Abstract
Abbreviations: BL: Burkitt’s Lymphoma; CBC: Complete Blood Count; HU: Hydroxyurea; EBV: Epstein Barr virus; MI: Myocardial Infarction; CNS: Central Nervous System; CR: Cure Rate; EFS: Event-Free Survival; LDH: Lactate Dehydrogenase Enzyme; CD: Cluster Differentiating Factor; ALL: Acute Lymphocytic Leukemia; DLBL: Diffuse Large B Cell Lymphoma; NG: Nasogastric; GI: Gastrointestinal; HAART: Highly Active Antiretroviral Therapy
Introduction
Burkitt’s lymphoma is one of the most aggressive non-Hodgkin B cell lymphomas. Usual presentation in sporadic Burkitt’s lymphoma which is the prevalent form in the USA is abdominal symptoms 30-38% and CNS involvement in 13-17% cases [1,2]. The patient’s first presentation with leptomeningeal involvement is rare and carries a poor prognosis if left untreated due to comorbidites [3].
Case Description
A 70-year-old male presented with complaints of headache and blurry vision for 3 days. He was seen and evaluated for toothache at the dentist’s office and was given amoxicillin/clavulanic acid one week ago. After the intake of the first dose, he started experiencing blurry vision. Besides that, he noticed petechiae started appearing on his lower extremities. He reported an episode of nosebleed lasting for 12 hrs. a few weeks back. A review of the system was negative for weight loss and positive for fatigue. Vitals signs were stable. Physical exam revealed distended belly with splenomegaly. CBC was significant for WBC count of 47 k with a peripheral smear showing 70% blasts. Other significant labs included Hgb 9 mg/dl, uric acid 19, platelets 11k. The patient was admitted for workup of leukemia/ lymphoma with blast crisis and tumor lysis syndrome. He was initially treated with Hydroxyurea (HU). Over the next 2 days, his leukocytosis persisted for which HU dose was doubled. His symptoms of headache, nausea and blurry vision continue to get worse. CT abdomen without contrast showed splenomegaly but no evidence of lymphadenopathy. CT scan head without contrast did not show any acute intracranial abnormality.
Ophthalmological evaluation revealed left VI nerve palsy and suggested left eye patch application. Leukemia/lymphoma with meningeal involvement does present with cranial nerve palsy. Lumbar puncture could not be performed due to low platelets. As part of the leukemia workup, flow cytometry of peripheral smear was done, and bone biopsy performed. Seven hours after the bone biopsy was performed under local anesthesia, the patient’s clinical condition got deteriorated rapidly. He was transferred to ICU for tachypnea and altered mental status. The patient was intubated and started on the vasopressor’s support. He was started on broad-spectrum antibiotics. Due to low platelets, bleeding was observed from the NG tube and he was started on proton pump inhibitor. The patient was transfused cryotherapy, platelets, packed red blood cells. Steroids were added. The patient develops shock liver later during the hospital course. His oxygen requirements remained high, and later bronchoscopy revealed right main bronchus hemorrhagic mucous plugs with mucosa showing diffuse petechiae. His flow cytometry and bone biopsy revealed Burkitt’s lymphoma. Given old age, a recent history of MI, ventilator support and unable to tolerate the aggressive chemotherapy, the decision was made regarding comfort care. The patient was terminally extubated on the 7th day of admission.
Discussion
Burkitt’s lymphoma is one of the most aggressive tumors with a tumor doubling time of 25 hours. It has 3 subtypes based on epidemiology and clinical presentation. Of note, the histologic pattern of all the 3 subtypes remains the same.
Endemic Variant
Found in the equatorial distribution of African countries, strongly associated with EBV virus infection. High malaria prevalence in these countries leads to increase cell turnover making them more prone to genetic instability. Usually seen in African children 4–7 years old, with a male: female ratio of 2:1. It involves the bones of the jaw and other facial bones, as well as kidneys, gastrointestinal tract, ovaries, breast, and other extranodal sites. The incidence is estimated to be 50 times higher than in the U.S. EBV is found in nearly all cases.
