Research Article
Structure and Physiological Functions of Ghrelin
Amany M Basuny, Moustafa A AboelAnin* and Eman A Hamed
Author Affiliations
Department of Agricultural Biochemistry, Faculty of Agriculture, Beni-Suef University, Egypt
Received: October 12, 2020 | Published: October 20, 2020
Corresponding author: Moustafa A AboelAnin,Department of Agricultural Biochemistry, Faculty of Agriculture, Beni-Suef
University, Egypt
DOI: 10.26717/BJSTR.2020.31.005080
The endogenous ligand for growth-hormone secretagogue receptor (GHS-R) was
discovered in 1999 from stomach and named it “ghrelin,” after a word root (“ghre”)
in Proto-Indo-European languages meaning “grow”, since ghrelin stimulates growth
hormone (GH) release from pituitary. In addition, ghrelin stimulates appetite and
increases food intake by acting on the hypothalamic arcuate nucleus, a region known to
control food intake. Thus, ghrelin plays important roles for maintaining growth hormone
release and energy homeostasis in vertebrates. The diverse functions of ghrelin raise the
possibility of its clinical application for GH deficiency, eating disorder, gastrointestinal
disease, cardiovascular disease, osteoporosis and aging, etc.
Keywords:Ghrelin; Growth Hormone;
Hypothalamic; Food Intake; Stomach
Abbreviations: GHSR: Growth Hormone Secretagogue Receptor; CaMKK: Calmodulin
Kinase; CB1: Canna BinoidReceptor Type 1;ACC: Acetylcarboxylase; FAS: Fatty Acid
Synthase; CPT1: Carnitine Palmitate Transferase;ROS: Reactive Oxygen Species; UCP2:
Unconjugated Protein 2 (UCP2); NPY: Nerve Peptide Y (NPY); AgRP:Agouti Related
Peptide ; POMC: Proopio Milano Curtin
Biochemistry of Ghrelin
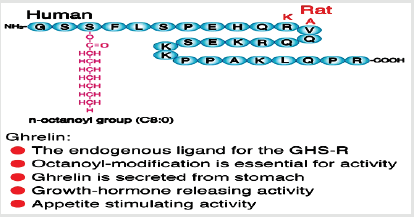
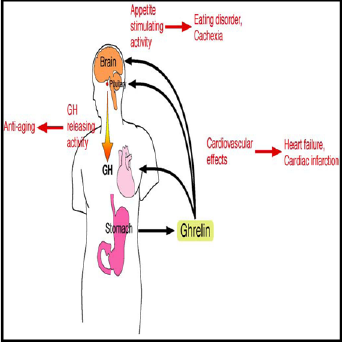
Ghrelin hormone composed of 28-amino acid peptide modified
at its third residue, a serine (Ser3), by a middle-chain fatty acid,
n-octanoicacid.The Ser3-acylation is necessary for its biological
activity, especially the binding and activation of the ghrelinhormone
receptor. Ghrelinhormonewas discovered in 1999 from stomach,
practiced potentappetite and growth hormone releasing stimulating
activities [1,2] (Figure 1).The name “ghrelin” is based on “ghre”,
which is the root ofthe word in the Indo-European primary languages
of the word “grow”, in reference to ghrelin hormone ability to induce
growth hormone release. Both the precursors of rat and human
ghrelin are consists of 117 amino acids. In these precursors, the
active ghrelin sequence of 28 amino acids immediately follows the
signal peptide. Ghrelin hormone(peptide hormone), serine 3 (Ser3)
is n-octanoylated and this modification is necessary for ghrelin
activity.The enzyme that catalyzes acyl adjustment in ghrelin has
not yet been specified. GlobalismInclusion of n-octanoic acid has
been suggested inamphibians, mammals, fish, andbirdsthat this
putative enzyme is somewhat specific in its choice of medium-chain
fatty acid substrates [3].
Mechanism of Action
Ghrelin emerged as the first circulating hunger hormone.
Ghrelin and Synthetic ghrelin imitative (the growth hormone
secretagogues) increase fat massand increase eat the food[4,5].An
action is practiced at the level of the hypothalamus.They activate
the cells in the arcuate nucleus which include the orexigenic
neuropeptide Y (NPY) neurons[6]. Ghrelin responsiveness of these
nerve cells bothinsulinsensitiveand leptin [7]. Ghrelinhormone activates the mesolimbic cholinergic dopaminergic bonus link, a
circuit that communicates the hedonic and reinforcing aspects of
natural bonus, like food as well as addictive drugs like ethanol[8].
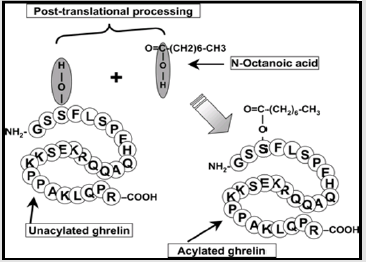
Ghrelin is Secreted in Two Forms
(Figure2)[9].
