ABSTRACT
Lipoproteins are large particles of lipids stabilized by apolipoproteins and they transport lipids in the plasma within the body by specific oriented pathways according to the kind of the lipoproteins. Apolipoproteins have structural and functional roles: they serve as recognition molecules for membrane receptors and are essential partners for enzymes (LPL, LCAT and CETP) that are involved in the metabolism and exchange of lipids. The composition of lipoproteins is slightly different between dogs, cats, horses and humans: VLDLs containing the ApoB48 molecule are identified in dogs and horses. The HDL lipoproteins are the major lipoproteins in animals, but the spherical triglyceride-rich HDL particles are virtually absent because the CETP activity is low. By limiting triglyceride exchanges between lipoproteins, these compositional differences impact the prevalence of atherosclerosis in animals in which the vascular lesions occur secondary to other endocrine and metabolic disorders.
Keywords: Lipoproteins; Apolipoproteins; HDL; Dog; Cat; Horse; Atherosclerosis
Abbreviations: HDL: Protein-Rich Lipoproteins; LDL: Low-Density Lipoproteins; VLDL: Very-Low-Density Lipoproteins; HDL: High-Density Lipoproteins; IDL: Intermediate-Density Lipoproteins; LPL : Lipoprotein lipase; LCAT: Lecithin Cholesterol Acyl Transferase; CETP: Cholesteryl Ester Transfer Protein
Introduction
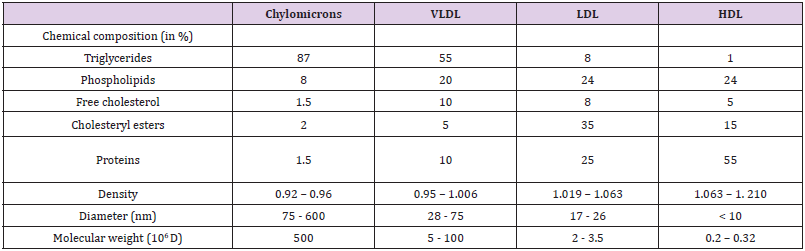
Lipoproteins are aggregates of proteins with lipids that serve in the lipid transport and distribution in blood plasma. The main physical properties of lipoproteins, namely density, size and molecular weight are directly related to their respective proportions of lipids and proteins that are the basis for the particle classification. Four major types of lipoproteins have been described in blood in humans and in domestic animals: chylomicrons secreted by the intestinal mucosa, Very-Low-Density Lipoproteins (VLDL) formed by the liver and progressively converted into Low-Density Lipoproteins (LDL), and High-Density Lipoproteins (HDL) formed by the liver and other organs. The triglyceride-rich chylomicrons and triglyceride-rich VLDL are characterized by a low density, d < 0.96 and d < 1.006 respectively, whereas the Protein-Rich Lipoproteins (HDL) exhibit the highest density, comprised between 1.063 and 1.21 in humans (Table 1) [1-3]. The distinct kinds of lipoproteins are involved in oriented distribution and metabolism of lipids and particularly of cholesterol (free or esterified) within the organism both because of a tissue-specific assembly and distinct recognition pathways. Basically, chylomicrons, produced by the intestinal mucosa from dietary fat and secreted into lymph, deliver fatty acids to muscle and adipose tissue after the endothelial lipoprotein lipase action and bring dietary cholesterol to the liver. In a second step, triglyceride-rich VLDL particles are formed by the liver and are progressively converted in LDL by molecular rearrangements, including gains in apolipoproteins and in cholesteryl esters from HDLs and depletion in triglycerides in the bloodstream.
Table 1: Overall chemical composition and principal physical properties of lipoproteins in humans [1,9,16].
Note: VLDL : Very Low Density Lipoprotein; LDL: Low Density Lipoprotein; HDL: High Density Lipoprotein
The cholesterol-rich LDL particles are later engulfed by peripheral cells throughout a receptor-dependent mechanism whereas the HDL particles made by various cells transport excess cholesterol back to the liver where it is catabolized in biliary acids and eliminated in the feces [1,4]. Apolipoproteins, circulating enzymes and specific receptors are largely implicated in the metabolism and disposal of lipoproteins from blood [1,5]. If one of these components is impaired, dyslipidemia may occur and lead to various lipid deposits in tissues, atherosclerosis and/or cardiovascular diseases in humans [1,5,6]. Except for Intermediate- Density Lipoproteins (IDL) [7], all kinds of lipoproteins with overall same molecular characteristics are found in dogs, cats and horses [8] but surprisingly, primary atherosclerotic dyslipidemia are rarely observed in these domestic animals. The present mini review aims to compare the apolipoproteins components with human molecules, to emphasize some marked differences in cholesterol transport and to correlate the major dyslipidemias occurring in the three animal species to eventual anomalies in the lipoprotein metabolism.
