ABSTRACT
The administration of a low-carbohydrate, and high-fat ketogenic diet (KD) has been demonstrated to exert beneficial effects on gastrointestinal mucosa in rats. Therefore, the present study was aimed to investigate whether the KD treatment, able to reduce the inflammation at the intestinal level, could also affect rat liver function. After weaning at 5 weeks of age, 12 Wistar rats were fed standard diet (control group), and 13 animals were fed KD. After ten weeks of treatment, animals were sacrificed and the liver from each animal was collected and stored at -80°C until assayed. Fatty acids extracted from liver tissue were quantified by a gas chromatographic method. Western blotting analysis and ELISA method were used to evaluate the inflammation and antioxidant biomarkers in rat liver, respectively. The levels of single saturated fatty acids and monounsaturated fatty acids were significantly decreased in hepatic tissue after KD treatment. A significant decrease was also observed for the content of arachidonic acid, an omega-6 polyunsaturated fatty acid. All main omega-3 polyunsaturated fatty acids, except for eicosapentaenoic acid, were significantly increased in liver tissue from rat treated with KD. Compared to the control group, a significant increase of the SOD 1 and SOD 2 protein enzymes, was found in the liver of KD-treated rats. KD treatment on the one hand promotes the increase of tissue inflammation status in the rat liver, on the other it was able to activate defense mechanisms to protect liver environment.
Keywords: Ketogenic Diet; Wistar Rats; Liver; Fatty Acids Profile
Introduction
The ketogenic diet (KD) ‒ characterized by a low carbohydrate content and high-fat levels ‒induces a metabolic shift to the use of ketones as a source of energy for cells. It is also defined as a carbohydrate-restricted diet with a 4:1 ratio of fats to proteins and carbohydrates [1,2]. Several clinical studies have shown that KD a reduction of midlife mortality and a prolongation of health due to its ability to control body weight and its beneficial effects in the prevention of type 2 diabetes, cancer, and neurological diseases [3- 6]. Recently, in a rat model of irritable bowel syndrome (IBS), KD was able to restore intestinal cell damage through the upregulation of endocannabinoids receptors (CB1-R and CB2-R) [7] improving intestinal permeability. Besides, the KD administration positively affected gastrointestinal mucosa in IBS rats and elicited molecular compensatory mechanisms able to reduce inflammation and oxidative stress in the gut, restoring mitochondrial function and baseline autophagy [7,8].
Literature data demonstrate that KD exerts beneficial effects on subjects with obesity [9,10]. Significant weight loss was associated with reduced visceral fat mass in overweight and obese patients treated with KD [11]. Moreover, consumption of a low-carbohydrate KD is involved in a control blood pressure values and the serum levels of total cholesterol, low-density lipoprotein (LDL), and triglycerides [12]. Besides, literature suggested KD as an effective way to treat non-alcoholic fatty liver disease (NAFLD) [13]. During the improvement of human NAFLD induced by a short-term KD (six days), liver redox status significantly changed. It appears that KD, at least as a short-term treatment, can significantly reduce liver fat content and insulin resistance [13]. However, in rodents, different studies have reported that KD can promote the progression of liver steatosis [14,15]. The greater palatability of high-fat diets justifies the greater food consumption by these animals, consequently increasing body weight [16]. High-fat diets seem to favor the fat accumulation in hepatocytes associated with increased lipid peroxidation metabolites in hepatic tissue [3,16]. Notably, the pro-oxidative environment caused by enhanced levels of dietary lipids, promotes the development and progression of steatosis in the liver [17]. The liver has a prominent role in carbohydrate and lipid metabolisms so that excessive amounts of free fatty acids in hepatocytes cause fatty liver, insulin resistance, and lipotoxicity [18-20].
Fatty acids constitute essential components of membranes that influence their physiological functions [21] and their tissue composition is the result of food intake, lifestyle, and environmental factors [22,23]. Based on this evidence and in line with the existing literature background, the purpose of the present study was to investigate whether a high-fat KD with beneficial effects at intestinal levels may affect liver function in Wistar rats. Through a lipidomic approach, the study was aimed to understand the relationship between KD and fatty acid profile of liver tissue and investigate the clinical implications that occur.
