Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Zavack� M* and Pobehov� J
Received: September 19,2018; Published: October 03,2018;
*Corresponding author: Martina Zavacka, PhD, MpH, Clinic of Vascular Surgery VUSCH, Inc, Ondavska 8, 040 11 Kosice, SR, t.c, Slovakia
DOI: 10.26717/BJSTR.2018.09.001820
Acute limb ischemy is caused by sudden inadequate blood flow of the limbs. It manifests by pain, disturbance of perception and movement of varying intensity and range. 15% cases is caudes by embolism, 85% is caudes by arterial thrombosis. The diagnostic and therapeutic approach depends on the degree of disability. Is expressed the Rutherford classification. Treatment is conservative, endovascular or surgical.
Acute limb ischemia is defined as a sudden inadequate blood circulation of the limbs or a sudden worsening of pre-existing ischemic disease of the limbs, manifested by pain, disturbance of perception and movement of different intensity and range, depending on etiology and localization of artery occlusion.
15% of cases is caused by embolism, 85% of arterial thrombosis, especially in patients with atherosclerotic disease [1].
In the Case of an Embolic Closure: 90% the source is cardiac (atrial fibrillation, wall intracardiac thrombosis in the heart attack,and the embolus of the valves, or the atrial myxoma.) The embolus embusses typically to areas of arterial bifurcation (aortic, femoral bifurcation, and may occur simultaneously in both limbs. The source of distal microembolization is dissrupted atherosclerotic plaques. In 5-10% of distal embolization cases, the source is unknown. With the increasing number of endovascular procedures, iatrogenic embolization increased because of the intra-arterial insertion of the instruments. Paradoxical embolization is, when the formation of thrombotic matter takes place in the venous river, which penetrates the right part of the heart through intracardiac communication (most often foramen ovale) into the artery system and subsequently embolises it (Figures 1 & 2) and (Table 1).
Mortality associated with an acute peripheral artery occlusion is high, averaging 10-25%. The thrombotic type of closure is formed on the basis of a preexisting atherosclerotic throat, with ischaemia manifestations less severe than with a sudden embolic closure, due to the possible presence of collateral circulation. The most common localization of the thrombotic cap of the peripheral artery is the superficial femoral artery [2-4].
Acute thrombotic or embolic closure of peripheral arteries also occurs in connection with arterial aneurysms, most commonly in the area of the popliteal artery. Popliteal aneurysms are degenerative in 90% of cases, and in 60-70% of cases they occur bilaterally. Flegmazia cerulea dolens is a rare cause of ALI, and may occur with a massive deep vein thrombosis that causes severe swelling of soft tissues, resulting in decreased arterial blood flow in the affected limb, and may develop ALI (Figures 3 & 4).
Uncommon causes of acute limb ischemia are drug associated thromboses (oral contraceptives, estrogen replacement preparations, anabolic steroids), hypercoagulable conditions, thrombophil- ic states (malignant processes), aortic dissection, arterial thrombosis due to radiation of activated arteritis [5].
Symptoms of acute limb ischemia include: pain, cold, sensitivity disturbance, movement disorder, paleness of the limb and non-palpation pulses under the closed artery. Exacerbated limb ischemia causes loss of motoric and sensory functions of the limb to stiffen muscles. The paleness of the limb is replaced after several hours by marble, the height of ischemia depending on the localization of the artery cap. In the case of emolition in aortic bifurcation, the development of a shock condition that develops on the basis of severe lower limb ischemia is dominated, and in the case of nasal thrombosis above the embolic closure visceral ischemia develops [6].
In diagnosis we start by collecting anamnestic data on the duration of the condition, subjective difficulties. We continue with physical examination, in particular palpation of the pulsations on the limbs, auscultation, examination of sensitivity and limb movement. Particularly helpful are special instrument examination methods (Table 3) [7].