Structures and Composition of Lipoproteins – Roles of Apolipoproteins
Lipoproteins are spherical macromolecules that share a same general structure consisting of a hydrophobic core of triacylglycerols and cholesteryl esters surrounded by an amphipathic shell of apolipoproteins and amphipathic lipids, phospholipids (phosphatidyl-cholines and sphingomyelins) and free cholesterol. The formation of spherical droplets rather than bilayer vesicles is linked to the apolipoprotein components. Indeed, apoproteins contain many segments of α-helix whose polar and/or ionized side chains are markedly aligned along one side and interact with ionized groups of phospholipids or with other polar surrounding molecules and whose non polar side chains are distributed on the opposite face of the helical cylinder and are associated with acyl residues of phospholipids by hydrophobic interactions [1,5]. Therefore, the α-helix segments float on the phospholipid surfaces like tree trunks on a river [5]. Consecutively, the more abundant the apolipoproteins, the more the particles are stable and tend to adopt a discoidal shape like nascent HDLs, while the aggregates are less stable when the lipid core is large, and the amphipathic shell is thin as in chylomicrons and in VLDLs [9].
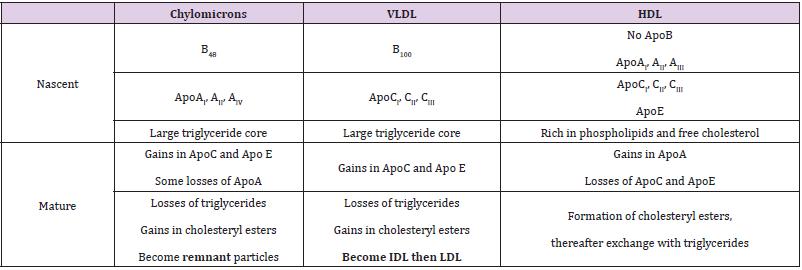
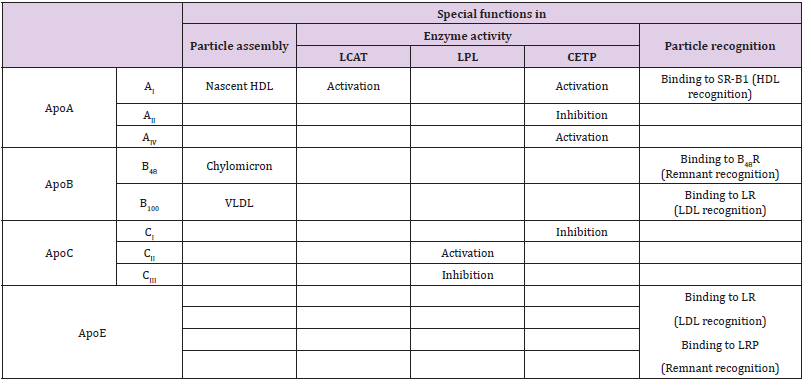
Various apolipoproteins have been identified and they are designated according to the Alaupovic nomenclature by an alphabetical letter followed by a roman numeral in the case of variants [9]. The most common are ApoA (AI, AII, AIII and AIV), ApoB (B48 and B100), ApoC (CI, CII and CIII) and ApoE. Two isoforms of the ApoB exist: B100 and B48. ApoB48, having 48% homology with liver ApoB100, is a truncated form due to alternative spicing of mRNA specifically carried out by the cells of the intestinal mucosa [10,11]. Other apolipoproteins (ApoF, ApoI…) have also been evidenced. Some of these have even been discovered recently such as ApoO [12] but they are scarcely found within lipoproteins and play only minor roles. The distribution of the main apolipoproteins in the different types of lipoproteins is shown in Table 2. In addition to the determining role on the overall shape of lipoproteins, the conformation of apoproteins may also be responsible for their allosteric effects on various enzymes and proteins involved in the fate of the particles (Table 3). For example, the ApoAI conformation influences more or less greatly the LCAT activity that catalyzes the formation of cholesteryl esters by transfer of an acyl group from lecithin to free cholesterol.
Table 2: Distribution of common apolipoproteins within lipoprotein particles and changes in the lipid composition according to their fate in blood [1,9,16].
Note: VLDL: Very Low Density Lipoprotein; IDL: Intermediate Density Lipoprotein; LDL: Low Density Lipoprotein; HDL: High Density Lipoprotein.
Table 3: Special functions of apolipoproteins in metabolism of lipoproteins [9,11,12,14-16,18].
Note: SR-B1: Scavenger Receptor B1: LR: LDL Receptor; LRP: LDL receptor Related Proteins; VLDL: Very Low Density Lipoprotein; LDL: Low Density Lipoprotein; HDL: High Density Lipoprotein; LCAT: Lecithin Cholesterol Acyl Transferase; LPL: Lipoprotein lipase; CETP: Cholesteryl Ester Transfer Protein.
