Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Kabanya C N1*, Karani A K2 and Mirie W3
Received: August 15, 2017; Published: September 07, 2017
Corresponding author: Felix Nunura, Division of Cardiovascular Medicine, Heart Institute of the Caribbean, 23 Balmoral Avenue, Kingston, Jamaica
DOI: 10.26717/BJSTR.2017.01.000332
Background: Heart Failure with reduced Ejection Fraction (EF) is likely itself a heterogeneous entity within the same complex clinical syndrome. The relative contribution of various risk factors and etiological conditions involved in the development of heart failure with Midrange (HFmrEF) and Reduced Ejection Fraction (HFrEF) in the Caribbean population is unknown. We aimed to determine the impact of cardiac risk factors and etiological conditions associated with reduced EF and identify common and distinctive risk profiles between HFmrEF vs HFrEF in a series of cases of Afro-Caribbean population with heart failure.
Methods and Results: We report a case series of 100 consecutive patients (52 % male, age: 65.5 ± 15.3 years) with objective evidence of cardiac dysfunction (EF: 34.8±7.8 %) assessed in the Heart Institute of the Caribbean, Jamaica, over the past two years (2015-2017). The study population was categorized according to LVEF as follows: Overall patients with left ventricular systolic dysfunction (LVEF < 50 %, n=100); HF with midrange LVEF (40-49 %, HFmrEF; n=34, Group 1) and HF with reduced ejection fraction (< 40 %, HFrE; n=66, Group 2). The mean number of risk factors for heart failure (n=100) per case was 1.23±1.09; 36 % had 1, 35 % had 2 or more, while 29 % had none. For the overall group of patients Hypertension -HTN was the most common (68%), followed by Diabetes -DM (27%) but 31% was non-hypertensive and nondiabetic. LVEF < 50 % was associated in 48 % with ischemic heart disease -IHD (20 % with documented myocardial infarction) but in52% with non-ischemic conditions as follows: Dilated Hypertensive Heart Disease-DHHH (24 %), Dilated cardiomyopathy -DCM (14%), and others (10 %). Comparing Group 1 and Group 2, the most significant variable was age (73.8±11.9, Group 1 vs 61.4±15.2, Group 2; p 0.0001). Cases with HFmEF and HFrEF had similar (P > 0.05) clinical characteristics (HTN : 76.4 vs 63.6%; DM: 27.2 % vs 25.7%; dyslipidemia 11.7 % vs 15.1%; smoking 5.8%vs. 7.5 % , obesity 20.5% vs 9 % and atrial fibrillation (20.5% vs 13.6%) , however there were significant differences in the prevalence of DHHD (11% vs. 30.3 %) , IHD (61.7% vs 40.9%) , combined hypertensive and ischemic heart disease (52.9% vs 33%), DCM(2.9 vs. 21.2%) and the presence of wide QRS : LBBB (2.9% vs 22.7 %).
Conclusion: Afro-Caribbean patients with heart failure and reduced -abnormal, no preserved- ejection fraction- is mainly hypertensive with or w/o diabetes and half of them develop leftventricularsystolic dysfunction due to non-ischemic causes. In addition, they demonstrate a distinct etiological but similar clinical profile when they are classified in HFmrEF and HFrEF .The impact of these findings remains to be determined in a larger prospective study.