Color Duplex Ultrasonography: This is a basic imaging examination methodology. Its benefits are financial difficulty, easy reproducibility, it is a non-burdening diagnostic methodology for the patient with a high sensitivity and specificity of 70-90%. This non-invasive methodology informs us about the quality of the vascular wall, lumen artery as well as hemodynamics in the investigated artery. It is capable of detecting signs of atherosclerotic process, characterizing the atherosclerotic plaque, localizing the constriction by determining its haemodynamic severity, diagnosing the thrombotic or embolic closure, and estimating its character (acute, subacute, or chronic) based on echogenicity [8].
CT Angiography: This is a non-invasive imaging methodology Its advantages include minimal invasiveness, ability to evaluate the vascular wall as well as surrounding tissue in detail. Disadvantages include the possible occurrence of allergic reactions, nephrotoxicity and other undesirable effects associated with administration of iodinated contrast agents as well as exposure to ionizing radiation [9] (Figure 5).
Figure 5: CTA finding of in-stent thrombosis in a patient after abdominal aorta stenting and two common pelvic arteries, different types of imaging.
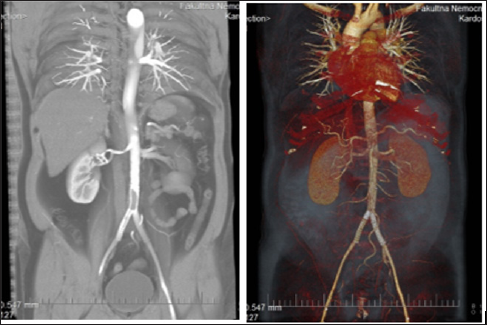
MR Angiography: Thisis a non-invasive investigative methodology providing significant advantages over CT angiography, including the absence of ionizing radiation and the relatively lower nephrotoxicity of gadolinium contrast agents used in MRA [10].
Angiography: It is a semi-invasive diagnostic methodology that offers the possibility of subsequent therapeutic use. Seldinger's catheterization technique, using iodinated contrast agents, obtains accurate data on anatomical proportions and pathological changes in the arteries examined. In the arterial imaging, contrast angiography remains a gold standard, which compares all other investigations of the methodology. Still, standard angiography remains the most accurate imaging algorithm for ALI, although there are cases where the flow to the hemorrhagic and plantar arteries is so slow that the DSA is unable to show the artery. An irreplaceable advantage is that, in addition to diagnostic value, angiography can also be used for subsequent therapy [11].
Conservative Treatment: In clinical practice, it is rarely applied and is left only for cases where the symptoms of ischaemia are mild or transient, and the artery is localized in the inferior limb or, in the case of high-risk patients who can not manage endovascular or surgical performance, and who are not an acute ischemia of the limbs threatening to live.
Anticoagulation Therapy: The most important treatment point in the conservative procedure is the administration of a primary dose of heparin that prevents the progression of thrombosis in the peripheral region and slows down irreversible ischemic changes. The dosage of heparin is dependent on the weight and overall clinical status of the patient. If we use the unfractionated form of heparin as an initial dose, we apply 70 IU / kg, then we continue with the continuous intravenous administration of heparin at 15 IU / kg / hr. In order to maintain therapeutic anticoagulation, we monitor the aPTT level, which should not deviate from the recommended range of 50-85 sec. A great advantage of using LMWH is that it is weakly reacting with platelets.
Vasoactive drugs and rheology: pentoxifylline, naphthydofuryl, prostaglandins
The pain in the ischemic limb is so pronounced that it often does not respond to drug therapy. We can share painkillers: I. Non-steroidal anti-inflammatory drugs - ibuprofen, paracetamol, metamizole. II. Opioid analgesics for mild to moderate pain - codeine, dihydrocodeine, tramadol, pethidine. Opioid analgesics for severe pain - morphine [12,13].