Facts of Ghrelin
Ghrelin consists of 28 amino acids that originate from 94
long precursors of amino acids (progerlin). The other products
of Progrelin are; des-Gln14-ghrelin (27 ghrelin), C-ghrelin, and
obestatin.
Effects of Ghrelin
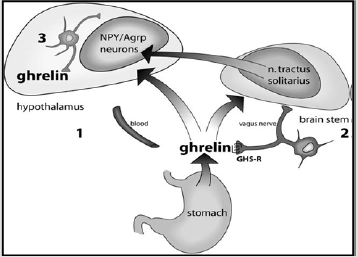
Ghrelin stimulates CRH by stimulating NPY, which inhibits
endogenous g aminobutyric acid (GABA) neurons thus releasing
the ventricular CRH nucleus from inhibiting [10,11](Figure
3).Ghrelinhormonesynthesised in the stomach reaches the ARC via the bloodstream and possibly other brain areas via an active
transport through the blood-brain barrier. Ghrelin hormone
synthesized in the periphery stimulates the vagal connections
that have been shown to express GHS-R, and the vagal connections
connect to the tractussolitarius nucleus in the brainstem which
then connects with the hypothalamus.Ghrelin is locally synthesized
in the hypothalamus and has direct links with the NPY / agoutibound
protein and other hypothalamic cells.
Ghrelin Physiological Functions
Ghrelin receptor (GHS-R), two types of GHS-R, GHS-R1a (385
amino acids) and GHS-R1b (295 amino acids).
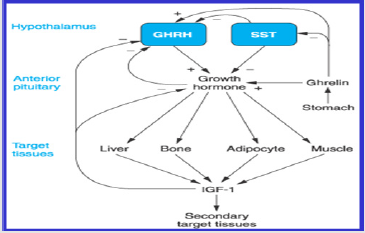
Ghrelin is a Potent Stimulator of GH Release(Figure 4).
A. Growth Hormone Secretion: Growth hormone (GH)
is secreted from the somatotroph cells of the pituitary gland.
The secretion is inhibited by Somatostatin (SS) and stimulated
by Growth Hormone Releasing Hormone (GHRH) and ghrelin.
Ghrelin stimulates the secretion of GH via binding to its
receptor GHS-R1a, which activates a G-protein Ga11, and this
activated G-protein then stimulates Phospholipase C. The
action of this lipase increases the interacellular concentration
of Inositol Triphosphate (IP3) which causes the release of Ca2+
from Interacellular stores, increase the Interacellular Ca2+ that
lead to the release of GH.Binding of Somatostatin and growth
hormone releasing hormone to their receptors (SS-R and
GHRH-R) on the cell surface lead to Inhibit (G0 and Gi) and
stimulate (Gs), stimulate (Gs) lead to stimulate adenlyatecyclase
(AC), the activation of AC increases the concentration of cyclic
AMP (cAMP), this in Turn stimulates protein kinase (PKA).
Activated PKA leading to influx of calcium ion Ca2+ into the Cell,
it leads to stimulate GH. Once ghrelin hormoneassociated GHSR-
1a, it obtains activated which in addition to activate Phospholipase C (PLC) bind to the inner parts of the receptors. Phospholipase
C contains at least eight isoforms.Phospholipase C isoforms
stimulate hydrolysis of some cell membrane phospholipids
particularlyPhosphatidyl Inositol 4, 5-diphosphate (PIP2) into
Inositol Triphosphate (IP3) and Diacylglycerol (DAG) which works
as two different messengers. Inositol triphosphate bound to the
receptor inositol triphosphate and is a Ca2+ channel with ligand
gates to theendoplasmic reticulum and catalysts for Ca2+ release in
the. Additional calcium enters from the extracellular medium via
voltageoperated L-type channels. Then the calcium ions act as a
second messengers and cause the smooth muscle to contract in the
celland causes secretory changes in the cell. Calcium ions interact
with the vesicular membrane and cause growth hormone-secreting
vesicles to fuse with the cell membrane;it is followed by exocytosis,
i.e. the extrusion of growth hormone outside the cell[12].
B. Ghrelin in Growth and Development: Ghrelin hormone
catalyzes the secretion of growth hormone in the hypothalamus,
a procedure that requires secretionGrowth Hormone
Releasing Hormone(GHRH)[13]. Diagram of the effect of
ghrelinhormoneonGrowth Hormone (GH) metabolism in adults.
