, Jimenez M, Gafni RI, Collins MT (2021) Tumor-induced Osteomalasia. Calcif Tissue Int 108(1): 128-142.
- Hewison M (1994) Tumor-induced Osteomalacia. Curr Opin Reumatol 6(4): 340-344.
- Chudley AE, Ninan A, Young GB (1981) Neurologic signs and hypophosphatemia with total parenteral nutrition. Can Med Assoc J 125(6): 604-607.
- Ravid M, Robson M (1979) Proximal myopathy caused by latrogenic phosphate depletion. JAMA 236(12): 1380-1381.
- Singhal PC, Kumar A, Deroches L, et al. (1978) Hypophosphatemia and rhabdomyolysis. J Clin Invest 62(6): 1240-1246.
- Liang G, Katz LD, Isonaga KL, Carpenter TO, Macica CM (2009) Survey of enthesopathy of X-linked hypophosphatemia and its characterization in Hyp mice. Calcif Tissue Int 85(3): 235-246.
- Liang G, Vanhouten J, Macica C M (2011) An atypical degenerative osteoarthropathy in Hyp mice is characterized by a loss in the mineralized by a loss in the mineralized zone of articular cartilage. Calcif Tissue Int 89(2): 151-162.
- Steele A, Gonzalez R, Garbalosa JC, Keith Steigbigel, Tania Grgurich, et al. (2020) Osteoarthritis, osteophytes, and enthesophytes affect biomechanical function in adults with X-linked hypophosphatemia. J Clin Endocrinol Metab 105(4): 1798-1814.
- Liamis G, Milionis HJ, Elisaf M (2010) Medication-induced hypophosphatemia: a review. Q J Med 103(7): 449-459.

 Case Report
Case Report