Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Masato Kantake*
Received: March 16, 2018; Published: April 03, 2018
*Corresponding author: Masato Kantake, Neonatal Medical Center, Juntendo University Shizuoka Hospital, 1192 Nagaoka, Izunokuni, Shizuoka, 4102295, Japan
DOI: 10.26717/BJSTR.2018.03.000907
The analysis of nuclear red blood cells (NRBCs) in pregnant woman is believed to be a practical, noninvasive prenatal test that is much useful for understanding the prenatal fetal status. Here, we show a simple, rapid and effective method to separate NRBC from maternal peripheral blood with preterm labor. We used MACS^ Technology which is based on MACS^ MicroBeads, highly specific antibodies coupled to super paramagnetic 50nm particles, MACS Columns containing a matrix of ferromagnetic spheres, and MACS Separators providing a strong magnetic field. This method could enrich the CD45-CD71+ cells from 0.3% of whole blood cells to 93% of separated cells. The yield of NRBCs from 2ml of maternal blood by this method is enough to be used for not only targeted DNA methylavtion analysis but whole exome and whole transcriptome analysis using next generation sequencer which may lead to great progress in perinatology.
It is now widely recognized that the environment in early life have important effects on adult health outcomes through epigenetic programming [1]. It has been thought that there are several developmental stages in which the environment induces the epigenetic modification of DNA. Among them, the prenatal period may be most crucial stage for these programming. Fetal cells have been known to be present in maternal blood since more than a century ago. It is known that each kind of fetal cells appears in maternal blood but there have been very few investigations tracing fetal white cells in the maternal circulation because of the lack of general antibody to distinguish fetal white cells from maternal white cells. Another important restriction of fetal lymphocytes is their longevity which is about 5 years of half-live [2,3].
Therefore, nucleated red blood cells have been considered a suitable target for prenatal diagnosis2. Numerous enrichment technics have been reported for the isolation of fetal NRBC from the maternal circulation as there are FACS, MACS, density gradient, and combination of them [4-8]. However, the extreme scarcity of fetal cells puts strong demands on the sensitivity and specificity. In the present study, we try a simple technology of NRBC separation from maternal blood using MACS technology without density gradient and lysis of RBC.
We tested 2 pregnant women with approvals of the ethical committee of Juntendo University and informed consents. Patientl is an 18years old woman bearing single male fetus. She came to our hospital at 26weeks of gestation to control of preterm onset of labor. Although tocolysis was done, she delivered vaginally a baby after spontaneous rapture of membrane within the day of admission. The blood sample was taken at the time of admission. Patient2 is a 42 years old woman bearing single female fetus. She came to our hospital at 27 weeks of gestation to control of preterm onset of labor and gestational diabetes mellitus. She can avoid a preterm delivery by tocolysis until 34weeks of gestation. The blood sample was taken at the time of admission. These 2 blood samples (2ml each) with anticoagulant were stocked at room temperature and analyzed within 12hours after sampling.
The separation protocol is following the manufacture's instruction.
Briefly,
a) Cell Labeling: We used direct magnetic labeling for anti CD45 (Straight FromTM Whole Blood CD45 MicroBeads human,Miltenyi Biotec GmbH, Gladbach, Germany) which is optimized to be mixed with whole blood, and indirect magnetic labeling for anti CD71. For sorting, anti CD71 primary antibody conjugated with Phycoerythrin (PE) and anti PE MACS MicroBeads.
b) Cell Separation: We performed CD45 negative selection and CD71 positive selection using autoMACS^Pro Separator (Miltenyi Biotec). During positive selection, the target cells are magnetically labeled and collected as the positive fraction. During depletion, the unwanted cells are labeled and depleted from the target cells. We performed CD45 negative sorting followed by CD71 positive sorting sequentially. The efficacy of separation was confirmed by using MACS Quant flowcytometry analyzer 10 (Miltenyi Biotec). The protocol used in this study is available from the author on request.
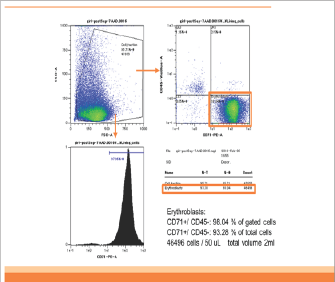
In patient 1, CD45-CD71+ cells were enriched from 0.3% in analyzed cells to 93% in enriched cells (310 folds). The yield of CD45-CD71+ cells was 2.5x106 cells from 2ml of maternal blood (Figurel). In patient 2, CD45-CD71+ cells were also enriched from 0.3% to 93% (310 folds). The yield was 3.7x106 cells from 2ml of maternal blood (Figure 2].