Sporadic
It includes those cases occurring with no specific geographic or climatic association. It accounts for 1% –2% of lymphoma in adults and up to 40% of lymphoma in children in the U.S. and western Europe.
Immunodeficiency Associated BL
Not associated with low CD 4 counts of less than 200/cubic
mm. BL accounts for 30-40% of non-Hodgkin’s lymphomas in HIV
patients. Before HAART, the incidence was 1000 times more compared
with non-HIV patients. Usually, patients are not diagnosed
with HIV and hence the discovery of BL is considered one of the
AIDS-defining conditions. HIV-associated Burkitt’s lymphoma
shares some pathogenetic features with endemic Burkitt’s lymphoma.
HIV infection, analogous to malaria, leads to polyclonal B cell
activation of EBV+ cells. Genetically unstable EBV +/- infected B
cells lead to Myc gene rearrangement finally leading to clonal proliferation
of B cells. HIV is indirectly involved in lymphoma development
by chronic antigenic stimulation, decrease immune surveillance
and cytokine dysregulation. The most common sites involved
are lymph node, bone marrow and extranidal sites with abdomen
most common.
Immunodeficiency associated BL is seen after prolong transplant
interval, approx. 5 yrs. per one case series, most of the patients
were solid organ transplant recipients, others were stem cell
transplant. EBV was also common in this subset of patients.
In the USA and Europe, the sporadic variant is prevalent with an
average incidence of 3 cases per 100 million population, accounts
for 1-2% of all lymphoma with male to female ratio of 3:1. [2,4] BL
can have a nodal or extranodal presentation. Extranodal presentation
seen in 80% cases, with 2 quarters involving GI tract and 1
quarter affecting head and neck region. The usual presentation of
Burkitt’s lymphoma is in the form of abdominal pain, nausea, vomiting,
abdominal mass, GI bleed and syndromes mimicking acute
appendicitis, small bowel obstruction or intussusception, perforation.
An abdominal presentation involving intestines ( jejunum, ileum,
cecum, stomach), lymph nodes, spleen, liver, kidney, pancreas,
breast, and ovaries have been reported in the literature. At the time
of diagnosis, patients have high tumor burden from bulky disease
and have elevated lactate dehydrogenase levels, with high uric acid
levels. Cases regarding atypical presentations such as BL mimicking
Cronh’s disease, origin from the nasal septum, gingival involvement,
Kruckenberg tumor by ovarian infiltration with spread to
stomach and presentation with peritoneal carcinomatosis has been
described in the literature [5-7].
The median age of presentation is 30 years. Rarely Burkitt’s
lymphoma can present with CNS or bone marrow involvement and
with leukemia presentation. WHO classified both forms of tumors
as one clinical entity?
2 different types of classification schemes are used.
a. An Arbor: in most adult trials are based on this classification.
The downside is that it does not describe the full extent of
extranodal involvement
b. St Jude or Murphey Classification: This classification considers
leukemia as a separate entity, unlike WHO classification.
Also, it is based on surgery intervention for diagnostic and therapeutic
purposes based on the debulking of Burkitt’s lymphoma
[4].
Today, the treatment of Burkitt’s lymphoma/leukemia is based
on aggressive chemotherapy even in patients with CNS and bone
involvement. Surgery is now not considered a required step in the
management of the Burkitt’s lymphoma due to better aggressive
chemotherapy regimens being available and increase local complications
risk [4]. Rarely, patients present with disease that is primarily
leukemic (classified as acute lymphoblastic leukemia [ALL], L3
type in the French American-British [FAB] Classification) Burkitt’s
lymphoma regardless of subtype expresses typically expresses pan
B cell lymphocyte surface antigen e.g. CD 19, CD20, CD 22, CD79a
and co-express CD 10, Bcl 6, CD 43, P 53 but not CD5, CD23, Bcl-
2, Tdt. Proliferation fraction is nearly 100% with doubling time
24-48 hours [5]. One of the characteristic of Burkitt lymphoma is
the translocation between the c-myc gene and the IgH gene (found in 80% of cases [t(8;14)]) or between c-myc and the gene for either
the kappa or lambda light chain (IgL) in the remaining 20%
[t(2;8) or t(8;22), respectively]. Other specific lymphoma-associated
translocations, such as IgH / BCL-2 and translocations involving
BCL-6 are absent. The myc/Ig translocation is detected by performing
fluorescence in situ hybridization (FISH) or long segment polymerase
chain reaction may increase the chance of identifying the
translocation.