Materials and Methods
Animals and Treatment
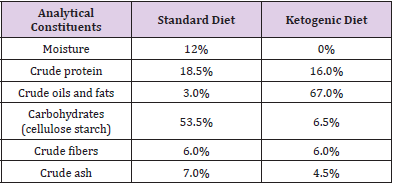
Twenty-five Wistar rats (5 weeks of age) were used in this study. All the animals received care in compliance with the “Guide for the Care and Use of Laboratory Animals” and the experimental procedures were approved by the Italian Ministry of Health (approval date: 28 November 2018, n. 901/2018-PR). The animals, at weaning, were divided at random into two groups and treated for ten weeks. Particularly, 12 animals were fed a standard diet (control group, CTR group), and 13 were fed a low-carbohydrate, high-fat KD (KD group). Diets (4RF21 standard diet and ketogenic diet) were purchased by Mucedola Srl, Settimo Milanese, Italy, and their composition is shown in Table 1. No sign of suffering, stress, or pain was observed in the animals during the experiment. At sacrifice, by anesthetic overdose, the liver from each animal was collected and stored at -80°C until assayed.
Fatty Acids Analysis
Liver tissue fatty acids analysis was carried out using Fatty Acid Extraction Kit Low Standard, according to the manufacturer’s instructions (Sigma-Aldrich, St. Louis, U.S.A.). Briefly, 0.15 g of the liver tissue was homogenized in 3 ml of extraction solvent. After homogenization, 5 ml of aqueous buffer were added, and the sample was filtered by a syringe. The eluted fraction containing the lipid extract was trans-esterified by adding boron trifluoride-methanol solution and hexane and incubated at 95° C for 1 hour. After adding hexane, the samples were vortexed and centrifuged at 500 x g for 5 minutes and the fatty acid methyl esters contained in the top hexane layer were evaporated. The dried pellet was reconstituted with 100 μl of hexane and injected into a gas chromatography equipment (Thermo Fisher Scientific, Milan, Italy). The quantification of fatty acids was carried out as previously described [24].
Western Blotting Analysis
Fifty μg of liver total proteins were subjected to 4-12% SDSPAGE polyacrylamide gel electrophoresis and transferred onto the PVDF membrane (Bio-Rad Laboratories, Milan, Italy). Subsequently, the membrane was incubated overnight at 4°C with the following primary antibodies: NAD-dependent deacetylase sirtuin-1 (SIRT1) (Bio-Rad Laboratories, Milan, Italy), peroxisome proliferatoractivated receptor-γ (PPAR-γ) (Santa Cruz Biotechnology, Santa Cruz, CA, U.S.A.), nuclear factor kappa-light-chain-enhancer of activated B cells (NFκB) (Santa Cruz Biotechnology, Santa Cruz, CA, U.S.A.), tumor necrosis factor (TNF-α) (Immunological Science), human neutrophil cytochrome b-light chain (p22/phox) (Abcam, Cambridge, UK), β-Actin (Cell Signaling Technology, Beverly, MA, U.S.A.). Then, the membrane was washed with TBS-T, and the secondary antibody conjugated with horseradish peroxidaseconjugated rabbit or mouse (Bio-Rad Laboratories) was used, where appropriate. The signal of each protein was visualized using the ChemiDoc™ system (Bio-Rad, Milan, Italy) and the Image Lab software. All data were normalized against β-actin expression.
Determination of Enzymatic Antioxidant Activity and Lipid Peroxidation
The levels of [Cu-Zn] Superoxide dismutase (SOD 1) and the Mitochondrial Superoxide dismutase (SOD 2) in the rat liver tissue were measured with Rat SOD 1 and Rat SOD 2 Enzyme-Linked Immunosorbent Assay (ELISA) Kits (Fine Test, Wuhan, China), respectively. Lipid peroxidation was measured in the rat liver by determining malondialdehyde (MDA), using the Rat MDA Assay Kit ELISA (My Bio Source, San Diego, CA, U.S.A.) by following the manufacturer’s instructions.
Statistical Analysis
Sample size calculation has been performed having a fixed power of 80% and a first type error of α of 0.025 (α ½ = 0.025); then, a minimum number of 6 animals for each experimental group was needed. The statistical differences among the two rat groups were evaluated by the student t-test and they were considered significant with a p-value <0.05.