ApoAI is composed of 10 α-helix segments organized into two antiparallel rings linked by disulfide bridges and at least two distinct conformations have been identified. LCAT activation is stronger when ApoAI is in a 5/5 conformation (the 5th segment of the first ring is connected to the 5th segment of the second ring) than when it is in a 5/2 conformation (the 5th segment of the first ring is connected to the 2nd segment of the second ring) [13]. Therefore, a LCAT deficiency as well as a deficit in ApoAI may lead to abnormalities in HDL maturation and indirectly in VLDL blood transformation (see below). The particle attack by lipoprotein lipases from endothelia only occurs when the lipoprotein gains ApoCII, a specific activator of the enzyme, leading to a release of fatty acids by hydrolysis of triglycerides and to a subsequent shrinkage of the lipid core. The triglyceride-rich particles (chylomicrons and VLDL) accumulate in the bloodstream even far from post-prandial periods when the lipoprotein lipase activity fails [14,15]. Plasma CETP (Cholesteryl Ester Transfer Protein) promotes the transfer of cholesteryl esters from HDLs to triglyceride-rich particles in exchange for triglycerides. A low CETP activity is associated with a decrease in LDL cholesterol and also with a disruption in HDL remodeling [16].
Some apolipoproteins also serve as recognition molecules for membrane receptors (Table 3). Several receptors have been identified for the lipoprotein uptake based on the ability to specifically bind some apolipoproteins. The hepatic LRP1 (LDL receptor Related Protein 1) binds ApoE but not ApoB100 and it mediates the uptake of ApoE containing particles, including chylomicron remnants and IDLs but it is not involved in the LDL clearance from blood [17]. The well-known LR (LDL receptor) also called ApoB/E receptor recognizes ApoE and ApoB100 (but not ApoB48) only when its binding domain has become available after progressive transformation of VLDLs. Therefore, it mediates the endocytosis of cholesterol-rich LDLs by peripheral cells [18] and also of chylomicron remnants in the liver to a lesser extent [17,18]. However, chylomicron remnants and LDL receptor-mediated endocytosis can be blocked by the presence of ApoCII (essential for lipoprotein lipase activity) and that is why lipoproteins still rich in triglycerides are not prematurely removed [1]. In addition, the modified LDLs, i.e. glycated or oxidized, become enable to bind to the LR [4,6]. The HDL particles are metabolized in the liver after uptake by a specific receptor SR-B1 (Scavenger Receptor B1) by interacting with ApoAI [19,20].
Apolipoproteins and Lipoprotein Associated Enzymes in Dogs, Cats and Horses (Table 4)
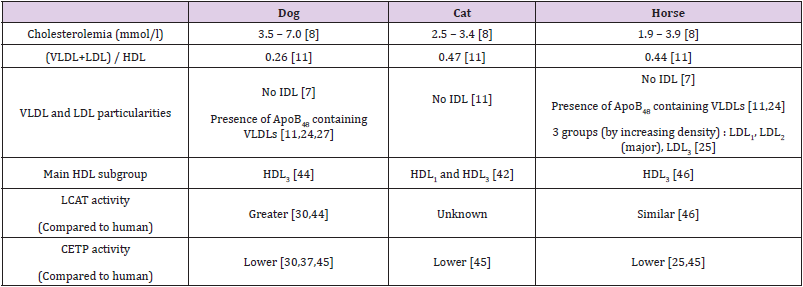
The ApoB100 and ApoB48 have been identified in the domestic animals and are identical to the human proteins [2,11,21-25]. However, the truncated ApoB form is markedly expressed both in the intestine and the liver in the dog and the horse [2,11,23,24,26] contrarily to the human and the cat [21]. The proportions of VLDL containing ApoB48 or ApoB100 are equal in the dog [27]. Although the ApoB48 containing lipoproteins are recognized by specific VLDL receptors [28] with a lower affinity than those containing ApoB100 [29], the VLDL receptor-mediated endocytosis is strong enough to considerably reduce circulating VLDLs [28] and indirectly LDLs in the dog. Consequently, it is considered that lipoprotein particles having ApoB48 are preferentially removed from blood throughout the VLDL receptor whereas those having ApoB100 are progressively transformed into LDL particles and eliminated from blood by the classical LDL capture pathway [24]. Some dog breeds such as beagle and miniature Schnauzer exhibit hypertriglyceridemia associated to a drastic reduction in the LDL/VLDL ratio [30] but the origin of this dyslipidemia is still unknown and the respective proportions of ApoB48 and ApoB100 in VLDLs have not yet been established.
Table 4: Plasma lipoproteins and cholesterol concentrations in dogs, cats and horses.
Note: VLDL: Very Low Density Lipoprotein; IDL: Intermediate Density Lipoprotein; LDL: Low Density Lipoprotein; HDL: High Density Lipoprotein; HDL1: Discoidal nascent HDL; HDL3: Cholesteryl ester-enriched HDL; HDL2: Triglyceride-enriched and cholesterol depleted HDL; LCAT: Lecithin Cholesterol Acyl Transferase; CETP: Cholesteryl Ester Transfer protein.