Ghrelin is excreted mainly by the stomach but also from the
hypothalamus. Ghrelin regulates Growth Hormone Releasing
Hormone. Growth Hormone Releasing Hormoneexpression in
the hypothalamus in vivo. It also directly stimulates growth
hormone releasing hormonefrom the pituitary, at least in vitro.
2-Reproductive Effects (Ghrelin Effects at the Level
of the Hypothalamic-Pituitary-Gonadal Axis): Schematic
representation of ghrelin hormone effects at the level of the
hypothalamicpituitarygenital axis. Ghrelin hormoneon the principle
produced by the stomach, can act through its functional receptor
GHS-R1a in endocrine or/and local manner in all male and female
reproductive tissues including hypothalamus, pituitary, ovary,
and testis. It isknown that ovarian steroid production (oestradiol
and progesterone) can alter the secretions of the pituitary and
hypothalamus.Moreover, hypothalamusinduced GnRH controls LH,
FSH secretion known to regulate gonadal functions.In mammalian
species, ghrelin hormone treatment inhibits the release of GnRH,
LH, and FSH at the hypothalamic and pituitary levels.Adverse
effects have been described in several species of fish.In gonads,
ghrelin hormone also exerts inhibitory effects by altering steroid
composition and germ cell production or viability in the ovaries
and testicles. On the other hand, ghrelin hormone treatment
reduces proliferation of Leydig cells whereas it increases those of
granulosa cells. SCF pathway: Stem Cell Factor pathway. ↓: decrease,
↑: increase, and inhibition.
Ghrelinand Apoptosis: The schematic diagram summarized
the molecular mechanisms by which DOX-induced excessive
autophagy, apoptosis and cell size decrease in cardiomyocytes were
inhibited by ghrelin supplement. In response to DOX exposure, the
increased autophagy is paralleling with the apoptotic level, and
cell size is decreased, which are associating with the increase in
ROS content. Ghrelin attenuates the DOX induced Cardiomyocyte
apoptosis and size decrease by suppressing the excessive autophagy
level through the inhibition of ROS level and activation of mTOR
pathway, which depends on AMPK signaling inhibition and p38-
MAPK signaling activation. DOX, doxorubicin; Reactive Oxygen
Species (ROS).
Thyroid Hormone Esmodulate the Bioactivity, Secretion
and/or Metabolism of Ghrelin and Obestatin: The present study
had a limitation to elucidate the mechanism(s) of effect of TSH and
thyroid hormones on gut peptides and vice versa. At present, we
may consider that thyroid hormone demodulate the bioactivity,
secretion and/or metabolism of ghrelin hormone and obestatin.
Additionally, gut-derived metabolic hormones may be involved
in regulating the hypothalamus, pituitary and thyroid functions.
However, effect of obestatin on the thyroid axis still remains an
open question, and further examinations will be necessary.
Ghrelin Interactions in the Feeding and Sleep Circuits:
Fasting Or feeding leads to changes in leptin,glucose, and ghrelin
levels, which may affect the transition from sleep to waking.Acyl
ghrelin appears to regulate nocturnal growth hormone secretion
through a direct effect on the pituitary gland.Increased growth
hormone may be necessary for glucose balance during sleep.The
gastric or hypothalamic ghrelin hormone also activates the orexin
neurons in LHA, which in turn activates the NPY / AgRP neurons
in the ARC (ghrelin or oxygenic pathway). At the same time, this
originating stimulant produces SST secretion that inhibits the
action of GHRH, thus impeding the production of growth hormone
in the pituitary gland. The net effect is to enhance arousal and
compulsive behavior.On the other hand, Sleep disturbances lead
to elevated ghrelin with decreased levels of leptin that directly
increase the activity of the orexin system, affecting the animals’
sleep wakefulness state and their complete behavior.
Appetite: Act in the arcuate nucleus by stimulating neurons
NPY / AGRP (Y / Agouti Associated Neuropeptide) →↑ Appetite
(orexigenic effect).The clinical application of ghrelin and the diverse
functions of ghrelin increase its clinical applicability.Attempts
at clinical use of ghrelin are now underway. Ghrelin is basically
a peptide hormone that provides cells with nutrition, energy
and regulates metabolic activities.The target diseases of ghrelin
will not only be growth hormone deficiency but also nutritional
disorder and weight loss due to various reasons.Moreover, ghrelin
will be applied to the elderly to maintain an esteem of “quality of
life” through the prevention and treatment of osteoporosis and
the improvement of muscle strength through the direct action of
ghrelin and the indirect action of the growth hormone released by
ghrelin.The clinical application of ghrelin is now in its second phase
to target chronic anorexia nervosa and cachexia.In the near future,
we hope ghrelin will be used to treat these ailments (Figure 5).
