Abstract
The photophysical property, cytotoxicity of hematoporphyrin derivative BL-1 in vitro and in vivo were investigated as photosensitizer for photodynamic therapy (PDT). Strong absorption and high singlet oxygen quantum yield of compound BL-1 were exhibited. Significant phototoxicity and inhibition to tumor were also showed. So compound BL-1 was suggested as the potential and promising antitumor photosensitizer for PDT.
Keywords: Photosensitizer, Photodynamic therapy, Hematoporphyrin derivative, Tumor
Abbreviations: PDT: Photodynamic Therapy; PS: Photosensitizer; HPD: Hemato porphyrin Derivative; HMME: Hematoporphyrin Monomethyl Ether
Short Communication
Photodynamic therapy (PDT) is an emerging non-invasive treatment based on a combination of photochemistry and photophysics. PDT has more advantages compared with traditional chemotherapy and surgical treatment, which is more targeted, less harmful to healthy tissue [1-4]. When the photosensitizer (PS) molecule is illuminated by a light of particular wavelength, it passes from the first excited singlet state to the first excited triplet state T1, which will initiate a photochemical reaction by generating reactive free radicals or transferring its energy to the ground state oxygen molecule (3O2) to produce singlet oxygen (1O2) that can oxidate the bio-molecules of diseased cells and apoptosis or necrosis was resulted [5,6]. Photodynamic therapy was discovered nearly 100 years ago but medically approved drugs and technologies are still limited [7,8].
The first photosensitizer used in clinics was Hematoporphyrin Derivative (HPD) which was called Photofrin or porfimer sodium. It is a mixture of six hematoporphyrin derivatives with long retention in skin which resulted in that patient need to avoid the light for 4 weeks. Hemeporfin (HMME) is a mixture of two enantiomers which was first discovered by Xu [9]. HMME has better performance than porfimer sodium. It shows low toxicity to normal tissues with rapid metabolism property. For nearly a century, the design and synthesis of hematoporphyrin derivatives are still of concern to many scientists. Here the photophysical property, cytotoxicity of new hematoporphyrin derivative BL-1 in vitro and in vivo were investigated As Photosensitizer for Photodynamic Therapy (PDT).
Results and Discussion
Photophysical Properties
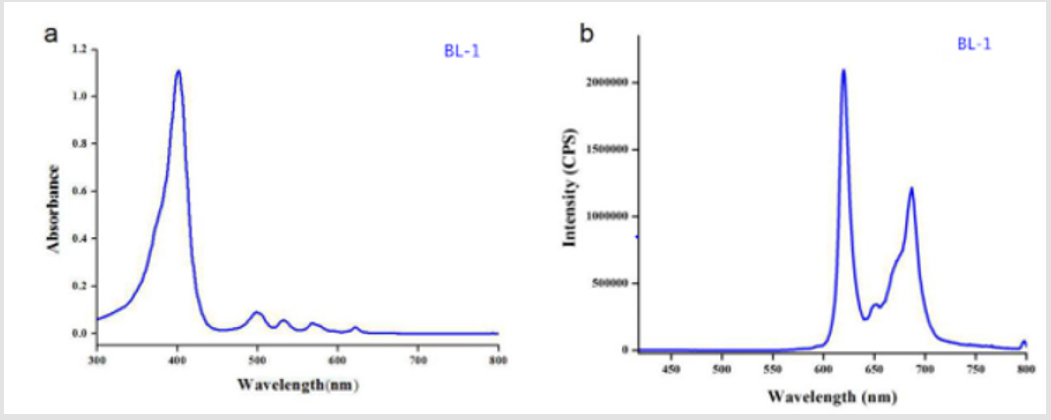
Compound BL-1 was dissolved in DMSO. UV-vis spectrum and fluorescence emission spectrum of Compound BL-1were investigated. As shown in (Figure 1) BL-1 had the characteristic soret and Q band absorptions at 401nm (soret), 498nm, 532nm, 569nm and 622nm (Q band), respectively. Compound BL-1 displayed a fluorescence excitation band at approximately 400nm and emission band at 620nm. HMME was chosen as the control compound.
Figure 1: UV-visible absorption and fluorescence spectra of BL-1.
(a) Uv-vis absorption spectrum of BL-1 (10 μM in DMSO).
(b) Fluorescence emission spectrum of BL-1 (10μM in DMSO).