In horses, as the VLDL particles converge with LDLs in the β zone on electrophoresis, it is difficult to measure their circulating concentrations [31,32]. Nevertheless, three subgroups of LDL particles [33], LDL1, LDL2 and LDL3 respectively named according to an increasing density have been evidenced in horses since 1993 [25]. As the LDL2 fraction is predominant [22,25], it is suggested that the blood LDL maturation is rather slow. The ApoA [23,26] and ApoC [3,23,26] subtypes and ApoE [23] have been characterized in lipoprotein particles in the dog and the horse. These apolipoproteins show strong homology with human proteins despite small differences. In dogs, ApoAIV is smaller than the human homolog [34], the ApoAI genomic sequence has 83% homology with the human DNA sequence although the two proteins have a quite similar structure [35,36]. The ApoAII is quite identical to the human protein [37] and presents a homodimer conformation in dogs and horses like in humans [38]. By contrast, the apolipoprotein sequences are poorly studied in cats. The small number of studies conducted in the species may be linked to the nibbling feeding behavior, leading to impossible measurement of circulating lipoprotein concentrations in the post-prandial periods [39].
The major apolipoprotein in the HDL particles is the ApoAI in animals [22,37] as in humans [19,20]. Indeed, this protein plays an important role in HDL biogenesis in the peripheral cells by association with membrane lipids (free cholesterol and phospholipids) and interaction with the ATP-binding cassette protein A1 (ABCA1) [19]. The resulting nascent particle, called HDL1, adopts a discoidal shape, containing a segment of phospholipid bilayer that is protected from water exposure by two ApoAI molecules arranged in an anti-parallel, double-belt conformation around the edge of the disc [19,20]. Thereafter, because of the action of LCAT activated by the ApoAI, the cholesteryl ester enriched lipid core becomes larger and ovoid, leading to spherical HDL particles called HDL3 stabilized by the flexible ApoAI that bends and forms a surrounding trefoil scaffold structure [19,20]. Finally, the CETP protein mediates exchanges of cholesteryl esters from HDLs and triglycerides from chylomicrons and VLDLs, leading to triglycerideenriched and cholesterol-depleted HDL particles, totally spherical, called HDL2 [40]. The CETP efficiency is markedly high in humans since it would allow the transfer of more than 70% of the newly formed cholesteryl esters [12]. The major lipoproteins in dogs, cats and horses are HDL particles unlike in humans where cholesterol is mainly associated with LDLs [11,25, 41-43]. The proportions of HDL particles are superior to 65% in dogs and cats, and 60% on average in horses in which they can even reach 80% in a few studies [31].
In Cairn Terriers, 82% of cholesterol is transported by HDLs and the hypercholesterolemia of Shetland sheepdogs would be linked to high circulating HDL concentrations [44]. More interestingly, the most commonly encountered HDL particles in dogs have a relatively spherical shape, remain rich in cholesteryl esters and correspond to the human HDL3 subgroup while nascent (HDL1) and triglyceride-rich mature (HDL2) HDL lipoproteins are minor fractions [44]. Two distinct HDL fractions are also identified in cats and are assimilated to nascent discoidal HDLs and to cholesteryl ester rich particles, respectively. As in dogs, triglyceride-enriched particles are scarcely observed [42]. In parallel, a greater HDL associated LCAT activity is found in dogs than in humans [30,44] whereas the CETP efficiency appears depressed in dogs and in cats [30,37,45]. Despite a LCAT activity identical in horses and in humans, HDL3 subgroup particles are predominant in horses [46] and the low frequency of triglyceride enriched HDLs may be related to low CETP activity [25,45].
Major Dyslipidemias and Associated Anomalies in Lipoprotein Metabolism in Dogs, Cats and Horses
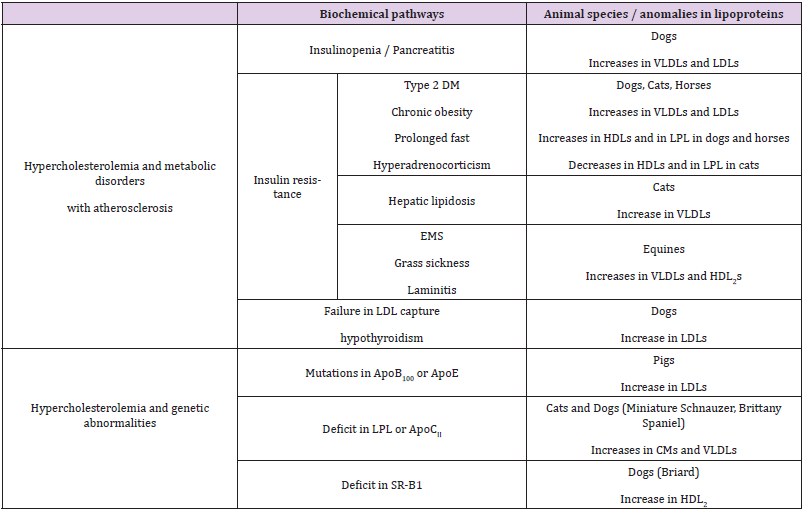
The Table 5 list the major dyslipidemias occurring in animal species and the probable relation with impairment in the fate of lipoproteins. Atherosclerosis in the domestic animals, particularly in dogs, is described secondary to endocrine diseases [47]. First, insulinopenia due to pancreatic failure observed in natural pancreatitis [27,48] or experimentally induced by oleic acid in dogs [35] leads to an exaggerated fatty acid metabolism and consequently to an amplified biogenesis of VLDL lipoproteins mainly containing ApoB100 and ApoAIV by the liver. Insulin resistance occur in type 2 diabetes mellitus in cats, dogs and horses as in humans [49,50] and in chronic obesity [49,51-56] or it is coupled with an exacerbated secretion of hyperglycemic hormones such as glucagon after a prolonged fast and glucocorticoids in primary hyperadrenocorticism [48,49]. In every of these situations, insulin resistance causes excessive lipomobilization throughout transacylase inhibition and lipase activation in adipocytes and thereafter increased hepatic biogenesis of VLDLs later transformed in LDL in bloodstream [48,49]. Increased lipoprotein lipase activity and elevated blood concentrations of HDL particles that provide the allosteric enzyme activator ApoCII molecules are coupled to increased blood VLDLs during chronic obesity in horses [46,57] and dogs [53]. On the other hand, decreases in HDL particles [56,58] and in lipoprotein lipase activity [47] are noted in obese cats.