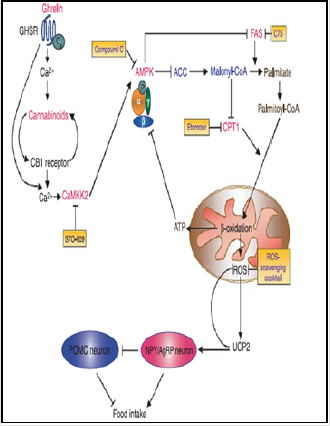
A. Mechanisms of the Appetite: Activating the particles
highlighted in red increases food intake, whereas the activation
of molecules highlighted in purple leads to inhibition of
food intake.In this figure, we depicted a simplified linear relationship between the elements that make up the ghrelin
signal chain. Clearly, the different components of this cascade
can also interact within and outside this pathway, activating
other distinct downstream signaling components, here not
reported, either sequentially or simultaneously, suggesting a
much more complex regulation.Growth Hormone Secretagogue
Receptor(GHSR); Calmodulin kinase (CaMKK); Canna Binoid
receptor type 1 (CB1); AMPK, AMP Activated Protein Kinase;
Acetylcarboxylase (ACC); Malonyl coenzyme A; Fatty Acid
Synthase (FAS); Carnitine palmitate transferase 1 (CPT1);
Reactive oxygen species (ROS); Unconjugated protein 2 (UCP2);
Nerve peptide Y (NPY);Agouti Related Peptide (AgRP); (POMC)
Proopio Milano Curtin. The effects of ghrelin hormone on
appetite are mainly mediated in the hypothalamus through
stimulation of neuropeptide Y (NPY), a potent orexigenic agent,
and of agouti related protein (AgRP), a melanocortin receptor
inverse agonist [14](Figure 6).
Diagram of ghrelin effect on energy metabolism in adults.
Ghrelin is excreted mainly by the stomach but also from the
hypothalamus. Ghrelin stimulates the appetite in the hypothalamus
by stimulating the neuropeptide Y (NPY), which is a powerful
originating factor, and agouti-binding protein (AgRP), which is a
melanocortin receptor reverse agonist. These actions are mediated
through (GHS-R). Ghrelin is also thought to induce adipogenesis
through independent GHS-R mechanisms.
B. Ghrelin and Regulate Appetite: The arcuate nucleus
(ARC) of the hypothalamus and brainstem is an important
area involved in the regulation of appetite, body weight and
energy balance [15]. The variety of hypothalamic appetite
regulators divided into two groups: The orexigenic types (appetite stimulators) which include the Neuropeptide Y
(NPY), the Agouti Related Peptide (AgRP), ghrelin, orexin and
cannabinoids, while the anorectics (appetite suppressants)
which include Proopiomelanocortin (POMC), and Cocaine
and Amphetamine Regulated Transcript (CART), Thyrotropin
Releasing Hormone (TRH), Corticotropin Releasing Hormone
(CRH), Peptide YY (PYY), Cholecystokinin (CCK) and Glucagon
Like Peptide (GLP 1) [16](Figure 7).
Ghrelin is a peptide made of 28 amino acids, synthesized mainly
by Oxidizing glands in thestomach (Hebebr and Remschmidt,
1995). Ghrelin is acylated in the third residue which is a serine,
introducing fatty acids (n-octanoyl) is essential for its activity[17].
It is one of the major signaling mechanisms the start of the meal
[18].
Ghrelin and Control of Energy Balance
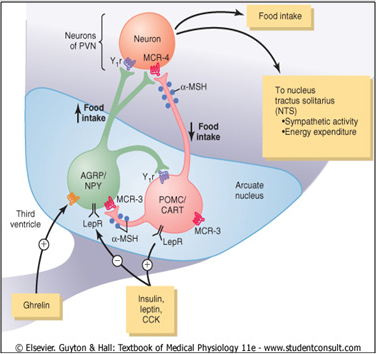
Control of energy balance through two types of neurons of
the arcuate nuclei: (1) Proopiomelanocortin (POMC) neurons that
release amelanocyte- stimulating hormone (α-MSH) and Cocaineand
Amphetamine Regulated Transcript (CART), reducing food
intake and increasing energy expenditure; and (2) neurons that
produce Agouti Related Protein (AGRP) and Neuropeptide Y (NPY),
increasing food intake and decreased energy expenditure. α MSH
released by POMC neurons stimulates melanocortin receptors
(MCR-3 and MCR-4) in the Paraventricular nuclei (PVN), Which
are then activated neuronal pathways that project to the Nucleus
Tractussolitarius (NTS) and increase sympathetic activity and
energy expenditure. AGRP act like an antagonist of MCR-4. leptin
,Insulin, and Cholecystokinin (CCK) are hormones that inhibit
AGRP-NPY neurons and stimulate adjacent POMC-CART neurons,
thereby decreased food intake. Ghrelin, a hormone secreted from
the stomach, activates AGRP-NPY neurons and motivate food
intake. leptin receptor (LepR); neuropeptide receptor (Y1R) [19].