Singlet Oxygen
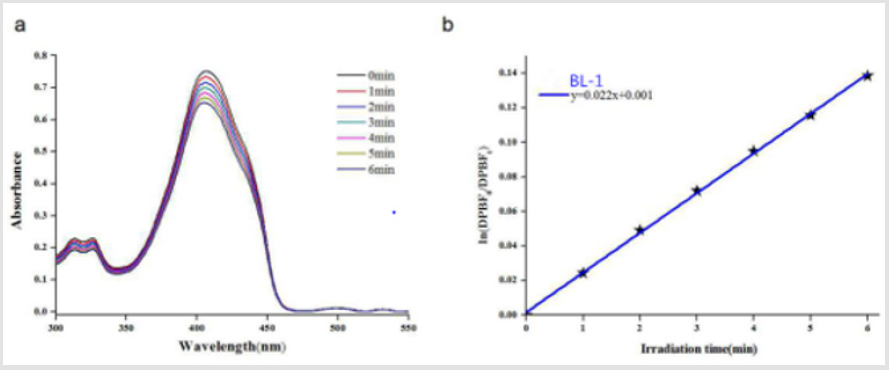
The efficiency of singlet oxygen can be used to evaluate the cytotoxicity of the photosensitizer. The singlet oxygen generation of the photosensitizer is detected by DPBF chemical oxidation method for the singlet oxygen capacity of the photosensitizer. The slope of the function is obtained from a plot of ln [DPBF0]/[DPBFt] versus illumination time t. As shown in (Figure 2), the absorption intensity of DPBF (λ = 410nm) continuously decreased in the presence of BL-1 as the irradiation time increasing. The rate of singlet oxygen generation was 0.0022min-1.
Figure 2: (a) Absorption spectra for the photodecomposition of DPBF by 1O2 after irradiation of compounds BL-1. (b) First-order plots for the photodecomposition of DPBF photosensitized by compounds BL-1.
MTT Assey
High phototoxicity and low dark toxicity are important factors for evaluating the effectiveness of photosensitizers. The MTT assay was used to evaluate the phototoxicity and dark toxicity of the compounds BL-1. The control group was set with different concentrations of DMEM in DMSO. The data demonstrated that DMSO had no effect on the survival rate of experimental cells (data not shown). At the same dose of light, drug concentration was inversely correlated with cell viability. At the same concentration of PS, the light dose was also negatively correlated with cell viability.
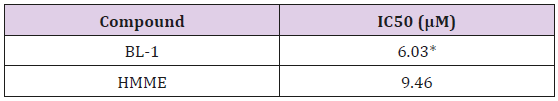
The IC50 (the concentration of a photosensitizer inhibits 50% of the cells under light) values were shown in (Table 1). The IC50 value of BL-1 to Eca-109 tumor cells was smaller than HMME. These results suggested that BL-1 was a promising photosensitizer in vitro.
Table 1: Phototoxicity for BL-1 and HMME toward Eca-109 cells.
Note: Visible light 622 nm, light dose 16 J/cm2.
*P < 0.05 represents a significant difference relative to the control
group
Photodynamic Activity In Vivo
PDT preferentially damages photosensitizer target cells. This statement can be explained by the current research about HIF mechanism. When the tumor site is hypoxic, HIF is highly expressed. This process promotes the growth of blood vessels and red blood cells at the tumor site. Porphyrins are coenzymes of red blood cells. Therefore, it will preferentially accumulate in the tumor site, and has a certain tumor targeting effect [10-12]. The antitumor activity of compound BL-1 was evaluated on Balb/c nude mice bearing Eca- 109 tumor. Tumor inhibition rates were measured by calculating the weight of nude mice tumors. The experiment was divided into the control group, the light group, the PS group and PDT group. The tumor weight of the nude mice gradually increased in the control group, the light group and the photosensitizer group. The weight of tumors is gradually decreasing as the photodynamic therapy group grows with time. Tumor inhibition rate is shown in (Table 2). It is suggested that the compound BL-1 exhibited efficient photodynamic antitumor efficacy on Balb/c nude mice at lower concentration. Therefore, BL-1 is a promising antitumor PS in PDT application.
Table 1: The inhibition rate of phototoxicity for BL-1 toward tumor inhibition rate.
Note: *P < 0.05, ***P < 0.001 represents a significant difference relative to the control group.
Material and Methods
Instruments and Reagents
All the chemicals and reagents were obtained from Sinopharm Chemical Reagent Co., Ltd. and Bide Pharmatech Ltd., and used without further purification. All solutions were freshly prepared with deionized water. The UV-vis absorption spectrum was recorded on an ultraviolet-visible spectrophotometer (Model V-530, Japan). Fluorescence spectrum was recorded on a fluorescence spectrophotometer (FluoroMax-4, France).
Singlet Oxygen Determination
The singlet oxygen capacity of the photosensitizer was measured by DPBF chemical oxidation. A DMSO solution containing 0.5μM PS and 20μM PBF was irradiated with a laser intensity of 622nm at 5mW/cm2, and the natural logarithm of DPBF absorption at 410nm was plotted and fitted by a first-order linear least squares model to obtain a singlet state of the photosensitive process. The rate constant of the methylene blue sensitized DPBF photooxidation reaction was converted to the 1O2 quantum yield [13].