Table 5: Dyslipidemias and relations with lipoprotein metabolism in domestic animals (dogs, cats and equines).
Note: CM : Chylomicron ; VLDL: Very Low Density Lipoprotein; IDL: Intermediate Density Lipoprotein; LDL: Low Density Lipoprotein; HDL: High Density Lipoprotein; HDL1: Discoidal nascent HDL; HDL3: Cholesteryl ester enriched HDL; HDL2: Triglyceride enriched and cholesterol depleted HDL; LPL: Lipoprotein lipase; SR-B1: Scavenger Receptor B1; DM: Diabetes mellitus; EMS: Equine metabolic syndrome
Cats are particularly susceptible to hepatic lipidosis. This metabolic disease, considered as the second cause of hepatic fat overload, occurs after an episode of anorexia in obese cats and may result from a dietary taurine deficiency [59]. Despite a high capacity to secrete VLDL particles (from 2% of circulating lipoproteins in healthy cats to 19% in diseased animals) [59], fats and especially cholesteryl esters accumulate in hepatocytes, suggesting impairment in cholesterol elimination. While cholic acid can be conjugated with either glycine or taurine for biliary elimination in humans, dogs and horses, only tauryl-cholate exists in cats [60]. Consequently, a lack of taurine or impaired tauryl transferase activity may contribute to cholesterol accumulation in depleted animals. A prolonged fasting induces a severe hyperlipidemia in horses known as equine metabolic syndrome [49] that is coupled with a laminitis predisposal [61]. Hyperlipidemia is caused by increases in blood VLDLs and in mature HDL2 particles [49]. These two specific pathological situations in cats and in equines are often aggravated by a massive dietary fat intake following a fasting period: an increased chylomicron secretion by intestine is added to the previously described abnormalities of the circulating lipoprotein concentrations. The mixed metabolic disorder is called grass sickness in equines [62] and obese donkeys are particularly susceptible to it [52].
By increasing the life span of lipoproteins circulating in the bloodstream, a failure in the LDL capture by peripheral cells promotes chemical modifications (oxidation and glycation) and enhances the risk for atherosclerosis. If genetic mutations of the LDL receptor, leading to familial hypercholesterolemia, are well known in humans, such inherited disorders of the receptor molecules have not been identified in domestic animals. In these later, a low LDL receptor density on cell membranes is related to some endocrine deregulations; as T3 induces the LDL receptor expression, atherosclerosis can complicate hypothyroidism, frequently in dogs and probably also in horses although often confused with equine metabolic syndrome [48]. Additionally, genetic structural abnormalities of the ApoB100 and/or ApoE molecules, that prevent binding to the LDL receptor have been reported in humans and pigs [63,64] but not in pets and horses.
Some genetic alterations in lipoprotein metabolism are scarcely reported in domestic animals. Although more than 50 mutations in the gene coding for the lipoprotein lipase have been detected in humans and though the feline DNA and protein sequences have at least 90% homology with human molecules [39], a single glycine-toarginine substitution at position 412, leading to enzyme deficiency, has been demonstrated in a cat colony [65]. The marked increase in circulating concentrations of chylomicrons and VLDL particles is correlated with clinical deleterious signs such as uveitis (due to the large lipoprotein accumulation in the aqueous humor) [39,65], growth delay (linked to a deficient peripheral utilization of lipids) and pancreatic lesions (resulting from mechanisms of lipotoxicity) [45,66-68]. Some dog breeds such as Miniature Schnauzers [30] or Brittany Spaniels [69], seem genetically predisposed to developing hypertriglyceridemia. Deficits in the lipoprotein lipase activity or in ApoCII molecules are suspected [70]. A few cases of hypercholesterolemia characterized by an increase in mature (HDL2) concentrations have been reported in Briard dogs [49,71]. A decreased expression of SRB1 receptors has been hypothesized.