Two groups of neurons in the arcuate nucleus are known by
the neuropeptides that they coexpress, the Neuropeptide Y (NPY)
and Agouti Related Protein (AgRP) Orexigenic Neurons as well as
the Proopiomelanocortin (POMC) and Cocaine and Amphetamine
Related Transcript (CART) Anorexigenic Neurons. These co
expressing neurons they are differentially regulated by circulating
adiposity signals, satiety signals, differentially activate second
order neurons that control food intake and energy expenditure
[19]. It stores (long term energy availability),orchestrate hormonal,
autonomic responses via differential regulation of downstream
neurons in the hypothalamus and other brain regions.
The Role of Ghrelin’s Proautophagic Properties in Cellular
Homeostasis
Ghrelin role Proautophagic Properties in Cellular Homeostasis
(A) Ghrelin enhances autophagy in a GHS-R1a-dependent
manner. Activated AMPK inhibits mTOR via activation of TSC and inactivation of Raptor. RaptorinducedInhibitory Phosphorylation
of ULK1 is decreased, leading to activation of ULK1 Kinase activity,
and activated ULK1 triggers autophagy [20].Ghrelin exerts a
cytoprotective effect by inducing autophagy in neurons, Intestinal
Epithelial Cells (IECs), and Vascular Smooth Muscle Cells (VSMCs).
Ghrelin’s proautophagic property improves hepatosteatosis by
increasing the abundance of mtDNA and inducing mitochondrial
FFA B oxidation. CaMKKb and the SIRT1 p53 axis also mediate
signaling to AMPK in the setting of autophagy, as in the case of
hypothalamic ghrelin signaling. (B) Under fasting, fat depleted
conditions, Growth hormone maintains blood sugar levels by
stimulating hepatic autophagy and subsequent gluconeogenesis.
Ghrelin is necessary to maintain growth hormone levels under
hunger, fat depleted conditions. Growth hormone signaling induces
autophagy via pSTAT. The molecule that connects the growth
hormonepSTAT axis and autophagy is currently unknown. (C)
Desacyl ghrelin stimulates AMPK activity, induces autophagy,
and reduced apoptosis andROS accumulation, thereby protecting
Cardiomyocytes from ischemic injury.
Ghrelin activates AMPK in hepatocytes, promotes autophagy,
motivate mitochondrial biogenesis, and induces mitochondrial
FFA b-oxidation, and so on ameliorates hepatic triglyceride over
accumulation[21].(A)ghrelin attenuates hepatic lipotoxicity by
enhancing autophagy via restoration of the AMPK/mTOR signaling
pathway [22]. The ghrelin autophagy axis is essential for survival
in famine. Under fasting, fat depleted conditions, organismsactivate
hepatic autophagy to perform gluconeogenesis andmaintain blood
glucose levels. This process is mainly orchestrated by the action
of GH [23].Under hunger,fat depleted conditions, GOAT knockout
mice exhibit insufficientGHup regulation, a decline in hepatic
autophagy, and lethal hypoglycemia[24] (B).A comprehensive
screen based on in vivo delivery of arrayed cDNA libraries aimed
at identifying tissue protective factors revealedStrong and specific
expression of the ghrelin gene in cardiac and skeletal muscles after
acute ischemia[25]. Transduction of the ghrelin gene into the heart
rescues Cardiomyocytes from ROS accumulation and apoptosis,
Restores heart function after myocardial infarction in an autophagy
manner (C).Desacyl ghrelin also reduce ROS production, lowers
tissue inflammation and reinforces insulin stimulated glucose
uptake in skeletal muscle in an autophagy dependent manner[26].
4.6.9. Ghrelin is Anti-Inflammatory[27-30]: It has been
shown that ghrelin is able to exert anti-inflammatory actions
by inhibiting the production of inflammatory cytokines.Ghrelin
practice anti-inflammatory actions in inflammatory bowel disease,
sepsis,pancreatitis, arthritis, and diabetic nephropathy[31-39].
Administration of ghrelin before the development of experimental
pancreatitis improved pancreatic blood flow, Lower IL1𝛽levels, and stimulated pancreatic cell proliferation[33]. In sepsis, ghrelin,
via an upregulation of MAPK phosphatase 1, lowerNorepinephrine and TNF𝛼levels known to cause hepatocellular dysfunction and upregulation of Proinflammatory Cytokines[40]. Furthermore,
organ blood flow is improved by ghrelin via an inhibition of NFkB(
Wu, et al.) and HMGB1 production by activated macrophages is
inhibited by ghrelin [32].
