Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Takeshi Morinaga1, Kei Horino1, Shinya Shimada1*, Keisuke Morita1, Kenichiro Yamamoto1, Satoshi Ikeshima1 and Hideo Baba2
Received: November 09, 2017; Published: November 16, 2017
Corresponding author: Department of Surgery, Kumamoto General Hospital, Japan Community Health Care Organization, Kumamoto, 10-10 Tohri-cho, Yatsushiro, Kumamoto 866-8660, Japan
DOI: 10.26717/BJSTR.2017.01.000536
It is extremely rare for metastatic tumors derived from extra-abdominal sites to arise in the small intestine [1, 2]. Herein, we report a case of the metastasis of esophageal squamous cell carcinoma (SCC) to the small intestine. A 72-year-old male, who had previously undergone subtotal esophagectomy for esophageal carcinoma at our hospital, was admitted with abdominal pain and abdominal distension. Abdominal computed tomography revealed an increase in the thickness of part of the small intestinal wall, which had caused mild bowel obstruction. The patient had no severe symptoms, so we initially administered conservative therapy (a fast venal transfusion), and his symptoms temporarily improved. However, the ileus did not improve so we conducted laparotomy. A solitary tumor was identified in the ileum, which was determined to be the cause of the intestinal obstruction, but no other tumors were seen at intra-abdominal sites. Based on the postoperative pathological findings, the lesion was diagnosed as a metastatic tumor derived from esophageal SCC. After surgery, the ileus vanished, and the patient was discharged without symptoms. We encountered an extremely rare case of small intestinal metastasis from esophageal carcinoma.
Recently, some cases of small intestinal metastasis from extraabdominal sites have been reported, but such cases are rare. In patients with esophageal carcinoma, we often encounter liver, lung, and bone metastases, but small intestinal metastasis from esophageal carcinoma is extremely rare. It was first reported by Wang et al. in 1985 [1-4]. Herein, we report a case of small intestinal metastasis from esophageal carcinoma combined with bowel obstruction. We had performed subtotal esophagectomy and 2-field lymphadenectomy because of esophageal squamous cell carcinoma (SCC) 6 years ago to treat the primary tumor.
A 72-year-old male underwent subtotal esophagectomy and 2-field lymphadenectomy because of esophageal SCC in the middle thoracic esophagus (pT1bN0M0 stage II) 6 years ago [5]. After surgery, he received chemotherapy, involving 5-fluorouracil, cisplatin, and docetaxel, but it was interrupted during the first course due to kidney dysfunction. Four years and 9 months later, lung metastasis was detected in the right lung on computed tomography (CT). We performed a partial pulmonary resection and found other tumors, which were not detected on pleural preoperative imaging. All of the tumors were resected, and the diagnosis of metastasis from esophageal SCC was confirmed. No additional treatment was administered because the patient exhibited renal hypo function. One year later, some pleural dissemination and lung metastases appeared; however, the patient did not have any symptoms. He subsequently visited our hospital regularly. About 6 years after the subtotal esophagectomy, he presented to our hospital with abdominal pain.
A physical examination revealed slight abdominal pain in his lower abdomen, abdominal distension, and a surgical scar from the medial incision made in the abdomen during the subtotal esophagectomy. A laboratory analysis produced the following results: white blood cell count, 6.3 × 103/mm3 (normal range, 3.5-9.0 × 103/mm3); hemoglobin count, 10.8 g/dL (normal range, 13.5-18.0 g/dL); platelet count, 42.7 × 104/mm3 (normal range, 13.0-37.0 × 104/mm3); total bilirubin, 0.5 mg/dL (normal range, 0.4-1.5 mg/dL); albumin, 3.3 g/dL (normal range, 4.1-5.1 g/dL); C-reactive protein; 3.68 mg/dL (normal range, 0-0.3 mg/dL); blood urea nitrogen, 60.0 mg/dL (normal range, 8.0-20.0 mg/dL); and creatinine, 1.71 mg/dL (normal range, 0.-1.1 mg/dL). The patient’s serum level of SCC antigen was 10.8 ng/ml (normal range, <1.5 ng/ ml), and his serum level of carcinoembryonic antigen was 6.6 ng/ ml (normal range, <5 ng/ml). All of these parameters, except the white blood cell count and total bilirubin level, exhibited abnormal values.
Abdominal CT revealed an increase in the thickness of part of the small intestinal wall, which had caused bowel mild obstruction (Figure 1). It was possible that the patient had a malignant neoplasm, such as small intestinal carcinoma or a gastrointestinal stromal tumor. His symptoms included slight abdominal pain and abdominal distension, so we initially administered conservative therapy, and his symptoms temporarily got better. However, his ileus did not improve so we conducted a laparotomy. A solitary tumor was identified in the ileum about 80 cm proximal to the terminal ileum, and it was determined to be the cause of the bowel obstruction. No other tumors were seen in the abdominal cavity, including the small intestine (Figure 2). Thus, we conducted a partial resection of the small intestine.