Results
The Effects of the Ketogenic Diet on Body Weight and Liver Tissue Fatty Acids Profile
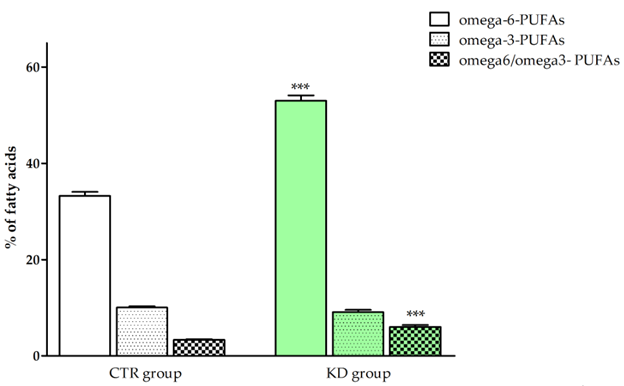
Figure 1: Total levels of omega-3- and omega-6- PUFAs, and the omega-6/omega-3- PUFAs ratio in rat liver tissue of the CTR and KD group. All data are expressed as mean ± standard deviation (SD) of three different experiments. p-Value indicates significant differences among the groups (p < 0.05, Student t-test); *** p< 0.001.
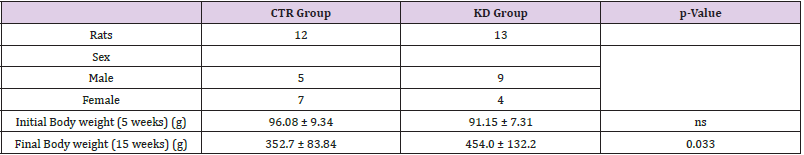
Table 2: Characteristics of experimental rat groups.
Note: All data are expressed as mean ± standard deviation (SD); p-Value indicates significant differences among the groups (p < 0.05, Student t-test); ns: not significant.
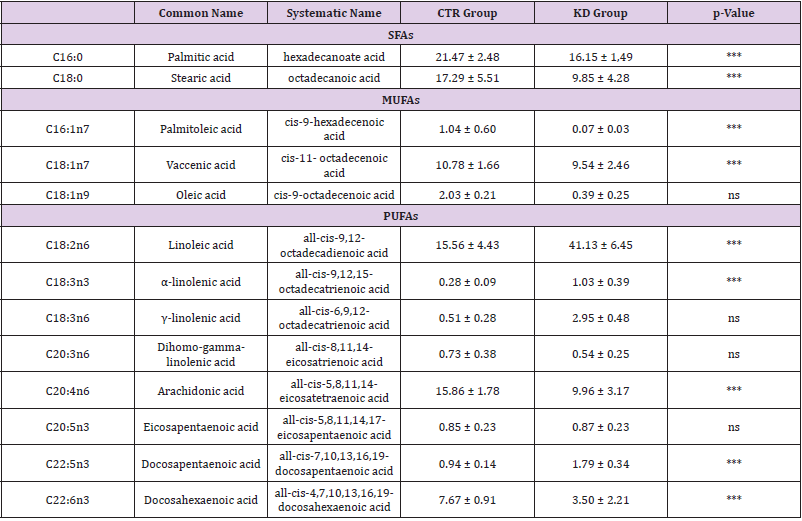
Table 3: Mean percentage of fatty acid composition in liver tissue of the rats treated with the standard diet (CTR group) and KD diet (KD group).
Note: All data are expressed as mean ± standard deviation (SD); p-Value indicates significant differences among the groups (p < 0.05, Student t-test); ns: not significant.
Table 2 shows the characteristics of two experimental rat groups. Compared to the CTR group, a significant increase in body weight was observed in animals after KD treatment (352.7±83.84 vs 454.0±132.2, p=0.033). Interestingly, the administration of KD affected the tissue fatty acids profile in the liver (Table 3). After 10 weeks of KD treatment, the levels of single saturated fatty acids (SFAs), such as palmitic and stearic acid, and single monounsaturated fatty acids (MUFAs) were significantly decreased compared to CTR group. A significant decrease was also observed for the content of arachidonic acid (AA), an omega-6 polyunsaturated fatty acid. In contrast, linoleic acid (LA), an essential omega-6 fatty acid, was significantly increased in liver tissue from KD-fed rats, probably due to the higher oils content in the KD. After KD treatment, all main omega-3 polyunsaturated fatty acids, except for eicosapentaenoic acid (EPA), significantly increased in the liver tissue. Figure 1 summarizes the differences in the levels of total omega-6-, omega-3- Polyunsaturated fatty acids (PUFAs), and the omega-6/omega-3- PUFAs ratio detected between the two groups of rats. In particular, the significant increase in total omega-6- PUFAs justified the increase of omega-6/omega-3- PUFAs ratio observed in the KD group. Saturated fatty acids (SFAs), monounsaturated fatty acids (MUFAs), and polyunsaturated fatty acids (PUFAs). Data are expressed as mean ± standard deviation (SD) of three different experiments; p-Value indicates significant differences among the groups (p < 0.05, Student t-test); *** p<0,0001, ns: not significant.