Cell Lines and Culture Conditions
The cells were obtained from the Cell Culture Center of the Chinese Academy of Sciences. The cell-related reagents were purchased from Shanghai Mingrong Biotechnology Co., Ltd. The cells were cultured in 10% Fetal Bovine Serum (FBS), 50mg/mL penicillin and 50mg/mL. In medium culture, 5% carbon dioxide was added to a humidified incubator at 37oC. Cells in the exponential growth phase were used in each experiment.
MTT Assay
The experiment of cell phototoxicity and dark toxicity was performed using the MTT assay. Cells of various concentrations were cultured in DMEM medium containing 10% (v/v) FBS and allowed to ingest for 24 hours in the dark. Cell viability was assessed by 3-(4, 5-dimethyl-2-thiazolyl)-2, 5-diphenyl-2H-bromide triazole (MTT) colorimetric assay 24 hours after treatment.
In Vivo Experiments
When the size of the tumor reached about 100 mm3 (about 14 days after inoculation), the tumor-bearing mice were divided into 4 groups containing 4 mice per group: control group, light (without drug), PS (without light) and PS-PDT (with drug and light). After PDT, tumor regeneration or healing of the mice was observed daily.
Conclusion
The photophysical and photochemical properties of new hematoporphyrin derivative BL-1 were determined. In vivo and in vitro tests were performed to evaluate the ability of compound BL-1 to destroy tumor. The compound exhibited long maxima absorption wavelengths and higher phototoxicity compared to HMME. BL-1 exhibited better inhibitory effect on Eca-109 tumor cells than HMME. Therefore,compound BL-1 could be a potential anti-tumor photosensitizer in photodynamic therapy.
Acknowledgment
This work was supported by the National Natural Science Foundation of China (No. 21977016), Foundation of Shanghai Science and Technology Committee (No. 17430741800, 17430711900, 18430713000, 18430731600;19410711000).
References
- Jonathan P, Conor L, Kimberley S, Brian W, Tayyaba Hasan, et al. (2010) Imaging and photodynamic therapy: Mechanisms, monitoring, and optimization. Chem Review 110(5): 2795-2838.
- Awan MA, Tarin SA (2006) Review of photodynamic therapy. Edu Review 4(4): 231-236.
- Taub AF (2007) Photodynamic therapy: other uses. Derm Clinics 25(1): 101-109.
- Sobotta L, Sniechowska J, Ziental D (2019) Chlorins with (trifluoromethyl)phenyl substituents- Synthesis, lipid formulation and photodynamic activity against bacteria. Dyes and Pigments 160: 292-300.
- Caminos DA, Durantini EN (2005) Synthesis of asymmetrically meso-substituted porphyrins bearing amino groups as potential cationic photodynamic agents. J. Porphyrins Phthalocyanines 9: 334-342.
- Allison RR, Sibata CH (2010) Oncologic photodynamic therapy photosensitizers: A clinical review. Photodiagnosis Photodyn Ther 7(2): 61-75.
- Abrahamse H, Hamblin MR (2016) New photosensitizers for photodynamic therapy. Biochem J 473(4): 347-364.
- Wang L, Cao D (2012) Research advances of porphyrin photosensitizers in photodynamic therapy. Chin J Org Chem 32(12): 2248-2264.
- Xu DY, Chen WH, Zhang H (1993) Study on photodynamic therapy of hematoporphyrin monomethyl ether (HMME). Chin J Laser Med (1): 3-7.
- Xie J, Cheng C, Jie Y, Ma H, Guo Z, et al. (2019) Expression of lactate dehydrogenase is induced during hypoxia via HIF-1 in the mud crab scylla paramamosain.Comp Biochem Physiol. Part C 225: 108563.
- Khan MS, Hwang J, Lee K, Choi Y (2019) Anti-tumor drug-loaded oxygen nanobubbles for the degradation of HIF-1alpha and the upregulation of reactive oxygen species in tumor cells. Cancers 11(10): 1464.
- Nishide S, Matsunaga S, Shiota M, Yamaguchi T, Takehiro Yamaguchi, et al. (2019) Controlling the phenotype of tumor-infiltrating macrophages via the PHD-HIF axis inhibits tumor Growth in a Mouse Model. iScience 19: 940-954.
- Spiller W, Kliesch H, Wohrle D, Hackbarth S, Günter Schnurpfeil, et al. (1998) Singlet oxygen quantum yields of different photosensitizers in polar solvents and micellar solutions. J Porphyrins Phthalocyanines 2(2): 145-158.

 Short Communication
Short Communication