Conclusion
The same kinds of lipoproteins with identical physical properties are present in humans and in domestic animals although IDL particles have been not identified in dogs, cats and horses. Animal apolipoproteins have a great homology with human molecules and share similar functions in stabilizing lipoprotein particles, regulating activity of enzymes involved in their maturation and recognition by specific membrane receptors. However, two types of VLDL particles, one containing the ApoB48 molecule the other the ApoB100, are identified in the dog and in the horse; the fate of the triglyceride rich particles may depend on the nature of the ApoB molecule. Unlike the humans, the HDL particles are predominant in domestic animals, the major fractions being nascent discoidal and intermediate (rich in cholesteryl esters) spherical particles according to the LCAT activity, especially high in dogs. Mature spherical triglyceride-rich HDL particles are virtually absent in domestic animals because of a low CETP activity. In relation with such specificities in lipoprotein metabolism, atherosclerosis in domestic animals only occurs secondary to endocrine and metabolic disorders, some of them being species-specific, such as hepatic lipidosis in cats and equine metabolic syndrome.
References
- Mc Gilverny RW, Goldstein GW (1983) Chapter 29 Turnover of fats and lipoproteins: the cholesterol connection (3rd)., In: Mc Gilverny RW and Goldstein GW (Eds.)., Biochemistry a functional approach WB Saunders Company, Philadelphia, pp. 555-571.
- Shah AS, Tan L, Jon JL, Davidson WS (2013) Proteomic diversity of high-density lipoproteins: our emerging understanding of its importance in lipid transport and beyond. J Lip Res 54(10): 2575-2585.
- Siri Tarino PW, Krauss RM (2016) The early years of lipoprotein research from discovery to clinical application. J Lip Res 57(10): 1771-1777.
- Bret L, Delcamps C (2020) La circulation des lipides les lipoproté Biochimie structural Cours, fiches et QCM Ellipses 2020. PASS licence santé, pp. 306-356
- Voet D, Voet JG (1998) Biochimie Protéines liées aux lipides et lipoproté Biochimie. In: Voet D and Voet JG (Eds.)., De Boeck university, France, pp. 314-325.
- Bonnet J (2005) Athérosclé EMC Cardiol Angeiol 2(4): 436-458.
- Oda H, Mori A, Hirowatari Y, Takoura T, Manita D, et al. (2017) Cholesterol concentrations in lipoprotein fractions separated by anion-exchange–high-performance liquid chromatography in healthy dogs and dogs with hypercholesterolemia. Res Vet Sci 114: 163-169.
- Kaneko JJ, Harvey JW, Bruss ML (2008) Clinical biochemistry of domestic animals. Academic Press.
- Delignette E (1993) Apoprotéines et nutrition Thèse de pharmacie. Grenoble Université Joseph Fournier.
- Botham KM, Weil A, Rodwell VW, Kennely PJ, Bender DA (2017) Biochimie de Harper De Boeck Superieur.
- Greeve J, Altkemper I, Dieterich JH, Greten H, Windler E (1993) Apolipoprotein B mRNA editing in 12 different mammalian species: hepatic expression is reflected in low concentrations of apoB-containing plasma lipoproteins. J Lip Res 34(8): 1367-1383.
- Lamant M (2006) Caractérisation d’une nouvelle apolipoprotéine humaine, l’Apo O Thèse de pharmacologie. Toulouse Université Paul Sabatier Toulouse III.
- Cooke AL, Morris J, Melchior JT, Street SE, Jerome WG, et al. (2018) A thumbwheel mechanism for APOA1 activation of LCAT activity in HDL. J Lip Res 59(7): 1244-1255.
- Lund-Katz S, Murley YM, Yon E, Gillotte KL, Davidson WS (1996) Comparison of the structural and functional effects of monomeric and dimeric human apolipoprotein A-II in high density lipoprotein particles. Lipids. 31(11): 1107-1113.
- Gong EL, Stoltzfus LJ, Brion CM, Murugesh D, Rubin EM (1996) Contrasting in vivo effects of murine and human Apolipoprotein A-II role of monomer versus dimer. J Biol Chem 271(11): 5984-5987.
- Nakajima K, Nagamine T, Fujita MQ, Ai M, Tanaka A, et al. (2014) Apolipoprotein B-48. In Adv in Clin Chem Elsevier 64: 117-177.
- Cooper AD (1997) Hepatic uptake of chylomicron remnants. J Lip Res 38(11): 2173- 2192.
- Vergès B (2018) Le métabolisme des lipoproté Méd Clin Endocrinol Diab 2: 7.
- Zhou L, Li C, Gao L, Wang A (2015) High density lipoprotein synthesis and metabolism (Review). Mol Med Rep 12(3): 4015-4021.
- Shen WJ, Azhar S, Kraemer FB (2018) SR-B1 a unique multifunctional receptor for cholesterol influx and efflux. Annu Rev Physiol 10 (80): 95-116.
- Yu L, Boyle P, Schneider JA, Segawa E, Wilson RS, et al. (2013) APOE ε4, Alzheimer’s disease pathology, cerebrovascular disease, and cognitive change over the years prior to death. Psychol Aging 28(4): 1015-1023.