Ghrelin decreased IL6 levels and symptoms of arthritis in
an animal model [31-55]. IL8 and IL6levels induced by insoluble
fibrillary𝛽amyloid protein deposition in mouse microglia are lower by desacyl ghrelin but not by acyl ghrelin probably by
amechanisminvolving, as already eluded to, an unidentified
receptor distinct from GHS-R1A [41].Anti-inflammatory and
antihyperalgesic effects of both desacyl ghrelin and acyl ghrelin
have been shown in rats [38]. The development of experimental
diabetic nephropathy in mice can be prevented by acyl ghrelin
acting on GHS-R1A[39]. Inflammatory bowel disease, especially
Crohn’s disease, is improved by administering ghrelin [36,41-55].
Ghrelin is a peptide hormone that the stomach secretes
primarily into the bloodstream, but other tissues have been shown
to also synthesize it. Ghrelin can exert its effects through systemic
or atomicin/ paracrine actions. The GHS-R1A receptor binds to acyl
ghrelin and presumably mediates its biological effects. None the
less, it is I realized that either GHSR1A homo- or heterodimmers
could be participate in the ghrelinmediated actions. The formation
of homo and heterodimmers is adding another level of complexity
in the understanding of the actions of ghrelinGrowing sets of
evidence support an increasing number of functions for desacyl
ghrelin. So far, the exact mechanisms and a potential specific
receptor have eluded determination. Much work remains to be
done to determine if this additional level of complexity is indeed
accounting for the biological effects of ghrelin.Varied physiological
andnumerouseffectsof ghrelin, It has also been reviewed in this
paper, have been reported. And so on, it appears important to
perform further studies to better understand the fine underlying
mechanisms accounting for these pleiotropic ghrelin actions.
Current understanding of ghrelin biology and biological functions
has led to the development of pharmacological tools modulating
ghrelin actions and the evaluation of their clinical applications.
The authors report no conflicts of interest in this work.
- Kangawa K (1999) Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 402: 656-660.
- Kola B, Hubina E, Tucci SA, Kirkham TC, Garcia EA, et al. (2005) Cannabinoids and ghrelin have both central and peripheral metabolic and cardiac effects via AMP-activated protein kinase. Journal of Biological Chemistry 280: 25196-25201.
- Nishi Y, Hiejima H, Hosoda H, Kaiya H, Mori K, et al.(2005): Ingested medium-chain fatty acids are directly utilizedfor the acyl modification of ghrelin. Endocrinology 146: 2255-2264.
- Tschöp M, Smiley DL, Heiman ML (2000) Ghrelin induces adiposity in rodents. Nature 407(6806): 908-913.
- Lall S, Tung LY, Ohlsson C, Jansson JO, Dickson SL (2001) Growth hormone (GH)-independent stimulation of adiposity by GH secretagogues. Biochem.Biophys. Res Commun 280(1): 132-138.
- Dickson SL, Luckman SM (1997) Induction of cfos messenger ribonucleic acid in neuropeptide Y and growth hormone (GH)-releasing factor neurons in the rat arcuate nucleus following systemic injection of the GH secretagogue, GH-releasing peptide-6. Endocrinolog 138(2): 771-777.
- Hewson AK, Tung LY, Connell DW, Tookman L, Dickson SL (2002) The rat arcuate nucleus integrates peripheral signals provided by leptin, insulin,and a ghrelin mimetic. Diabetes 51(12): 3412-3419.
- Jerlhag E, Egecioglu E, Dickson SL, Douhan A, Svensson L, et al. (2007) Ghrelin administration into tegmental areas stimulates locomotors activity and increases extracellular concentration of dopamine in the nucleus accumbens. Addiction Biology 12(1): 6-16.
- Sato T, Nakamura Y, Shiimura Y, Ohgusu H, Kangawa K, et al.(2012).Structure, regulation and function of ghrelin. J Biochem 151(2): 119-128.
- Cone RD, Cowley MA, Butler AA, Fan W, Marks DL, et al. (2001) The arcuate nucleus as a conduit for diverse signals relevant to energy homeostasis. International Journal of Obesity Related Metabolic Disorders 25(5): 63-67.
- Cowley MA, Smith RG, Diano S, Tschop M, Pronchuk N, et al. (2003) The distribution and mechanism of action of ghrelin in the CNS demonstrates a novel hypothalamic circuit regulating energy homeostasis. Neuron 37: 649-661.
- Masayasu K,Kojima KK (2005) Ghrelin: Structure and Function. Physiol Rev 85(2): 495-522.
- Tannenbaum GS, Epelbaum J, Bowers CY (2003) Interrelationship between the novel peptide ghrelin and somatostatin/growth hormone-releasing hormone in regulation of pulsatile growth hormone secretion. Endocrinology 144(2): 967-974.