Thrombolytic Therapy: Is endovascular treatement. It means percutaneous, catheter-directed local continuous intra- arterial thrombolysis (LKT), which is an accepted and less invasive treatment method compared to a surgical solution. The aim is to restore flow in the affected artery, to uncover the lesion (stenosis) if present and to restore collateral circulation. There is no damage to the small arteries, endothelium respectively (thrombogenesis, vasospasm), while still being able to convert to angiosurgery if necessary.
a) Diagnosed cerebrovascular accident (including TIA for the last 2 months).
b) Active bleeding.
c) Fresh bleeding into the gastrointestinal tract over the last 10 days.
d) Neurosurgical performance (intracranial, spinal) over the last 3 months.
e) Intracranial trauma over the last 3 months.
a) Cardiopulmonary resuscitation during the last 10 days.
b) Large non-vascular operations or trauma during the last 10 days.
c) Uncontrolled hypertension (systolic blood pressure> 180 mmHg, diastolic> 110 mmHg).
d) Puncture of an uncompromising artery.
e) Intracranial tumor .
f) Recent eye surgery.
a) Hepatic failure associated with coagulopathies.
b) Bacterial endocarditis.
c) Pregnancy.
d) Diabetic hemorrhagic retinopathy.
Percutaneous aspiration thrombectomy is one of the oldest endovascular techniques where we aspire (aspirin) the thrombotic and embolic masses from the artery. The aspiration catheter is placed in close proximity to the thrombus, then manually creates a vacuum in the syringe. As a result of the vacuum, the thrombotic and embolic masses are sucked through the aspiration catheter into the syringe. Indications for PAT are fresh, non-adherent thrombotic matter or emboli.
New endovascular mechanical thrombectomy systems include thrombotic fragmentation/aspiration, isolated pharmaco- mechanical thrombolysis, and ultrasonic accelerated thrombolysis. Percutaneous mechanical thrombectomy (PMT) can be very useful as a primary, as an adjuvant therapeutic alternative for patients with ALI. The advantage of PMT is the ability to quickly and successfully remove thrombotic matter, significantly reducing the duration of ischemia.
The indication of surgical revascularization depends on the clinical stage of ALI, the patient's co-morbidities and the etiology of acute limb ischemia. Embolic closures can often be resolved by embolectomy, while thrombotic closure may be endarterectomy, more often bypass reconstruction. The operation in most patients with the embolic closure of the marginal artery of the lower limb may be initiated by transfemoral approach through arteriotomy in the femoral artery, allowing for vascular access for balloon catheter thrombectomy from aortic bifurcation to ankle. Rarely, it is necessary to provide a knee-jerk approach. In the absence of the outflow tract or the absence of a peripheral Doppler signal after thromboembolism, it is necessary to perform angiography of the affected limb in order to consider a further procedure. Angiography after surgery is generally recommended, unless the embolus has been completely removed and the patient has palpable peripheral pulsations.
Amputation: The need for amputation for ischemia of less than 4 hours is 3%, for ischemia between 4-6 hours about 10%, for 6-12 hours about 25% and for ischemia lasting for more than 12 hours, the risk of amputation is increased to 50-60 %.
Necrotic Rhabdomyolysis: For prolonged lower limb ischia, cross-striped muscle necrosis occurs, which then releases large amounts of myoglobin, potassium and hydrogen ions. As a result of the processes described, there is a metabolic disorder that causes myoglobinuria and acute renal failure, hyperkalaemia with heart rhythm disorders.
Compression Syndrome: Compartment syndrome causes pressure to rise in closed fascial spaces and consequently to functionally and morphologically affect the vascular, nerve and muscle structures within the compartment. Clinically, the compartment syndrome is manifested by severe pain in the fascicular fascicula of the affected area, the swelling of the foreleg, and the sensory or motor deficiency of the limb may also appear. In non-traumatic acute limb ischemia, the development of compartment syndrome is approximately 8%, whereas traumatic endemic ischemia is reported to occur in up to 30% of cases. The medication treatment of the compartment syndrome is the application of anti-edematous agents - 20% Mannitol i.v., aspiration of diuresis and correction of the internal environment. Depending on the clinical condition, fasciotomy of the anterior, lateral or posterior compartment may be performed; in serious cases, the fasciotomy of several compartments may be made at the same time (Figure 6).