- Watson TDG (1991) Lipoprotein metabolism in the Horse. PhD Ann Arbor ProQuest Dissertations & Theses.
- Lalla OF, Gofman JW (2006) Ultracentrifugal analysis of serum lipoproteins. Methods Biochem Anal 1: 459-478.
- Jeusette I, Istasse L, Diez M (2004) Métabolisme lipidique et hyperlipémies chez le chien. Ann Méd Vét 148: 79-89.
- Watson TDG, Packard CJ, Shepherd J (1993) Plasma lipid transport in the horse (Equus caballus) Comp Biochem. Physiol Part B Comp Biochem 106(1): 27-34.
- Xiangdong L, Yuanwu L, Hua Z, Liming R, Qiuyan L, et al. (2011) Animal models for the atherosclerosis research: a review. Protein Cell 2(3): 189-201.
- Chikamune T, Katamoto H, Nomura K, Ohashi F (1998) Lipoprotein profile in canine pancreatitis induced with oleic acid. J Ve Med Sci 60(4): 413-421.
- Dieckmann M, Dietrich MF, Hertz J (2010) Lipoprotein receptor an evolutionarily ancient multifunctional receptor family. Biol Chem 391(11): 1341-1363.
- Hui DY, Innerarity TL, Milne RW, Marcel YL, Mahley RW (1984) Binding of chylomicron remnants and beta-very low-density lipoproteins to hepatic and extrahepatic lipoprotein receptors A process independent of apolipoprotein B48. J Biol Chem 259(24): 15060-15068.
- Xenoulis PG, Steiner JM (2010) Lipid metabolism and hyperlipidemia in dogs. Vet J 183(1): 12-21.
- Leat WMF, Northrop CA, Buttress N, Jones DM (1979) Plasma lipids and lipoproteins of some members of the order perissodactyla. Comp Biochem Physiol Part B Comp Biochem 63(2): 275-281.
- Asadi F, Asadian P, Shahriari A, Pourkabir M, Kazemi A (2011) Serum lipid and lipoprotein patterns of iranian horses. Rev Sci Techn OIE 30(3): 955-960.
- Champain D (2012) Métabolisme des lipoprotéines et biomarqueurs dans les maladies cardiovasculaires. Diplome d’ingénieur CNAM. Lille Conservatoire national des arts et des métiers 30(3): 955-960.
- Weisgraber KH, Bersot TP, Mahley RW (1978) Isolation and characterization of an apoprotein from the d<1.006 lipoproteins of human and canine lymph homologous with the rat A-IV apoprotein. Biochem Biophys Res Comm 85(1): 287-292.
- Swaney JB (1980) Characterization of the high-density lipoprotein and its major apoprotein from human, canine, bovine and chicken plasma. Biochem Biophys Acta 617(3): 489 502.
- Demoor L (1996) Conception d’un modèle synthétique d’apolipoprotéine - Application à l’étude des propriétés physico-chimiques et biologiques d’un domaine de l’apolipoprotéine A-1. Thèse pour l’obtention du titre de Docteur de l’université des sciences et technologie de Lille Université des sciences et technologie de Lille.
- Hollanders B, Mougin A, N Diaye F, Hentz E, Aude X, et al. (1986) Comparison of the lipoprotein profiles obtained from rat, bovine, horse, dog, rabbit and pig serum by a new two-step ultracentrifugal gradient procedure. Comp Biochem Physiol B Comp Biochem 84 (1): 83-89.
- Puppione DL, Fisher WH, Park M, Whitelegge JP, Schumaker VN, et al. (2004) Sequence of horse (Equus caballus) apoA-II. Another example of a dimer forming apolipoprotein. Comp Biochem Physiol Part B Biochem Mol Biol 138(3): 213-220.
- Ginzinger DG, Lewis ME, Ma Y, Jones BR, Liu G, et al. (1996) A mutation in the lipoprotein lipase gene is the molecular basis of chylomicronemia in a colony of domestic cats. J Clin Invest 97(5):1257-1266.
- Eieenberg S (1984) High density lipoprotein metabolism. J Lip Res 25: 1017-1058.
- Chapman MJ (1980) Animal lipoproteins chemistry structure and comparative aspects. J Lip Res 21(7): 789-853.
- Demacker PNM, Van Heijst PJ, Hak Lemmers HLM, Stalenhoef AFH (1987) A study of the lipid transport system in the cat, Felix domesticus. Atherosclerosis 66(2): 113-123.
- Watson TDJ (1996) Lipoprotein metabolism in dogs and cats. Comp Haematol Internat 6(1): 17-23.
- Downs LG, Bolton CH, Crispin SM, Wills JM (1993) Plasma lipoprotein lipids in five different breeds of dogs. Res Vet Sci 54(1): 63-67.
- Guyard Dangremont V, Desrumaux C, Gambert P, Lallemant C, Lagrost L (1998) Phospholipid and cholesteryl ester transfer activities in plasma from 14 vertebrate species. Relation to atherogenesis susceptibility. Comp Biochem Physiol Part B Biochem Mol Biol 120(3): 517-525.