- Chen HY, Trumbauer ME, Chen AS, Weingarth DT, Adams JR et al. (2004) Orexigenic action of peripheral ghrelin is mediated by neuropeptide Y and agouti-related protein. Endocrinology 145(4): 2607-2612.
- Druce M,Bloom SR (2006) The regulation of appetite. Arch Dis Child 91: 183-187.
- Scerif M, Goldstone AP, Korbonits M (2011) Ghrelin in obesity and endocrine diseases. Mol Cell Endocrinol 340(1): 15-25.
- Yang J, Brown MS, Liang G, Grishin NV, Goldstein JL (2008) Identification of the acyl-transferase that octanoylates ghrelin, an appetite-stimulating peptide hormone. Cell 132(3): 387-396.
- Cummings DE, Frayo RS, Marmonier C, Aubert R, Chapelot D (2004) Plasma ghrelin levels and hunger scores in humans initiating meals voluntarily without time- and food-related cues. Am J Physiol Endocrinol Metab 287(2): 297-304.
- Barsh GS, Schwartz MW (2002) Genetic approaches to studying energy balance: perception and integration. Nature 3(8): 589-600.
- Kim J, Kundu M, Viollet B,Guan KL (2011) AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat. Cell Biol 13(2): 132-141.
- Ezquerro S, Me´ ndez Gime´ nez L, Becerril S, Moncada R, Valentı´ V, et al. (2016) Acylated and desacyl ghrelin are associated with hepatic lipogenesis, b-oxidation and autophagy: role in NAFLD amelioration after sleeve gastrectomy in obese rats. Sci Rep 6: 39942.
- Mao Y, Cheng J, Yu F, Li H, Guo C, et al. (2015) Ghrelin attenuated lipotoxicity via autophagy induction and nuclear factor-kB inhibition. Cell. Physiol Biochem 37: 563-576.
- Ezaki J, Matsumoto N, Takeda-Ezaki M, Komatsu M, Takahashi K, et al. (2011) Liver autophagy contributes to the maintenance of blood glucose and amino acid levels. Autophagy 7(7): 727-736.
- Zhang Y, Fang F, Goldstein JL, Brown MS, Zhao TJ (2015) Reduced autophagy in livers of fasted, fat-depleted, ghrelin-deficient mice: reversal by growth hormone. Proc Natl Acad Sci 112(4): 1226-1231.
- Ruozi G, Bortolotti F, Falcione A, Dal Ferro, M Ukovich, et al. (2015) AAV-mediated in vivo functional selection of tissue-protective factors against ischaemia. Nat Commun 6: 7388.
- Gortan Cappellari G, Zanetti M, Semolic A, Vinci P, et al. (2016) Unacylated ghrelin reduces skeletal muscle reactive oxygen species generation and inflammation and prevents high-fat diet-induced hyperglycemia and whole body insulin resistance in rodents. Diabetes 65(4): 874-886.
- Chang L, Du JB, Gao LR, Pang YZ, Tang CS (2003) Effectof ghrelin on septic shock in rats. Acta Pharmacologica Sinica 24(1): 45-49.
- Dembinski A, Warzecha Z, Ceranowicz P, Tomaszewska R, Stachura J, et al. (2003) Ghrelin attenuates the development of acute pancreatitis in rats. Journal of Physiology and Pharmacology 54(4): 561-573.
- Xia Q, Pang W, Pan H, Zheng Y, Kang JS, et al. (2004) Effects of ghrelin on the proliferation and secretion of splenic lymphocytes in mice, Regulatory Peptides 122(3): 173-178.
- Dixit VD, Schaffer EM, Pyle RS, Collins GD, Sakthivel SK, et al. (2004) Ghrelininhibitsleptin- and activation-induced pro-inflammatorycytokine expression by human monocytes and T cells. The Journal of Clinical Investigation 114(1): 57-66.
- Granado M Priego T Mart´ın, A I Villan´ua MA, L´opez-Calder´on A (2005) Anti-inflammatory effect of the ghrelin agonist growth hormone-releasing peptide-2 (GHRP-2) in arthritic rats. The American Journal of Physiology-Endocrinology and Metabolism 288(1): 486-492.
- Chorny A, Anderson P, Gonzalez Rey E, Delgado M (2008): Ghrelin protects against experimental sepsis by inhibiting high-mobility group box 1 release and by killing bacteria. Journal of Immunology 180(12): 8369-8377.
- Warzecha Z, Ceranowicz P, Dembinski A,Cieszkowski J, Kusnierz-Cabala B, et al. (2010) Therapeutic effect of ghrelin in the course of cerulein induced acute pancreatitis in rats. Journal of Physiology and Pharmacology 61(4): 419-427.
- Baatar D, Patel K, Taub DD (2011) The effects of ghrelin on inflammation and the immune system. Molecular and Cellular Endocrinology 340(1): 44-58.