A 64-year-old man, a long-term active smoker (5-6 per day), the hypertonic was in urgent mode transferred from the Surgery Department to the Clinic of Vascular Surgery, with sudden weakness and restlessness of both lower limbs. He had a history of two-week walking pain in his anamnesis. On arrival, the patient was afebrile, both of the lower limbs were cold, pale, with marbled skin and developed sensomotor deficit. Based on the input ultrasonographic examination of the vessels and the clinical picture, the condition was classified as an ALI stage Rutherford II b, requiring urgent operational review. In total anesthesia, thrombectomy of iliac arteries right, femoral, popliteal and hemorrhagic arteries on both sides, supplemented by endovascular treatment: mechanical thrombectomy, with implantation of stentgraph into and. iliaca external to the right, percutaneous transluminal angioplasty (PTA) and stent to a. iliaca communis on the left with a beneficial effect (Figure 7).
Patient after procedure, in spite of complex care, muscle necrosis occurred in the left anterior forearm, therefore a revision of the pre-collector arteries with the necessary amputation in the right thigh was indicated. The postoperative course was further complicated by hepato-renal failure in reperfusion syndrome, with a probable proportion of contrast-induced nephropathy. During the next treatment days, the condition stabilized, allowing gradual rehabilitation. The patient was transferred to the gradient surgical department to continue conservative therapy, but a re-deterioration of the medical condition occurred. As a result of complications, the patient dies. After the completion of our hospitalization, we obtained definitive results of a histopathological examination of the thrombotic arteries of the lower limb arteries, where, after repeated examination, including immunohistochemistry, a recent mixed thrombus and a largely uncorrotically altered malignant mesenchymal neoplasm was identified, the exclusion being malignant fibrous histiocytoma (MFH) (7).
A 65-year-old patient, in the past after ablation of the right breast cancer, after repeated chemotherapy and radiotherapy, was admitted to the Clinic of Vascular Surgery, VUSCH, a.s. for weekly worsening of cold, pain and partial numbness of the entire right upper limb (PHK). According to the patient's history, the condition developed / deteriorated within 3 months, during which there was also a significant decrease in weight. On receipt PHK was cold, fingers livid, with minimal sensitivity, still retaining momentum that was painful. Pulses for a. brachialis, a.radialis and a. ulnaris, or palpable. Pulse was palpable only for a. axillaris, which also confirmed the ultrasound finding of the thrombembol of the older data, coming from the transition and. subclavian / a. axilaris to the right, distal to the right. After the necessary preparation, the patient was operated, we extracted a large number of old but also fresh thrombi to the central and peripheral, but one of the peripheral arteries was already chronically closed (Figure 8).
Postoperatively PHK was perfused in its entirety, with palpable pulses in its entirety, with no movement or sensitivity. However, there was a pronounced reperfusion injury, combined with PHK chronic lymphoedema. Thrombembols sent for histological examination confirmed recurrence of the tumorous process, the patient was consulted with a clinical oncologist, treated therapist and subsequently dismissed from our Clinic.
Mortality associated with an acute peripheral artery occlusion is high, averaging 10-25%. The thrombotic type of closure is formed on the basis of a preexisting atherosclerotic throat, with ischaemia manifestations less severe than with a sudden embolic closure, due to the possible presence of collateral circulation. The most common localization of the thrombotic cap of the peripheral artery is the superficial femoral artery. The treatment options depend on the degree of ischemia and the overall condition of the patient. Complications of acute limb ischemy threaten the patient with loss of limb and loss of life.
Acute limb ischemy is caused by sudden inadequate blood flow of the limbs. It manifests by pain, disturbance of perception and movement of varying intensity and range. 15% cases is caudes by embolism, 85% is caudes by arterial thrombosis. Symptoms of acute limb ischemia include: Pain, cold, sensitivity disturbance, movement disorder, paleness of the limb and non-palpation pulses under the closed artery