- Coleman MC, Walzem RL, Kieffer AJ, Minamoto T, Suchodolski J, et al. (2019) Novel lipoprotein density profiling in laminitic, obese and healthy horses. Dom An Endocrinol 68: 92-99.
- Liu SK, Tilley LP, Tappe JP, Fox PR (1986) Clinical and pathologic findings in dogs with atherosclerosis: 21 cases (1970-1983). J Am Vet Med Assoc 189(2): 227-232.
- Marz W, Kleber ME, Scharnagi H, Speer T, Zewinger S, et al. (2017) HDL cholesterol reappraisal of its clinical relevance. Clin Res Cardiol 106(9): 663-675.
- Campbell TW, Thrall MA, Allison R, Weiser G (2012) Veterinary Hematology and Clinical Chemistry (2nd)., In: Campbell TW, Thrall MA, Allison R, Weiser G (Eds.)., Wiley-Blackwell, USA.
- Furuhashi M, Hotamisligh GS (2008) Fatty acid-binding proteins role in metabolic diseases and potential as drug targets. Nat Rev Drug Discov 7(6): 489-503.
- Mc Kenzie HC (2011) Equine Hyperlipidemias. Vet Clin North Am Eq Pract 27(1): 59-72.
- Ileri Buyukolu T, Guldur T (2005) Dyslipoproteinemias and their clinical importance in several species of domestic animals. J Am Vet Med Assoc 227(11): 1746-1751.
- Tropf M, Nelson OL, Lee PM (2017) Weng HY Cardiac and metabolic variables in obese dogs. J Vet Intern Med 31(4): 1000-1007.
- Bauer JE, Meyer DJ, Campbell M, Mc Murphy R (1990) Serum lipid and lipoprotein changes in ponies with experimentally induced liver disease. Am J Vet Res 51(9): 1380-1384.
- Bailhache E, Nguyen P, Krempf M, Siliart B, Magot T, et al. (2003) Lipoproteins abnormalities in obese insulin-resistant dogs. Metabolism 52(5): 559-564.
- Hoenig M, Wilkins C, Holson JC Ferguson DC (2003) Effects of obesity on lipid profiles in neutered male and female cats. Am J Vet Res 64(3): 299-303.
- Dreyfus JC (1990) Lipoprotéine lipase et obésité. Méd Sci 6 (6): 586.
- Muranaka S, Mori N, Hanato Y, Saito TR, Lee P, et al. (2011) Obesity induced changes to plasma adiponectin concentration and cholesterol lipoprotein composition profile in cats. Res Vet Sci 91(3): 358-361.
- Valtolina C, Favier RP (2017) Feline hepatic lipidosis. Vet Clin North Am Small Anim Pract 47(3): 683-702.
- Smaili W, Benayad A, Oubaha S, Krati K, Samlani Z (2019) Obésité abdominale et anomalies lipidiques. Résumé de congrè Journées Francophones d’Hépato-gastroentérologie et d’Oncologie Digestive.
- Roquigny C (2020) Master méd La gestion diététique dans le traitement du syndrome métabolique équin: approche théorique et pratique. Université de Liè
- Milne EM, Doxey DL, Gilmour JS (1990) Serum lipids and lipoproteins in equine colic and grass sickness. Res Vet Sci 48(2): 170-174.
- Rabes JP, Varret M, Boileau C (1997) L’hypercholestérolémie familiale 25 ans aprè II- Formes non-liées au récepteur des LDL. Méd Sci 13(12): 1409.
- Hasler-Rapacz JO, Nichols TC, Griggs TR, Bellinger DA, Rapacz J (1994) Familial and diet-induced hypercholesterolemia in swine. Lipid, ApoB, and ApoA-I concentrations and distributions in plasma and lipoprotein subfractions. Arterioscl Thromb J Vasc Biol 14(6): 923-930.
- Ginzinger, Clee, Dallongeville, Lewis, Henderson, et al. (1999) Lipid and lipoprotein analysis of cats with lipoprotein lipase deficiency. Eur J Clin Invest 29(1): 17-26.
- Hoffman JM, Creevy KE, Franks A, O Neill DG, Promislow DEI (2018) The companion dog as a model for human aging and mortality. Aging Cell 17(3):12737.
- Cailloux G (2005) Influence de l’obésité dans l’évolution du diabète sucré chez le chien - Etude bibliographique. Thèse vété Toulouse Université de Paul Sabatier.
- Dubreuil E (2013) Alimentation et athérosclé Thèse de pharmacie. Université de Lorraine.
- Johnson MC (2005) Hyperlipidemia disorders in dogs. Compend. Vet 2: 9.
- Ford RB (1993) Idiopathic hyperchylomicronaemia in miniature schnauzers. J Small Anim Pract 34(10): 488-492.
- Watson P, Simpson KW, Bedford PG (1993) Hypercholesterolaemia in briards in the United Kingdom. Res Vet Sci 54(1): 80 -85.

 Mini Review
Mini Review