The Effects of the Ketogenic Diet on Liver Protein Levels
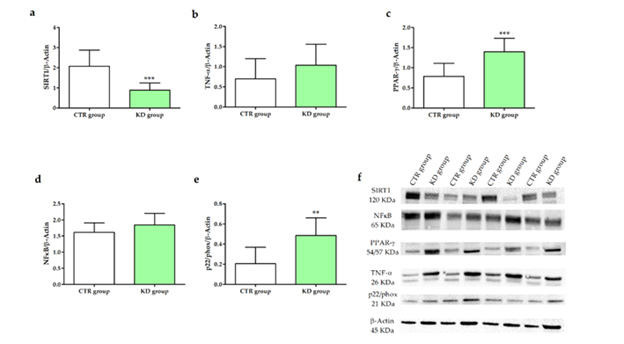
The effects of KD administration on the rat liver levels of SIRT1, TNF-α, PPAR-γ, NFκB, and p22/phox are shown in Figure 2. The group receiving the KD treatment presented higher levels of TNF-α, PPAR-γ, NFκB, and p22/phox protein than the control group. In contrast, a statistically significant reduction in SIRT1 protein levels was observed in rats fed KD. The significant increase of the PPAR-γ and p22/phox protein expression demonstrated that KD caused an inflammatory environment in the liver, which was also justified by a significant reduction in SIRT1 protein.
The Effects of the Ketogenic Diet on Enzymatic Antioxidant Activity and Lipid Peroxidation
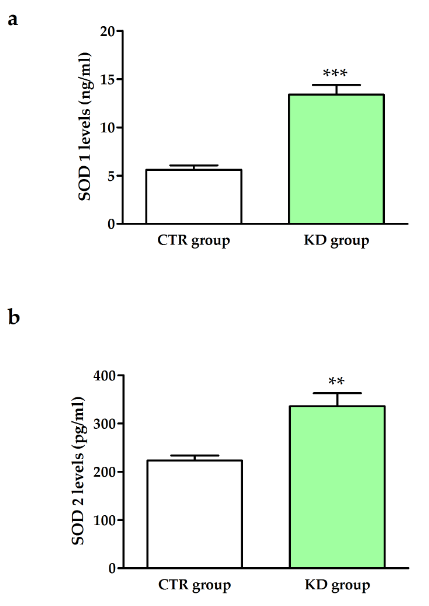
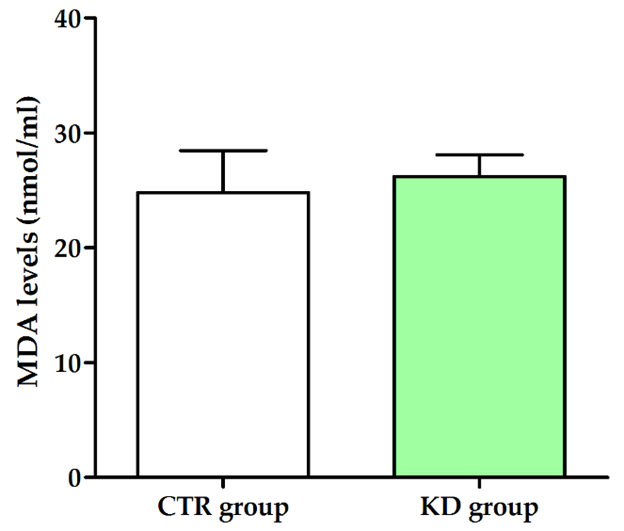
The analysis of enzymatic antioxidant activity based on the determination of SOD 1 and SOD 2 demonstrated that the rats fed KD showed higher value than the controls (Figure 3). No difference was observed in the hepatic MDA levels between the two experimental rat groups (Figure 4), suggesting that the higher degree of inflammation in the liver tissue occurred without significant changes in the oxidative status.
Figure 2: Western blotting analysis of SIRT1 (a), TNF-α (b), PPAR-γ (c), NFκB (d) and p22/phox (e) protein expression, normalized with β-Actin protein, in CTR and KD group. Panel f shows representative blots evaluated in two experimental groups. All data are expressed as mean ± standard deviation (SD) of three different experiments. p-Value indicates significant differences among the groups (p < 0.05, Student t-test); ** p<0.001 and *** p< 0.0001.