Keywords: Afro-Caribbean; Heart Failure; HFmrEF; HFrEF; Risk factors
Abbreviations: EF: Ejection Fraction; HFmrEF: Heart Failure With Midrange; HFrEF: Heart Failure Reduced Ejection Fraction; HF: Heart Failure; PAR: Population Attributable Risk; ARIC: Atherosclerosis Risk in Communities study; MESA: Multi-Ethnic Study of Atherosclerosis; DM: Diabetes Mellitus
The full impact of cardiovascular disease risk factors on incident disease and cardiovascular mortality in Jamaica and much of the Caribbean is still unknown [1]. Data from St James Study in Trinidad and the Barbados Registry of Stroke have highlighted the importance of hypertension and diabetes as predictors of coronary artery disease [2] and stroke [3] respectively; however we lack local information on the incidence of heart failure (HF) in the Afro-Caribbean community. The results of a study [4] of acute heart failure admissions to an UK city-Centre hospital that serves a multiethnic population showed that South Asian and Afro- Caribbean patients were younger than their white counterparts, indicating an earlier onset of disease. Moreover, a reanalysis of the same study, (4.1) taking into account population data, has suggested that the relative risk of heart failure in those aged 60-79 years was 3•1 (95% CI 1•9-4•9) for African Caribbean and 5•2 (3•7-7•4) for Indo-Asians. Furthermore, there were ethnic differences in the cause of heart failure, with hypertension most common among Afro-Caribbean and coronary artery disease and diabetes most common among South Asians. On the other hand, in adult African Americans the prevalence of HF has been estimated at 3% higher than white counterparts [5] and this is not explained by coronary artery disease, as the prevalence of CAD has been defined lower in black patients with HF [6]. The Population Attributable Risk (PAR) for HF has been shown to be greatest for coronary disease (20%) and hypertension (20%) in the Olmsted County Study [7]. Data on the burden of HF in diverse populations are scarce.
The Atherosclerosis Risk in Communities study (ARIC) [8] and the Multi-Ethnic Study of Atherosclerosis (MESA) [9] reported that HF incidence was higher in blacks than in whites. In both studies, the greater HF incidences in blacks was related to their greater burden of atherosclerotic risk factors as well as to socioeconomic status. Data on the incidence and prevalence of HF according to EF and how it may have changed over time are very limited. The available evidence suggests that the prevalence of HF with preserved EF increased over time [10]. In a report (2007) of 100 Jamaican heart failure patients by Lalljie [11]. 49% had echocardiograms, of these 39% had ejection fractions (EF) > 40%, 34 % had EF 21-40 % and 27% had EF < 20%. Hypertensive heart disease was found in 54%, hypertensive cardiomyopathy in 14% and ischemic heart disease in 26%.Evidence-based therapies for heart failure (HF) differ significantly according to left ventricular ejection fraction (LVEF). However, few data are available on the phenotype and prognosis of patients with HF with midrange LVEF of 40% to 55% (HFmrEF), and the impact of recovered systolic function on the clinical features, functional capacity, and outcomes of this population is not known [12]. Previous investigations in American [13] and Chinese [14] populations have demonstrated that subjects with HF and a normal EF (>55%) -HFNEF, differ in their clinical and demographic characteristics from subjects with mildly (40% to 55%) and severely (<40%) decreased EF. Accordingly, it is clear that differentiation of patients with HF based on LVEF is important, [15] however there are no comparative research among Afro-Caribbean patients with (HFmrEF) and (HFrEF). In the present study, we aimed; (I) to assess the clinical features, associated cardiovascular risk factors and etiological conditions in Afro-Caribbean patients with objective evidence of left ventricle systolic dysfunction-LVSD (EF <50%) , and (II), In accordance with the new terminology [16] to identify common and distinctive risk profiles between patients with HFmrEF (EF 40-49 %) and patients with HFrEF (EF < 40 %).
This is an observational study. The series of cases were 100 consecutive inpatients or outpatients seen at the Heart Institute of the Caribbean, Jamaica, referred to our institution by local community hospitals or general practitioners who underwent standardized clinical and cardiovascular testing, including echocardiography during the past two years (2015 to 2017). The study had been originally planned as a retrospective design; however, in a consecutive manner the subjects covered in the registry were prospectively expanded for the purpose of increasing study population.
Pertinent data were abstracted from our Electronically Medical Records (SMART-EMR) for all patients (n=100) included in this study and were independently reviewed by Cardiologists to adjudicate the presence or absence of HF based on the European Society of Cardiology (ESC) criteria [16]. The data reviewed for this adjudication process included demographic, past medical history, and available test reports including echocardiogram findings. From this population we identified the following group of patients:
a. Those with HF and abnormal EF (< 50 %, overall group),
b. Those with HF and a midrange EF (40% to 49 %, HFmrEF. Group 1), and
c. 3- Those with HF and a reduced EF (<40%, HFrEF, Group 2).
An EF >50 % (HFNEF or HFpEF) was employed to define normal because the lower boundary of EF for our cases were 50 %.