Sporadic Burkitt’s lymphoma, most presenting as leukemia,
one-third of cases had chromosomal breakpoints in the joining
region of IgH [5]. Criteria for diagnosis BL include immunohistochemical
stains CD 20 (+), CD10 (+), BCL-6 (+), and BCL-2 (− )
and a proliferative fraction> 95% as per WHO classification [6].
c-Myc gene translocation can be present; however, 10–15% BL can
be c-Myc negative. Features such as pleomorphic nuclei and BCL2
positivity are features of double-hit lymphomas or B-cell lymphomas,
unclassifiable with intermediate features between diffuse
large B-cell lymphoma (DLBCL) and BL. Also, DLBCL usually shows
a Ki-67 proliferation rate of about 65–70% (in BL it is essentially
100%). In addition, CD44 is most likely to be expressed in CD10+
positive DLBCL and its negative in BL [5]. In our patient FISCH analysis
for high grade/large b cell lymphoma was negative for Bcl6,
Myc, t 8:14, T 14:18. Bone marrow was hypercellular around 80%,
with near-total replacement by malignant population of lymphoid
cells with large nuclei, finely granular chromatin, occasionally distinct
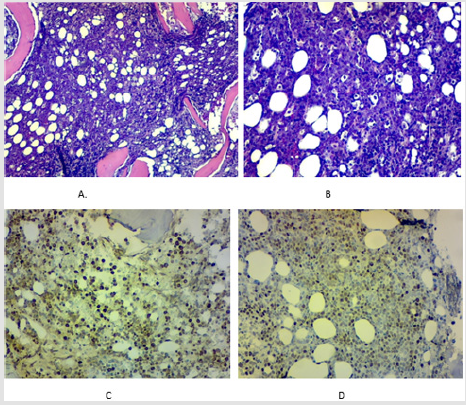
nucleoli and scant cytoplasm. Frequent mitotic (Figure 1)
with apoptotic cells imparting starry sky appearance were present.
Trilieage hematopoiesis was virtually absent. Peripheral smears
showed blasts and smudge cells. Out of 17 immunohistochemical
stains results were as follows:
Figure 1: A. H&E stain of the bone marrow biopsy showing
heavy infiltration of the bone marrow with leukemic cells.
B. shows starry sky pattern with empty spaces
surrounding macrophages due to increase phagocytosis of
the abnormal leukemia cells.
C. anti-c-my stain heavy staining pattern of the
leukemic cells.
D. Ki-67 stain heavy positivity staining.
CD45, CD20,CD10,Pax5 +
CD 138 patchy positive predominantly weak
BcL2, bcl6, sox 11, TDt negative
c-Myc + approx. 90%
IGM+
MUM1: negative
Ki-67 Near 100%.
The final impression was high-grade B cell lymphoma most
likely consistent with Burkitt’s lymphoma with lymphoma cells
nearly 100% of the bone marrow cells.
Flow cytometry analysis of blood showed CD10+ high-grade B
cell lymphoma. Negative prognostic factors in BL are as follows.
a. CNS involvement found by CN palsy or presence of blasts
in CSF
b. Bone marrow involvement
c. Delayed presentation and diagnosis
d. Old age
e. Advance stage
f. Poor performance status
g. Failure to achieve a cure rate
h. Bulky disease, high LDL
i. IN children age > 15 yrs. [6-9].