Figure 1a: Pre-Separation (Patient 1).
Figure 1:The result of flowcytometry analysis of patient 1. The FSC/SSC plots and anti CD45 and anti CD71 plots are shown. CD45-CD71+ cells were enriched from 0.3% in analyzed cells (Figure1a) to 93% in enriched cells (Figure1b) (310 folds). The yield of CD45-CD71+ cells was 2.5�106 cells from 2ml of maternal blood.
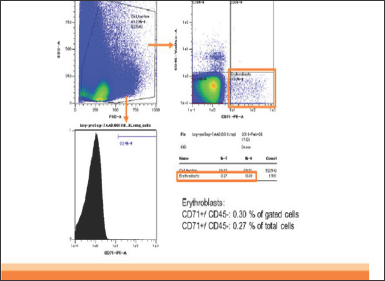
Figure 1b: Post-Separation (Patient 1).
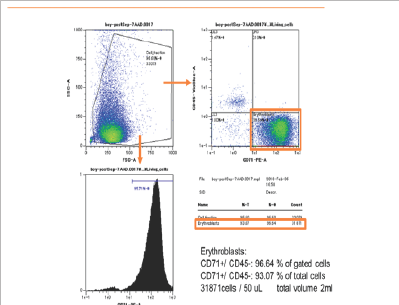
Figure 2a: Pre-Separation (Patient 2).
Figure 2: The result of flowcytometry analysis of patient 2. CD45-CD71+ cells were also enriched from 0.3% (Figure 2a) to 93% (Figure 2b) (310 folds). The yield was 3.7�106 cells from 2ml of maternal blood.
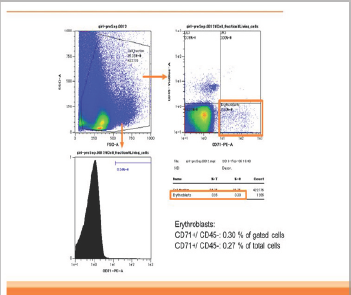
Figure 2b: Post-Separation (Patient 2).
Bianchi et al. precisely investigated the fetal nucleated cells in maternal blood2. According to it, 0.3% of mononuclear cells are transferrin receptor (TfR; CD71) positive in nonpregnant adult, and these cells are heterogenous in size and not coalesce into a discrete population. In contrast, the cord blood samples contained large percentages of CD71+ cells (39%). Contour plot analysis revealed a large population of these cells with intermediate light scattering properties. Samples from the pregnant woman contained more CD71+ cells than the samples from the nonpregnant adults (0.83%). Contour plots revealed the consistent presence of a small but defined cell population which is equivalent to those in cord samples. Erythroblasts have been shown to express the TfR antigen on their cell surfaces from the burst-forming units, erythroid, and stage up until and including nuclear extrusion (reticulocytes) stage. In the cord blood, large numbers of reticulocytes and NRBCs containing fetal hemoglobin were identified visually. In contrast, the samples from the nonpregnant adults consisted of lymphocytes, monocytes. None of the isolated cells in this group contained fetal hemoglobin. In the samples from pregnant women, the sorted cells were overwhelmingly recently enucleated erythrocytes (reticulocytes) the origin of which cannot be determined. NRBC containing fetal hemoglobin were occasionally identified.
Monocytes, lymphocytes containing adult hemoglobin were only infrequently observed. Taken together, CD71+ cells in pregnant woman contains fetal erythroblast and small number of maternal lymphocyte and a certain level of reticulocyte from unknown origin. Our strategy using CD45 negative selection to avoid the contamination of maternal monocytes or lymphocytes prior to CD71 positive selection is thought to be reasonable. Relatively high yield of CD45-CD71+ cells in the present study may reflect the contamination of recently enucleated erythrocytes (reticulocytes). From now on, the SRY gene quantitation of separated cells from male bearing mother's blood is needed. When the fetal cell ratio is high in some extent, our method may be useful for prenatal fetal DNA methylation analysis and for postnatal cell type specific analysis.
The author thanks to Drs. Toshitaka Tanaka and Naho Ikeda for sample collection. The author also thanks to Dr. Souhei Nakayama for technical assistance.