A complete echocardiogram was obtained with the use of an ultrasound instrument with a 3.5- to 4.5-MHz sector scanner. Twodimensional guided M-mode measurements of systolic and diastolic chamber dimensions and wall thickness were obtained according to recommendations of the American Society of Echocardiography (ASE) [17] Alular regurgitation and stenosis and other Doppler variables were assessed by standard techniques. Among groups, we evaluated the relation between 2 determinants: Left ventricle enddiastolic dimension (mm) and Left Ventricle Ejection Fraction (%). The following was the criteria (non mutually exclusive) to establish the etiology of the Heart Failure:
Dilated Hypertensive heart Disease (DHHD) / Hypertensive Dilated Cardiomyopathy:
A. History of Hypertension (HTN), as the only known risk factor
B. No history of previous Myocardial infarction (MI)
C. Absence of resting, regional, left ventricle wall motion abnormalities-LVWMA (Echo) or Pathological Q waves (ECG) suggestive of prior MI
D. Normal Angiogram or Cardiac Nuclear Scan (if available). Ischemic Heart Disease (IHD):
A. Definite history of previous MI or Prior documented Myocardial infarction
B. No history of HTN
C. Presence of resting, regional LVWMA (Echo) or Presence of pathological Q waves (ECG)
D. Abnormal Angiogram (epicardial coronary artery disease).
Combined Hypertensive and Ischemic Heart Disease:
A. History of Hypertension
B. Positive or Negative history of Diabetes
C. Resting, regional LVWMA (Echo)
D. Presence of pathological Q waves (ECG)
Valvular Heart Disease (VHD): obvious hemodynamically significant valvular dysfunction. 5- Dilated Cardiomyopathy-DCM:
A. Negative history of Hypertension, Diabetes, or previous MI
B. Cardiomegaly and generalized LV dysfunction. Absence of regional LVWMA.
Restrictive cardiomyopathy- RCM:
A. Obvious restrictive physiology
B. Atrial enlargement and normal LV dimensions
C. Infiltrative (i.e. Amyloidosis) pattern usually with LV hypertrophy.
a. Peripartum Cardiomyopathy: DCM at the end of pregnancy or after delivery.
b. Non compaction cardiomyopathy (NCCM): typical spongy appearance of Left ventricle walls.
Data are expressed as mean ± SD. The final analysis was done for comparisons of data (means, T-Test and Proportions) among groups for differences in age, gender, cardiovascular risk factors, associated heart disease, cardiac rhythm, and other cardiovascular testing when available. The primary focus of these comparisons was in subjects with HF and midrange EF of 40% to 49 %, referred to as Group 1 and subjects with HF and reduced EF < 40 % , referred as Group 2. All statistical analyses were performed with Medcalc version 17.0.4 statistical software. A p value <0.05 was considered statistically significant.
Tables 1-3 show the demographic and clinical characteristics of the Overall Group of patients (N=100 ) with left ventricular systolic dysfunction (LVSD) ,LVEF < 50 %. The mean number of risk factors for heart failure per case was 1.23±1.09; 36 % had 1, 35 % had 2 or more, while 29 % had none. Hypertension -HTN was the most common (68%), followed by Diabetes -DM (27%) but 31% was nonhypertensive and non-diabetic. LVSD was associated with ischemic heart disease -IHD in 48% and with non-ischemic conditions in 52 % as follows: Dilated Hypertensive Heart Disease-DHHH (24 %),Dilated Cardiomyopathy -DCM (14%), and others (10 %). The ECG showed Atrial Fibrillation in 16 % and LBBB was noted in 16 %.
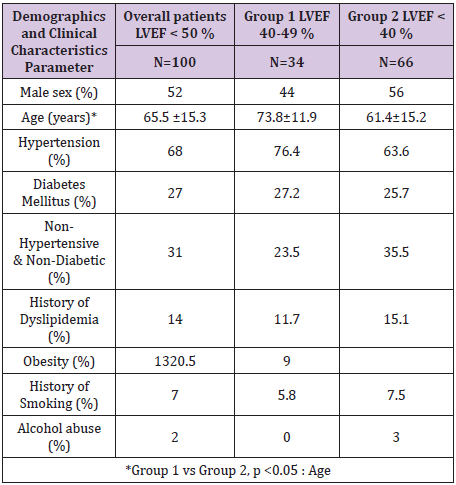
Table 1: Afro Caribbean Heart Failure.