Figure 3: SOD 1 (panel a) and SOD 2 (panel b) levels in the CTR and KD groups. All data are expressed as mean ± standard deviation (SD) of three different experiments. ** p<0.001 and *** p< 0.0001.
Figure 4: MDA levels in CTR and KD groups. All data are expressed as mean ± standard deviation (SD) of three different experiments.
Discussion
The effects of KD on liver metabolism and function can be mixed. The present study shows that KD, on the one hand, promotes the increase of tissue inflammation in the liver, on the other, it activates defense mechanisms to protect the liver environment. Significant changes in liver tissue fatty acids profile have been observed after KD intake in rats. Most of these changes can be considered as environmental factors that lead to improving or counteracting tissue inflammation. The anti-inflammatory effects of KD are confirmed by the decrease of the AA content in liver tissue. While the increase of total omega-6-PUFAs detected in rats fed KD is probably due exclusively to the levels of LA derived by the diet. Several studies showed that the high-LA diet could cause no significant increase in AA [25,26]; the LA derived from the diet could be less exposed to enzymatic turnover and, therefore, AA production starting from its precursor is reduced. SIRT1 is known to regulate several downstream pathways eliciting antiinflammatory effects [27,28]. Higher SIRT1 protein levels have been observed during calorie restriction in tissues, as the brain, muscles, liver, and kidneys [29-31]. SIRT1 seems to regulate both liver and adipose tissue inflammation, and its protective function in high-fatinduced liver pathology has been demonstrated in SIRT1+/- mice [32]. Different studies have reported that the expression of SIRT1 is downregulated in most metabolic disorders, including obesity [29,33], demonstrating that the reduction of SIRT1 expression is often linked to an upregulation of pro-inflammatory cytokines, as TNF-α.
Here, KD treatment exerted a reduction in liver SIRT1 protein levels and a concomitant induction of TNF-α, NFκB, and p22/phox protein expression. In a normal condition, NFκB has been observed in the cytoplasm of hepatocytes in a latent form but, when exposed to pro-inflammatory stimuli, it participates in the activation of tissue inflammation [31]. In addition, the upregulation of NFκB was reported to be closely connected with the overexpression of the p22/phox protein [32]. Experimental studies have also shown that all these proteins are linked together, actively intervening in inflammatory processes [34-36]. The significant induction of PPAR-γ, detected in liver tissue of rats fed KD agreed with other studies showing increased PPAR-γ expression following excessive fat intake [37,38]. PPAR-γ is a transcription factor that controls hepatic fatty acids metabolism and, therefore, it results in enhanced hepatic steatosis and abnormal conditions of liver function [39].
Simultaneously, the diet-induced pro-inflammatory state in the rat liver was somewhat offset by increased antioxidant activity, increased total PUFA levels, and a significant reduction in SFA and MUFA. In the present study, the beneficial effect of KD is confirmed by the significant increase in SOD 1 and SOD 2 (Figure 3), in rats fed KD compared to the control. SODs are present in all aerobic living cells since O2 is a common product of oxygen metabolic reactions. SOD 1 is the main intracellular form of SOD and accounts for 80% of the total SOD protein. Previously, some studies declared SOD 1 to be predominantly cytosolic [40]. Nevertheless, later studies have found it in the whole cell, including in the mitochondrial intermembrane space and nucleus [41]. On the other hand, SOD 2 is located exclusively in the mitochondrial matrix (MM) [41]. Together, SODs provide to remove the damaging O2- free radicals from cells and tissues.
In addition to their conventional function as O2- detoxification enzymes, emerging evidence suggests that SOD 1 and SOD 2 are actively implicated in the modulation of several cellular processes [42,43]. One well-recognized mechanism is related to H2O2˗the dismutating product of O2- by SOD˗, can act as a second messenger in growth and metabolic processes regulation [44]. Thus, by increasing SODs, KD can induce antioxidant enzyme activity in liver tissue, probably in response to pro-inflammatory dietary stimuli. The tissue fatty acids profile observed in the present study after KD treatment agrees with experimental evidence underlying the role of SFAs and MUFAs in regulating cell membrane fluidity [45]. A low content of SFAs and MUFAs seems to improve the fluidity of the cell membrane and allows to counteract a possible cell damage, rapidly [45].