Short term multi-regimen chemotherapy such as CALGB 10002,
CODOX-M/IVAC, EPOCH, and Hyper CVAD with rituximab is highly
effective with a complete remission rate up to 90%. In a small study
involving 19 HIV-free patients, the use of a less toxic dose-adjusted
EPOCH (etoposide, prednisone, vincristine, cyclophosphamide, and
adriamycin) plus rituximab (DA-REPOCH) led to an event-free survival
of 96% and overall survival of 100% [5]. In adults, good outcomes
have been achieved, with CR rates of 65%–100% and overall
survival (OS) rates of 50%–70%. The institution of the CODOX-M/
IVAC regimen (Magrath protocol)-two cycles of CODOX-M (cyclophosphamide,
vincristine, doxorubicin, high-dose methotrexate,
and intrathecal therapy) alternating with IVAC (ifosfamide, etoposide,
high-dose cytarabine, and intrathecal therapy)-for high-risk
disease, and for those with low-risk disease (e.g., one extranodal
site or completely resected abdominal disease with normal LDH),
three cycles of CODOX-M. Children and adults treated with this regimen
had similar outcomes; the EFS rate at 2 years was 92% for
the group as a whole. However, there were significantly associated
toxicities, including frequent myelosuppression, mucositis, neuropathy,
and some treatment-related deaths.
To counteract these toxicities numerous changes in protocols
have been made with good outcomes. New therapies are required to minimize toxicities, especially in older patients. Relapses remain
a problem, particularly among higher-risk patients. Novel therapies
that have not been tested but could be useful include those
targeted against the c-myc gene, DNA methyltransferase inhibitors,
proteasome inhibitors, and cyclin-dependent kinase inhibitors. Patients
presenting with the leukemic disease as in our case, previously
classified as ALL, L3 type. traditional therapies used to treat
lymphoblastic leukemia worked poorly. Newer chemotherapeutic
regimens are associated with a better outcome but that too comes
at the expense of serious toxicities. Our case is unique due to the
fact that this patient did not have any abdominal presentation. Even
imaging such a CT scan abdomen did not reveal any lymphadenopathy.
His MRI brain did not suggest any leptomeningeal involvement,
but his cranial nerve VI palsy highly supported the brain involvement.
Due to advance age, high bulk of disease, several co-morbid
conditions e.g. MI, acute renal failure, acute respiratory failure, and
ventilator dependency, we did not proceed with the chemotherapy
options. Despite the fact that even with CNS involvement there has
been successful remission achieved, in this unique case, the risk of
chemotoxicity outweighs the benefits of chemotherapy treatment.
Conclusion
Burkitt’s lymphoma in old age with CNS involvement is a rare presentation with very poor prognosis and non-promising outcomes without intervention.
References
- Treatment of Burkitt leukemia/lymphoma in adults – UpToDate.
- E Sariban, B Edwards, C Janus, I Magrath (1983) Central nervous system involvement in American Burkitt’s lymphoma. J Clin Oncol 1(11): 677-681.
- JL Ziegler, AZ Bluming, RH Momtow, L Fass, PP Carbone. Central Nervous System Involvement in Burkitt’s Lymphoma
- KA Blum, G Lozanski, JC Byrd (2004) Adult Burkitt leukemia and lymphoma. Blood 104(10): 3009-3020.
- JA Ferry (2006) Burkitt’s Lymphoma: Clinicopathologic Features and Differential Diagnosis. Oncologist 11(4): 375-383.
- S Gurzu, T Bara, TJ Bara, M Turcu, CV Mardare, et al. (2017) Gastric Burkitt lymphoma. Medicine (Baltimore) 96(49): e8954.
- P Naik, J Wang, MJ Brazeau, D Rosario (2016) An Atypical Presentation of Sporadic Jejunal Burkitt’s Lymphoma. Case Rep Gastrointest Med 2016: 1-4.
- G Aslan (2013) Unusual presentation of sporadic Burkitt’s lymphoma originating from the nasal septum: A case report. J Med Case Rep 7(1): 60.
- F Ziade, N von der Weid, M Beck Popovic, A Nydegger (2012) Burkitt's lymphoma-An atypical presentation. BMC Pediatr 12(1): 617.

 Case Report
Case Report