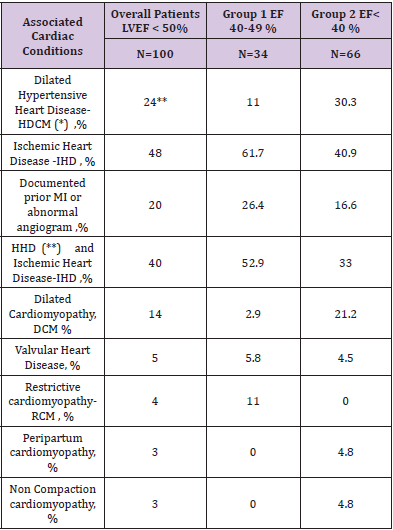
Table 2: Afro Caribbean Heart Failure.
*HHD. Hypertensive Heart Disease
**DHHD or Hypertensive Dilated Cardiomyopathy (HDCM): These patients had past history of hypertension and prominent left ventricular dilatation with reduced left ventricular contractility.
***Group 1 vs Goup 2, p < 0.05: DHHD, IHD, HHD & IHD, DCM, and RCM.

Table 3: Afro Caribbean Heart Failure.
Tables 1-3 show the demographic and clinical data for the different series of cases withHFmrEF; LVEF 40-49 %; n=34, Group 1 and HFrEF; LVEF < 40 %; n=66, Group 2. Patients differed significantly in Age but did not differ significantly in gender and conventional risk factors. Compared with subjects with HFrEF, those with HFmrEF were older and more often women. In subjects with HFrEF, hypertension was less frequent compared with subjects with HFmrEF but the combined history of hypertension and diabetes was more frequent in HFmrEF than in subjects with HFrEF. Although the cases did not differ significantly with regard to the prevalence of atrial fibrillation (20.5 vs 13.6 %) and valvular heart disease-VHD (5.8 vs. 4.5 %), the prevalence of a definite history of prior myocardial infarction (26.4 vs 16.6 %) and combined hypertensive and ischemic heart disease (52.9 vs 33 %), was more common in subjects with HFmrEF . Dilated hypertensive heart disease (11 vs 30 %) and dilated cardiomyopathy (2.9 vs 21.2 %) were significantly more common in subjects with HFrEF than in those with HFmrEF. A history of diabetes mellitus was similar in both groups (27.2 vs. 25.7 %) however the comparison of markers of worst prognosis for HF demonstrated more significant myocardial remodeling (LVEDD: 55.4±8 vs. 68.6± 10.7 mm) and more prevalence of wide QRS complex-LBBB (2.9 vs. 22.7 %) in subjects of Group 2 (HFrEF).
The major findings of the present study were that
A. Fifty-two % of the Afro-Caribbean patients with objective evidence of Heart Failure develop LVSD (EF < 50 %) associated with non-ischemic conditions and
B. Except for age, patients with HFmrEF share similar clinical features and frequency of risk factors with HFrEF patients, however they differ in the etiologic factor associated with cardiac failure.
To the best of our knowledge, this is the first 100 case series studies that describe comparatively, the incidence and the influencing factors of Afro-Caribbean patients with HF based on LVEF in an observational study.
The majority of Jamaicans (92.1%) identify as black [18] however there are otherethnic groups are [19]: Mixed (6.1%), Asian (0.8%), others (0.4%). Much of Jamaica’s black population is of African or partially African descent with many being able to trace their origins to West Africa. It has been previously observed that [20]:
A. Coronary Heart Disease (CHD) and Peripheral Arterial Disease (PAD) are less prevalent among Afro-Caribbeans compared to Caucasian and South East Asian ethnic groups (prevalence of CHD range from 0-7 % in Afro-Caribbean to 2-22 % in Caucasians)
B. Strokes are more common among Afro-Caribbean. Unfortunately, there are inadequate data on morbidity and mortality from cardiovascular disease (CVD), particularly across the socio-economic spectrum in Afro-Caribbean populations.