Moreover, the therapeutic potential of PUFAs, mainly omega-3 PUFAs, on metabolic liver diseases is well known [46]. EPA and Docosahexaenoic acid (DHA) also seem to exert protective effects against bile acids-induced cell death in human hepatocytes [47,48]. In addition, diet rich in omega-3 PUFAs are recommended for its benefits in reducing platelet adherence [49], lowing cholesterol, and reducing pro-inflammation and pro-oxidation mediators [50]. Several investigations described that the first line of defense of the cell against environmental stresses is the membrane and its content in SFAs, MUFAs, and PUFAs is important for the correct physiological functions [23]. The current study shows the possibility that the KD, through the lipidome, could improve the diet-induced inflammatory state of liver tissue. Herein, this finding implicates a significant and novel role for the tissue lipids profile in regulating complex metabolic pathways in the liver.
Conclusion
The positive effects of KD in rat liver tissue support the role of diet in modulating tissue fatty acids composition and in regulating molecular pathways related to counteracting inflammation. The clinical relevance of the study consists in suggesting a controlled use of KD in the subjects with metabolic disorders, likely for a short period, to avoid a diet-induced inflammatory effect in the liver.
Author Contributions
Conceptualization M.N.; methodology, T.L., G.R.C., V.D.N., M.C., I.G., E.M.; formal analysis, T.L. G.R.C, V.D.N.; data curation, T.L., V.D.N.; writing—original draft preparation, T.L., M.N.; writing—review and editing, T.L., G.R.C., V.D.N., M.C., F.R., M.G.C., V.T.; supervision, M.N. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by R.C. 2020-2021, Prog. No. 15 and 16 (D.D.G. n. 700/2020).
Institutional Review Board Statement
The study was approved by the Italian Ministry of Health (approval date: 28 November 2018, n. 901/2018-PR) according to European Union guidelines (Directive2010/63/EU for animal experiments).
Data Availability Statement
Data are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lutas A, Yellen G (2013) The ketogenic diet: metabolic influences on brain excitability and epilepsy. Trends Neurosci Educ 36(1): 32-40.
- Zilberter T, Zilberter Y (2018) Ketogenic Ratio Determines Metabolic Effects of Macronutrients and Prevents Interpretive Bias. Front Nutr 5: 75.
- Okuda T (2019) A low-carbohydrate ketogenic diet induces the expression of very-low-density lipoprotein receptor in liver and affects its associated metabolic abnormalities. NPJ Sci Food 3: 25.
- Shai I, Schwarzfuchs D, Henkin Y, Shahar DR, Witkow S, et al. (2008) Weight loss with a low-carbohydrate, Mediterranean, or low-fat diet. N Engl J Med 359(3): 229-241.
- Westman EC, Tondt J, Maguire E, Yancy WS Jr (2018) Implementing a low-carbohydrate, ketogenic diet to manage type 2 diabetes mellitus. Expert Rev Endocrinol Metab 13(5): 263-272.
- Schugar RC, Crawford PA (2012) Low-carbohydrate ketogenic diets, glucose homeostasis, and nonalcoholic fatty liver disease. Curr Opin Clin Nutr Metab Care 15(4): 374-380.
- Gigante I, Tutino V, Russo F, De Nunzio V, Coletta S, et al. (2021) Cannabinoid Receptors Overexpression in a Rat Model of Irritable Bowel Syndrome (IBS) after Treatment with a Ketogenic Diet. Int J Mol Sci 22(6): 2880.
- Chimienti G, Orlando A, Lezza AMS, D Attoma B, Notarnicola M, et al. (2021) The Ketogenic Diet Reduces the Harmful Effects of Stress on Gut Mitochondrial Biogenesis in a Rat Model of Irritable Bowel Syndrome. Int J Mol Sci 22(7): 3498.
- Abduraman MA, Azizan NA, Teoh SH, Tan ML (2021) Ketogenesis and SIRT1 as a tool in managing obesity. Obes Res Clin Pract 15(1): 10-18.
- Muscogiuri G, Barrea L, Laudisio D, Pugliese G, Salzano C, et al. (2019) The management of very low-calorie ketogenic diet in obesity outpatient clinic: a practical guide. J Transl Med 17(1): 356.
- Jayarathne S, Koboziev I, Park OH, Oldewage-Theron W, Shen CL, et al. (2017) Anti-Inflammatory and Anti-Obesity Properties of Food Bioactive Components: Effects on Adipose Tissue. Prev Nutr Food Sci 22(4): 251-262.
- Leon BM, Maddox TM (2015) Diabetes and cardiovascular disease: Epidemiology, biological mechanisms, treatment recommendations and future research. World J Diabetes 6(13): 1246-1258.
