Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Shefali K Sharma*, Prasanth Balasubramanian, Varun Dhir, Aman Sharma and Sanjay Jain
Received: January 24, 2018; Published: February 02, 2018
Corresponding author: Shefali Khanna Sharma, Department of Internal Medicine, Postgraduate Institute of Medical Education and Research, 4th floor, F block, Nehru Hospital, Chandigarh, India, 160012
DOI: 10.26717/BJSTR.2018.02.000721
Chronic inflammatory conditions like Rheumatoid Arthritis (RA) causes deposition of Amyloid Associated (AA) in various tissues or organs resulting in secondary or reactive systemic amyloidosis. We have reported here a 58-year old lady, known case of RA since 17 years with very poor compliance to treatment who presented with chronic diarrhea since 2 months, on evaluation, upper GI endoscopy and biopsy revealed positive for serum amyloid associated on innumohistochemistry staining, suggestive of secondary systemic amyloiosis. Symptoms gradually disappeared with the prompt institution disease modifying anti-rheumatic drugs (DMARDs). GI amyloidosis has been reported up to 60% of the patients with secondary amyloidosis and must not be forgotten while evaluating the cause for chronic diarrhea in a patient with chronic inflammatory condition like RA.
Keywords: Rheumatoid arthritis, amyloidosis, secondary amyloidosis, gastrointestinal amyloidosis
Abbreviations: RA: Rheumatoid Arthritis; AA: Amyloid Associated; GI: Gastro Intestinal; AL: Amyloid Light chain; DMARDs: Disease Modifying Anti-Rheumatic Drugs
Figure 1: Flexion of the metacarpophalangeal joint and extension of the interphalangeal joint of the thumb (Z-deformity) and flexion deformity of the 2nd to 5th proximal interphalangeal joints.
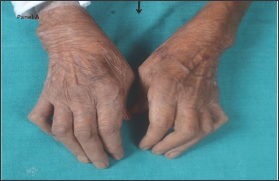
Amyloidosis refers to deposition of mis folded protein fibrils in extracellular tissues causing local or systemic manifestations. Chronic inflammatory conditions like Rheumatoid Arthritis (RA) causes deposition of Amyloid Associated (AA) in various tissues or organs resulting in secondary or reactive systemic amyloidosis [1] One such manifestation is due to the deposition of AA in gastrointestinal (GI) system results in GI amyloidosis. We have reported here a case of chronic diarrhea in a patient with RA, which on detailed evaluation was found to be due to secondary amyloidosis (Figure 1).
A 58-year old lady, with history suggestive of inflammatory joint pains since 17 years and very poor compliance to treatment, presented with chronic diarrhea since 2 months. On examination, patient had Z-shaped thumb deformity and flexion deformity of her fingers which are typically seen in rheumatoid arthritis. On evaluation, patients' complete blood counts were Hemoglobin (Hb-7.3gm/dL, Total Leucocyte Count (TLC)- 11.2x 109/L with differential leucocyte count-polymorphs 70%, lymphocytes 22%, eosinophils 4% and basophil 4%, platelet Count 236 x 103, ESR was 55 mm/hr and peripheral smear showed microcytic to normocytic RBCs and hypochromasia. Biochemistry investigations revealed serum sodium 135mmol/L, serum potassium 3.5mmol/dL, blood urea nitrogen- 48mg/dL, creatinine 2.1mg/dL, and Total protein- 4.8 mg/dL, serum albumin 1.6 with albumin: globulin ratio 1: 3.8, Serum calcium- 8.8mg/dL. X ray of the involved joints showed peri-articular osteopenia with symmetrical narrowing of the joint spaces. 24-hour urinary protein showed massive proteinuria (3.3gm/24 hrs). Stool workup was negative for microorganisms and fat globules (Figures 2 & 3). Upper GI endoscopy was done which revealed Amyloid deposits that were positive for Serum Amyloid Associated protein. As patient developed leucopenia secondary to methotrexate 2 months prior to the current admission, it was deferred and she was started on Lefunomide after which her proteinuria and diarrhea gradually improved.
Figure 2: Periarticular osteopenia and symmetrical narrowing of the joint spaces of metacarpophalangeal and proximal interphalangeal joints.
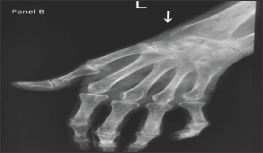
Figure 3: Pink colored amyloid deposits in the lamina propria, stained by congo red when seen sunder light microscope.
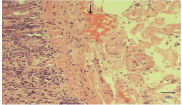
GI amyloidosis is seen in up to 60% of the patients with secondary or reactive systemic amyloidosis, whereas, up to 8% of the patients with primary or Amyloid Light chain (AL) amyloidosis [2,3] GI amyloidosis has also been seen associated with dialysis- related, hereditary, localised and senile amyloidosis, although rarely. The pathogenesis of GI amyloidosis is due to direct mucosal infiltration, neuromuscular damage or rarely due to autonomic neuropathy (Figure 4). Tada et al. [4] reported the endoscopic and biopsy findings in 37 patients with GI amyloidosis and the frequency of amyloid deposition was maximum in the duodenum (100%) followed by stomach (95%), colorectum (91%), and esophagus (72%). Also, the degree of deposition of amyloid in the duodenum correlated with the frequency of positive endoscopic findings like polypoid protrusions, granular appearance etc [4]. GI amyloidosis manifests as chronic diarrhea leading to protein losing gastroenteropathy, steatorrhea, hematemesis or motility dysfunction that includes constipation, vomiting, bloating, pseudoobstruction [5] GI amyloidosis is diagnosed usually by upper GI endoscopy and biopsy. Management is aimed at the proper treatment of the primary disease.
Figure 4: Amyloid deposits showing apple green bifringence under the plane-polarized microscope

GI amyloidosis is not an uncommon manifestation of secondary amyloidosis and must not be forgotten while evaluating chronic diarrhea in a patient with chronic inflammatory condition like RA.