The prevalence of hypertension in Jamaicans was initially estimated in 30.8% in the 15-and-over age group [21]; however a more recent survey estimates the traditional CVD risk factors as follows : hypertension, 25%; diabetes, 8%; hypercholesterolemia, 12%; obesity, 25%; and smoking 15% [1]. Therefore it is obvious that the burden of cardiovascular disease risk factors in Jamaica remains very high.
Heart failure (HF) is usually a progressive condition that begins with risk factors for LV dysfunction (e.g., hypertension), proceeds to asymptomatic changes in cardiac structure (e.g., LV hypertrophy) and function (e.g., impaired relaxation), and then evolves into clinically overt HF, disability and death [22]. In the present study we examined the characteristics of 100 patients with EF <50 % .68% were hypertensive and 32 % were non-hypertensive. It is difficult to know in which proportion these hypertensive patients evolved towards one of the different possibilities of progression from hypertensive heart disease (HHD) to cardiac failure. It is now well recognized that clinical heart failure can occur either in the setting of reduced LVEF or preserved- LVEF. Drazner [23], has proposed several possible pathways in the progression from hypertension to heart failure, accordingly, a direct pathway from hypertension to dilated cardiac failure (increased LV volume with reduced LVEF) can occur without or with an interval myocardial infarction (MI) however concentric LVH progresses to dilated cardiac failure most commonly via an interval myocardial infarction . Other important modulators of the progression of hypertensive heart disease are obesity, diabetes mellitus, age, environmental exposures, and genetic factors. 24 % of our hypertensive patients had dilated cardiac failure (Dilated HHD) without a definite history of myocardial infarction (and absence of regional wall motion abnormalities in the echo study). Hypertensive heart disease simulating dilated cardiomyopathy has been previously documented [24]: these patients had past history of hypertension and prominent left ventricular dilatation with reduced left ventricular contractility, but no left ventricular wall thickening. Among cases reported by Lalljie [11] 14% were considered as hypertensive cardiomyopathy. In short, the consideration of a direct pathway from hypertension to dilated HHD seems to be plausible. It has recently been recognized that in Afro-Caribbean patients with heart failure the most common cause of heart failure was non ischemic dilated cardiomyopathy (NIDCM) [25-35] (27.5% vs whites, 19.9%; P<0.001) and lower rates of ischemic cardiomyopathy were observed (13% versus 41%; P<0.001) and cardiac amyloidosis has been mentioned as the fourth most common cause (11.4 %) of heart failure in Afro- Caribbean. We found that 52 % of Afro-Caribbean Heart Failure in our population was associated with non-ischemic conditions. In this group are included a 24 % with dilated HDD and 14 % with dilated cardiomyopathy (DCM) of unknown etiology. Campbell et al [36] described twenty one cases of DCM documented with necropsy occurred in the Jamaica community and Tulloch Reid et al. [37] have reported the association of HTLV-1 seropositivity and unexplained dilated cardiomyopathy in Jamaican patients. Tulloch Reid has also reported the experience of 26 cases (45±11 years-old) with unexplained dilated cardiomyopathy at Kingston Public Hospital [38], however more data about HF in Afro-Caribbean community is needed .Since we do not routinely perform testing for cardiac amyloidosis (ATTR V122I), it is possible that some of these subjects may be confused with other etiologies. According with Dunguet et al. [26] about 4% of African-Americans possess the V122I variant of transthyretin, associated with cardiac amyloidosis (ATTR) and 10 % of Afro-Caribbean heart failure population have ATTR V122I, often misdiagnosed as hypertensive heart disease (HHD).