Figure 1: Abdominal CT revealed that part of the small intestinal wall had become thicker, causing bowel obstruction.
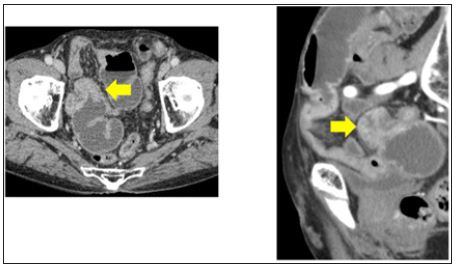
Figure 2: A solitary tumor was identified in the ileum about 80 cm proximal to the terminal ileum, and it was determined to be the cause of the patient’s bowel obstruction.

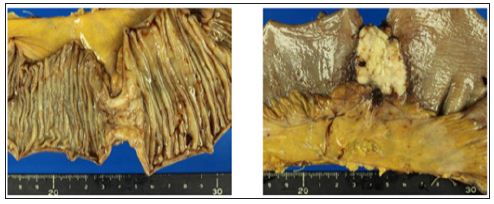
Macroscopically, the surgical specimen contained a mass measuring 3.0 × 2.5 cm in size, and it occupied the whole circumference of the ileal lumen, resulting in stenosis (Figure 3). Histologically, the lymphatic sub mucosal infiltration of viable SCC tumor cells was observed from the mucous membrane to the serosa. These histological findings revealed that the metastasis was not derived from peritoneal dissemination. In addition, there was no mesenteric lymph node metastasis from the SCC. The patient was discharged without symptoms, but ileus appeared again at 1 month after the partial resection of the small intestine, and so he was re-hospitalized. CT showed that the lung and liver metastases were getting worse, and some bone metastases had appeared.
Figure 3: The surgical specimen contained a mass measuring 3.0 × 2.5 cm, and it occupied the whole circumference of the ileal lumen.
Some types of cancer are reported to be associated with intestinal metastases, such as lung cancer, breast cancer, and malignant melanoma. However, metastatic intestinal tumors derived from extra-abdominal sites are very rare [1,2]. We usually examine esophageal cancer patients for liver, lung, and bone metastases, whereas intestinal metastatic tumors derived from esophageal cancer are extremely rare [3,4]. SCC of the esophagus is characterized by extensive lymphatic dissemination via the longitudinal lymphatic system, which runs along the esophagus and drains into the large number of (widespread) lymph nodes found in the thorax and abdomen [6]. It has been reported that the retrograde spreading of esophageal SCC cells into the cervical, mesenteric, or iliac lymph nodes is unusual [7,8].
The intra-abdominal region can be reached from the esophagus via the lymphatic network connecting the esophagus and intestines, lymphatic embolization, and peritoneal seeding of the tumor during surgery. The intra-abdominal region can also be reached via the hematogenous route, especially via the vertebral venous plexus [9,10]. In esophageal cancer, metastases can arise in the intestinal tract via any of these routes. In our case, it is unlikely that tumor seeding during the operation was responsible for the patient’s metastasis because only one small intestinal metastatic lesion formed. Microscopically, the lymphatic submucosal infiltration of viable SCC tumor cells was observed. Based on this finding, lymphoid metastasis is the most likely mechanism by which metastasis occurred in this case, but hematogenous spread cannot be excluded.
One interesting aspect of this case is why the solitary metastasis occupied the whole circumference of the ileal lumen. The lymphatic network develops on the mesenteric side of the small intestine, so mesenteric dissemination via the lymphatic network usually causes metastatic lesions to appear on the mesenteric side, as is seen in breast cancer. This might be explained by the characteristics of SCC. There have been several reports about small intestinal metastasis derived from esophageal SCC [3,4,11]. The tumors in these cases were detected based on their clinical symptoms, such small bowel obstruction and perforation, or were detected via imaging in symptomless patients.
In some cases, multiple metastases arose. In our case, an isolated small intestinal metastasis derived from esophageal carcinoma caused small bowel obstruction. The patient was disease-free for about 4 years and 9 months after undergoing subtotal esophagectomy until a metastatic lesion developed in his right lung. After undergoing partial intestinal resection, he wasdischarged, but the ileus recurred. CT showed that the lung and liver metastases had got worse, and some bone metastases subsequently appeared. Small intestinal metastases derived from esophageal carcinoma might be a significant finding that is generally associated with a poor prognosis. In conclusion, intestinal metastatic tumors derived from esophageal cancer are extremely rare. In addition, such tumors can develop with or without symptoms. Thus, physicians should keep in mind the possibility of small intestinal metastasis derived from esophageal carcinoma when patients who have a history of advanced esophageal cancer present with acute abdominal problems, such as bowel obstruction or perforation.