- Das UN (2011) Relationship between gut and sepsis: role of ghrelin. The World Journal of Diabetes 2(1): 1-7.
- Deboer MD (2011) Use of ghrelin as a treatment for inflammatory bowel disease: mechanistic considerations. International Journal of Peptides 189242: 8.
- Cheyuo C, Jacob A, Wang P (2012) Ghrelin-mediated sympathy inhibition and suppression of inflammation in sepsis. American Journal of Physiology-Endocrinology and Metabolism 302(3): 265-272.
- Sibilia V, Pagani F, Mrak E, Dieci E, Tulipano G, et al. (2012) Pharmacological characterization of the ghrelin receptor mediating its inhibitory action on inflammatory painin rats. Amino Acids 43(4): 1751-1759.
- Tsuchimochi W, Kyoraku I, Yamaguchi H, Toshinai K, Shiomi K, et al. (2013) Ghrelin prevents the development of experimental diabetic neuropathy in rodents. The European Journal of Pharmacology 702(1): 187-193.
- Jacob A, Rajan D, Pathickal B, Balouch I, Hartman A, et al. (2010) The inhibitory effect of ghrelin on sepsis-induced inflammation is mediated by the MAPK phosphatase-1. International Journal of Molecular Medicine 25(1): 159-164.
- Bulgarelli I, Tamiazzo L, Bresciani E,Rapetti D, Caporali S, et al. (2009) Desacyl-ghrelinand synthetic GH-secretagogues modulate the production of inflammatory cytokines in mouse microglia cells stimulated by𝛽-amyloid fibrils. Journal of Neuroscience Research 87(12): 2718-2727.
- Bennett PA, Thomas GB, Howard AD, Feighner S, Van der Ploeg LH, et al. (1997) Hypothalamic growth hormone esecretagogue-receptor (GHS-R) expression is regulated by growth hormone in the rat. Endocrinology 138(11): 4552-4557.
- Cummings DE, Purnell JQ, Frayo RS, Schmidova K, Wisse BE, et al. (2001) A preprandial rise in plasma ghrelin levels suggests a role in meal initiation in humans. Diabetes 50(8): 1714-1719.
- Egecioglu E, Stenstrom B, Pinnock SB, Tung LY, Dornonville de la CC, et al. (2008) Hypothalamic gene expression following ghrelin therapy to gastrectomizedrodents. Regulatory Peptides 146: 176-182.
- Guan XM, Yu H, Palyha OC, McKee KK, Feighner SD, et al. (1997) Distribution ofmRNA encoding the growth hormone secretagogue receptor in brain andperipheral tissues. Molecular Brain Research 48(1): 23-29.
- Higgins SC, Gueorguiev M, Korbonits M (2007) Ghrelin, the peripheral hunger hormone. Annals of Medicine 39(2): 116-136.
- Kojima M, Kangawa K (2005) Ghrelin: structure and function. Physiol Rev 85(3): 495-522.
- Kojima M, Hosoda H, Date Y, Nakazato M, Matsuo H, et al. (1999) Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 402(6762): 656-660.
- Neary NM, Druce MR, Small CJ, Bloom SR (2006) Acylatedghrelin stimulates food intake in the fed and fasted states but desacylated ghrelin hasno effect. Gut 55(1): 135.
- Seoane LM, Lopez M, Tovar S, Casanueva FF, Senaris R, et al. (2003) Agouti-related peptide, neuropeptide Y, and somatostatin-producingneurons are targets for ghrelin actions in the rat hypothalamus. Endocrinology 144(2): 544-551.
- Shiiya T, Nakazato M, Mizuta M, Date Y, Mondal MS, et al. (2002) Plasma ghrelin levels in lean and obese humans and the effect of glucose on ghrelin secretion. Journal ofClinical Endocrinology and Metabolism 87(1): 240-244.
- Smith RG, Jiang H, Sun Y (2005) Developments in ghrelin biology andpotential clinical relevance. Trends in Endocrinology and Metabolism 16(9): 436-442.
- Theander Carrillo C, Wiedmer P, Cettour Rose P, Nogueiras R, Perez Tilve D, et al. (2006) Ghrelin action in the brain controls adipocyte metabolism. Journal of ClinicalInvestigation 116(7): 1983-1993.
- Wu R, Zhou M, Das P, Dong W, Ji Y, et al. (2007) Ghrelin inhibits sympathetic nervous activity in sepsis. American Journal of Physiology-Endocrinology and Metabolism 293(6): 1697-1702.
- Zigman JM, Nakano Y, Coppari R, Balthasar N, Marcus JN, et al. (2005) Mice lacking ghrelin receptors resist the development of diet-induced obesity. Journal of Clinical Investigation 115(12): 3564-3572.

 Research Article
Research Article