This study shows the characteristics of 34 patients with HFmrEF (Group 1). It has been recognized (16) that patients with an LVEF in the range of 40-49% represent a ‘grey area’: Identifying HFmrEF as a separate group can stimulate research into the underlying characteristics, pathophysiology and treatment of this group of patients. Patients with HFmrEF most probably have primarily mild systolic dysfunction, but with features of diastolic dysfunction [16]. Aging has been associated with diastolic dysfunction. After comparing the two groups (HFmrEF vs HFrEF), we observed that the most significant variable was age (73.8±11.9, Group 1 vs 61.4±15.2, Group 2; p 0.0001). Although HFmrEF and HFrEF had similar clinical characteristics, patients with HFmrEF showed more association with Ischemic heart Disease (IHD), and combined hypertensive and IHD. Specifically, Group 1, more often consists of older women with long-standing hypertension, whereas the presence of coronary artery disease and a definite history of past myocardial infarction were more common than in subjects with HF and reduced EF. This group also showed only mildly dilated left ventricles (55.4 ± 8 mm). In a cohort of Chinese patients with mildly decreased EF (40 % to 55 %), despite only mildly dilated left ventricular dimensions, they had significant ventricular remodeling (e.g., rightward shift in end-diastolic pressure-volume relation) and decreases in chamber contractility [14]. The researchers comment that this is markedly different from the physiologic parameters displayed in the cohort with HF and EF>55%, and more similar to the phenotype observed in patients with overt systolic HF (with an EF <40%). Nadruz et al [12] have concluded that patients with HFmrEF demonstrate a distinct clinical profile from HFpEF and HFrEF patients. Within the HFmrEF population, recovered systolic function is a marker of more favorable prognosis. With current understanding, HFmrEF is distinct from HFrEF (EF < 40%) simply because there is no evidence-based specific therapy, and distinct from HFpEF (EF ≥ 50%) simply because EF is not preserved [27].
Heart failure (HF) has been singled out as an epidemic and is a staggering clinical and public health problem associated with significant mortality, morbidity, and healthcare expenditures, particularly among those aged ≥65 years [30]. Data for 66 patients with HFrEF was available for our analysis. The mean age was 61.4 years old but 39.3 % were less than 60 years-old and 18.2 % were less than 50 years old. It has been described that incident HF before 50 years of age is substantially more common among blacks than among whites. Hypertension, obesity, and systolic dysfunction that are present before a person is 35 years of age are important antecedents that may be targets for the prevention of heart failure [28]. Among the patients with HFrEF, 63.6 % were hypertensive, 25.7 % diabetic, and 13.6 % had atrial fibrillation. In the E-ECHOES Study [29] of Prevalence of Heart Failure and Atrial Fibrillation in Minority Ethnic Subjects, 17 Afro Caribbean -AC, (0.89%; 95% CI 0.52 to 1.42%) had an LVEF < 40% ( mean age 72.4 years ± 11 , 88.24% males ). 57.9 % were hypertensive, 26.3 % were diabetic and just 1.36 % had evidence of atrial fibrillation with a mean CHADS2 score of 2.3.
Assigning a cause to HF should be envisioned while focusing on clinically ascertained risk factors and acknowledging that multiple causes for HF often coexist and interact in a given patient [30]. HFrEF was associated with dilated hypertensive heart Disease- (DHHD) in 30.3 %, a definite history of myocardial infarction in 16.6 %, combined hypertensive and ischemic heart disease in 33 % and dilated cardiomyopathy-(DCM) in 19.6 %.It has been established that the etiology of HF is diverse within and among world regions. Many patients will have several different pathologies-cardiovascular and non-cardiovascular-that conspire to cause HF, and many patients with HF and ischemic heart disease (IHD) have a history of myocardial infarction or revascularization. However, a normal coronary angiogram does not exclude myocardial scar (e.g. by CMR imaging) or impaired coronary microcirculation as alternative etiology for IHD [16]. In a random sample of 13 angiograms (40) of our patients with HFrEF (38 % were hypertensive and diabetic and 30.7 % were non-hypertensive, non diabetic).We found six patients (46.1%) with epicardial coronary artery disease (IDCM) and seven (53.8 %) with angiographically normal coronary arteries (NIDCM). On the other hand, when we examined the differences between group 1 (HFmrEF) and group 2 (HFrEF) with respect to the etiological factors associated with left ventricle systolic dysfunction (LVSD), we observed more patients with DHHD, DCM and LBBB in the group 2 which underlines the importance of myocardial diseases as a causative factor in the Afro-Caribbean population.