- Luukkonen PK, Dufour S, Lyu K, Zhang XM, Hakkarainen A, et al. (2020) Effect of a ketogenic diet on hepatic steatosis and hepatic mitochondrial metabolism in nonalcoholic fatty liver disease. Proc Natl Acad Sci USA 117(13): 7347-7354.
- Dallak MA, Bin-Jaliah I, Albawardi A, Haidara MA, Sakr HF, et al. (2018) Swim exercise training ameliorates hepatocyte ultrastructural alterations in rats fed on a high fat and sugar diet. Ultrastruct Pathol 42(2): 155-161.
- Zhang H, Song C, Yan R, Cai H, Zhou Y, et al. (2021) High-fat diet accelerate hepatic fatty acids synthesis in offspring male rats induced by perinatal exposure to nonylphenol. BMC Pharmacol Toxicol 22(1): 22.
- Showalter MR, Nonnecke EB, Linderholm AL, Cajka T, Sa MR, et al. (2018) Obesogenic diets alter metabolism in mice. PloS one 13(1): e0190632.
- Leonardi DS, Feres MB, Portari GV, Zanuto ME, Zucoloto S, et al. (2010) Low-carbohydrate and high-fat diets on the promotion of hepatic steatosis in rats. Exp Clin Endocrinol Diabetes 118(10): 724-729.
- Larter CZ, Yeh MM (2008) Animal models of NASH: getting both pathology and metabolic context right. J. Gastroenterol. Hepatol 23: 1635-1648.
- Nassar ALD, Marot LP, Ovidio PP, de Castro GSF, Jordao AA (2014) Oxidative stress and fatty acid profile in Wistar rats subjected to acute food restriction and refeeding with high-fat diets. Acta Cir Bras 29: 178-185.
- Zelber-Sagi S, Nitzan-Kaluski D, Goldsmith R, Webb M, Blendis L, et al. (2007) Long term nutritional intake and the risk for non-alcoholic fatty liver disease (NAFLD): A population-based study. J Hepatol 47: 711-717.
- Marangoni F, Colombo C, Martiello A, Negri E, Galli C (2007) The fatty acid profiles in a drop of blood from a fingertip correlate with physiological, dietary and lifestyle parameters in volunteers. Prostaglandins Leukot. Essent. Fatty Acids 76: 87-92.
- Freedman SD, Blanco PG, Zaman MM, Shea JC, Ollero M, et al. (2004) Association of cystic fibrosis with abnormalities in fatty acid metabolism. N Engl J Med 350: 560-569.
- Fekete K, Györei E, Lohner S, Verduci E, Agostoni C, et al. (2015) Long-chain polyunsaturated fatty acid status in obesity: a systematic review and meta-analysis. Obes Rev (16): 488-497.
- Notarnicola M, Lorusso D, Tutino V, De Nunzio V, De Leonardis G, et al. (2018) Differential Tissue Fatty Acids Profiling between Colorectal Cancer Patients with and without Synchronous Metastasis. Int J Mol Sci 19(4): 962.
- Meuronen T, Lankinen MA, Karkkainen O, Laakso M, Pihlajamaki J, et al. (2021) FADS1 rs174550 genotype and high linoleic acid diet modify plasma PUFA phospholipids in a dietary intervention study. Eur J Nutr 61(2): 1109-1120.
- Lankinen MA, Fauland A, Shimizu B-i, Ågren J, Wheelock CE, et al. (2019) Inflammatory response to dietary linoleic acid depends on FADS1 genotype. Am J Clin Nutr 109: 165-175.
- PFluger PT, Herranz D, Velasco-Miguel S, Serrano M, Tschop MH (2008) Sirt1 protects against high-fat diet-induced metabolic damage. PNAS 105: 9793-9798.
- Pruitt K, Zinn RL, Ohm JE, McGarvey KM, Kang SH, et al. (2006) Inhibition of SIRT1 reactivates silenced cancer genes without loss of promoter DNA hypermethylation. PLoS genetics 2: e40.
- Gillum MP, Kotas ME, Erion DM, Kursawe R, Chatterjee P, et al. (2011) SirT1 regulates adipose tissue inflammation. Diabetes 60(12): 3235-3245.
- Picard F, Kurtev M, Chung N, Topark-Ngarm A, Senawong T, et al. (2004) Sirt1 promotes fat mobilization in white adipocytes by repressing PPAR-gamma. Nature 429: 771-776.
- Nisoli E, Tonello C, Cardile A, Cozzi V, Bracale R, et al. (2005) Calorie restriction promotes mitochondrial biogenesis by inducing the expression of eNOS. Science 310: 314-317.
- Xu F, Gao Z, Zhang J, Rivera CA, Yin J, et al. (2010) Lack of SIRT1 (Mammalian Sirtuin 1) activity leads to liver steatosis in the SIRT1+/- mice: a role of lipid mobilization and inflammation. Endocrinol 151: 2504-2514.
- Cho KW, Lumeng CN (2011) SirT1: a guardian at the gates of adipose tissue inflammation. Diabetes. 60(12): 3100-3102.
- Xia L, Tan S, Zhou Y, Lin J, Wang H, et al. (2018) Role of the NFkappaB-signaling pathway in cancer. OncoTargets and Ther 11: 2063-2073.
- Manea A, Manea SA, Gafencu AV, Raicu M (2007) Regulation of NADPH oxidase subunit p22(phox) by NF-kB in human aortic smooth muscle cells. Arch Physiol Biochem 113(4-5): 163-172.
- Gencoglu H, Orhan C, Tuzcu M, Sahin N, Juturu V, et al. (2020) Effects of walnut oil on metabolic profile and transcription factors in rats fed high-carbohydrate-/-fat diets. J Food Biochem 44: e13235.
- Mayoral R, Osborn O, McNelis J, Johnson AM, Oh DY, et al. (2015) Adipocyte SIRT1 knockout promotes PPARgamma activity, adipogenesis and insulin sensitivity in chronic-HFD and obesity. Mol Metab 4: 378-391.
- Tontonoz P, Spiegelman BM (2008) Fat and beyond: the diverse biology of PPARgamma. Annu Rev Biochem 77: 289-312.
- Evans RM, Barish GD, Wang YX (2004) PPARs and the complex journey to obesity. Nat Med 10: 355-361.
- Che M, Wang R, Li X, Wang HY, Zheng XFS (2016) Expanding roles of superoxide dismutases in cell regulation and cancer. Drug Discov Today 21(1): 143-149.
- Tsang CK, Yuan Liu, Janice Thomas, Yanjie Zhang, XFS Zheng (2014) Superoxide dismutase 1 acts as a nuclear transcription factor to regulate oxidative stress resistance. Nat Commun 5: 3446.
- Veal EA, Alison M Day, Brian A Morgan (2007) Hydrogen peroxide sensing and signaling. Mol Cell 26(1): 1-14.
- Forman HJ, Matilde Maiorino, Fulvio Ursini (2010) Signaling functions of reactive oxygen species. Biochemistry 49: 835-842.
- Younus H (2018) Therapeutic potentials of superoxide dismutase. Int J Health Sci (Qassim) 12(3): 88-93.
- Mauger M, Ferreri C, Chatgilialoglu C, Seemann M (2021) The bacterial protective armor against stress: The cis-trans isomerase of unsaturated fatty acids, a cytochrome-c type enzyme. J Inorg Biochem 224: 111564.
- Therien A, Cieslak A, Verreault M, Perreault M, Trottier J, et al. (2021) Omega-3 Polyunsaturated Fatty Acid: A Pharmaco-Nutraceutical Approach to Improve the Responsiveness to Ursodeoxycholic Acid. Nutrients.
- Yang J, Fernandez-Galilea M, Martinez-Fernandez L, Gonzalez-Muniesa P, Perez-Chavez A, et al. (2019) Oxidative Stress and Non-Alcoholic Fatty Liver Disease: Effects of Omega-3 Fatty Acid Supplementation. Nutrients 11(4): 872.
- Cieslak A, Trottier J, Verreault M, Milkiewicz P, Vohl MC, et al. (2018) N-3 Polyunsaturated Fatty Acids Stimulate Bile Acid Detoxification in Human Cell Models. Can J Gastroenterol Hepatol 2018: 6031074.
- Sun R, Wang X, Liu Y, Xia M (2014) Dietary supplementation with fish oil alters the expression levels of proteins governing mitochondrial dynamics and prevents high-fat diet-induced endothelial dysfunction. T Br J Nutr 112(2): 145-153.
- Minihane AM, Vinoy S, Russell WR, Baka A, Roche HM, et al. (2015) Low-grade inflammation, diet composition and health: current research evidence and its translation. Br J Nutr 114(7): 999-1012.

 Research Article
Research Article