As previously mentioned, it has been reported [26] that 10% of Afro-Caribbean heart failure population have ATTR V122I, often misdiagnosed as hypertensive heart disease (HHD), which informs our recommendation for more specific studies for cardiac amyloidosis in our population. On the other hand, the fact that 20% of patients with HFrEF have wide QRS complex (LBBB), should stimulate a desire to consider allocation of more resources to Cardiac Resynchronization Therapy (CRT) in the Caribbean community. It has been widely speculated that LBBB may be the first manifestation of heart disease whereas the clinical presentation of a dilated cardiomyopathy develops only some years later [31] and CRT has been recommended for symptomatic patients with HF in sinus rhythm with a QRS duration ≥150 msec and LVEF ≤35% despite OMT, in order to improve symptoms and reduce the HF morbidity and mortality [16].
According to Sanderson [32], HFrEF is mainly due to myocardial infarction or dilated cardiomyopathy that rapidly damages the myocardium. He emphasizes that cardiologists working in Africa, East Asia, and India often can see patients with HFrEF who have only hypertension as a risk factor, and presumably these patients passed through an HFpEF phase, however other associated risk factors may interact, including alcohol intake. Accordingly, the time course and pattern of development of heart failure (HF) is primarily caused by:1-Myocardial Infarction (MI), with pronounced remodeling and shape change leading to systolic heart failure (SHF), or 2- Hypertension, with or without diabetes mellitus (DM), leading to heart failure with , initially, a normal ejection fraction (HFpEF). Afro-Caribbean HF appears to be of worse severity than HF in white patients. When affected by heart failure, Afro-Caribbean patients might experience a greater rate of hospitalization and may be exposed to a higher mortality risk as well. Genomic medicine has yielded a number of candidate single nucleotide polymorphisms that might contribute to the excess pathogenicity of heart failure in African Americans [33] , but much more work needs to be done in other cohorts. Within the Afro-Caribbean patients with objective evidence of left ventricular dysfunction in the SOLVD Registry [34], 24% appeared to have hypertension as the sole plausible cause of LV dysfunction as compared to only 4% of white patients.
It seems likely that Afro-Caribbean hypertensive patients would evolve through an initial phase with preserved EF and depending on the associated factors, they eventually develop systolic dysfunction, initially mild (HFmrEF) and finally severe (HFrEF). The present study suggests that, when defined as a prior documented myocardial infarction or known epicardial coronary artery disease, IHD appears to be present in approximately 20 % of African-Caribbean patients with heart failure-a rate that is less than the half of that seen in white patients with heart failure. It is thus evident that a non-ischemic etiology would be responsible for heart failure in ≈52 % of African Caribbean patients with HF. Although 68% of patients with HF are hypertensive, the exact mix of HHD and idiopathic DCM (as well as the influence of other cardiomyopathies) is not discernible from the present data. Therefore, the contribution of hypertensive heart disease as the sole explanation of LV dysfunction is likely to be in the range of 24 %. The impact of an effective therapy on reversere modeling may further change this complex final outcome. Further research related to the use of Hydralazine-ISDN [35] in heart failure in Afro- Caribbean population seems warranted.
Socioeconomic and cultural factors and limited access to specialized diagnosis by the Afro-Caribbean population could affect the presented data. As Madu et al. [39] have previously reported, nearly 85% of the global CVD mortality and disease burden occur in developing countries, driven mainly due to the absence of appropriate infrastructure for specialized diagnostic and treatment services for CVDs. Furthermore, this study was performed in an Afro Caribbean Black population. Because other factors related to ethnicity and socio-economic status can be an important factor in disease mechanism and propagation, our study is not free from such confounding factors. It is therefore implicit that certain presumptions and conclusions about racial differences in cardiovascular disease expression must be interpreted cautiously, and a more rigorously obtained data will be helpful in further elucidating our findings [39].
It is increasingly recognized that the population of patients with Heart Failure and LVSD (EF < 50 %) constitutes a heterogeneous category with potentially multiple underlying pathophysiology mechanisms contributing to the observed phenotype. We demonstrate that Afro-Caribbean patients with LVSD are mainly hypertensive with or w/o diabetes and half of them develop ventricular systolic dysfunction due to non-ischemic causes. In addition they demonstrate a distinct etiologic profile when they are classified in the categories of HFmrEF and HFrEF.


