Abstract
Blended edible oils were formulated to enrich fatty acid profile with balance fatty acid ratio. Indigenous Rice Bran Oil (RBO) and Mustard Seed Oil (MO) were mixed in the ratio of 60:40, 70:30 and 90:10 (RBO:MO). Moisture content, density, specific gravity, refractive index, viscosity, free fatty acid, acid value, iodine value, unsaponifiable matter and peroxide value of the single vegetable oils and their blends were determined. Fatty acid composition such as Saturated Fatty Acid (SFA), Mono-Unsaturated Fatty Acid (MUFA), Poly-Unsaturated Fatty Acid (PUFA) of all oils were analyzed by GCMS. GCMS analysis of rice bran oil identified 6 fatty acids with ratio SFA:MUFA:PUFA=1.43:1.46:1.0 and mustard seed oil identified 8 fatty acids with ratio SFA:MUFA:PUFA=1.72:1:2.06. All blended oils revealed 9 fatty acids with ratio SFA:MUFA:PUFA=1.24:2.1:1.0;SFA:MUFA:PUFA= 1.34:2.56:1.0andSFA:MUFA:PUFA=1.2:1.6:1.0; for 60:40, 70:30 and 90:10 (RBO: MO) respectively. Among three blendings, SFA:MUFA:PUFA=1.2:1.6:1.0 for 90:10 (RBO: MO) ratio was near World Health Organization (WHO) guidelines. According to WHO and other health agencies, nutritionally superior oil must fall in the ratio of 1:1-3:1; SFA; MUFA and PUFA respectively. The results suggest that 90:10 (RBO:MO) blended oil can be good alternative oil for edible purposes with improve nutrition as well as desirable physico-chemical properties.
Keywords: Blended Oil; Rice Bran Oil; Mustard Oil; Fatty Acids; Saturation and Unsaturation
Abbreviations: RBO: Rice Bran Oil; MSO: Mustard Seed Oil; SFA: Saturated Fatty Acid; MUFA: Mono-Unsaturated Fatty Acid; PUFA: Poly-Unsaturated Fatty Acid; LDL-C: Low Density Lipoprotein Cholesterol; FFA: Free Fatty Acid; AV: Acid Value
Introduction
Vegetable oil plays a variety of in human nutrition due to its
different fatty acids. But high intakes of Saturated Fatty Acids (SFA)
increase Low Density Lipoprotein Cholesterol (LDL-C) as well as
cardiovascular diseases. On the other hand, unsaturated fatty acids
have the opposite effects. To combat this fatty acid ratio problem,
blending of vegetable oils is gaining popularity in the oil sector to
satisfy consumer demands. Rice Bran Oil (RBO), a by-product of
the rice milling industry contains unique antioxidant compound
gamma-oryzanol, high levels of phytosterols and tocols [1,2]. Due
to the presence of micronutrients in RBO, the value of other edible
vegetable oils can be nutritionally improved by mixing with RBO.
Choudhary, et al. [3] stated that blending of non-conventional RBO
with traditional oils (sunflower oil, canola oil etc.) can result in a
stable cooking oil that reduces the demand for traditional oils. On
the other hand, mustard oil which is rich in unsaturated fatty acids
widely used for both culinary and therapeutic purposes. Oil with
higher unsaturation are highly prone to oxidation [4]. Basically
fatty acid composition of the oils is the main factor responsible for oxidation and also for the formation of undesirable compounds
or flavours [5,6]. However, there is global increasing demand for
the production of a blending oil for edible purposes because of
improved fatty acids and nutritional benefits.
Blending is one of the methods used to modify oils and fats for
wide range of applications. Evidences suggest that no single oil
can provide the recommended dietary fatty acid ratio. According
to WHO and other health agencies, nutritionally superior oil must
fall in the ratio of 1:1-3:1; SFA, MUFA and PUFA respectively [7].
The objective of the present investigation was to design an oil
blend which may provide an ideal recommended fatty acid ratio
regarding nutrition.
Materials and Methods
Materials
Mustard seeds were procured from local market then fresh and healthy mustard seeds were stored for oil extraction. Freshly milled rice bran, outer layer of the grain was collected from rice milling industry, Rajshahi, Bangladesh and immediately processed after milling for stabilization. Primarily sieved with 100 mesh size to separate husk and broken rice grain. All solvents and chemicals used were analytical, HPLC and GC grade. Fatty acids standards were purchased from Sigma-Aldrich.
Extraction of Oils
Rice bran oil was extracted by solvent extraction method. The extraction process was conducted in the laboratory by Soxhlet apparatus for 6 hours using n-hexane as extracted solvent. The ratio of rice bran:hexane used was 1:3 (w/v). n-hexane has been selected because the solvent has better effect over other polar solvents like alcohol, ketone, aldehyde ether and ester etc. [8]. The oil was recovered by the evaporation of the solvent under reduced pressure using a Rotary vacuum evaporator and the percentage of oil content was calculated. Mustard oil was extracted by automated oil expelling machine. The refining process of the oils was carried out using the method as described by [9] with some modification.
Preparation of Blends
Rice bran oil and mustard seed oil were taken in the ratio of 90:10, 70:30 and 60:40 respectively. Each blend was placed in 250 ml beakers and were mixed by using a mechanical stirrer at 180 rpm for 15 min. Then the single oils and all blended oils were stored at room temperature in air tight glass bottle for physico-chemical and GCMS analyses.
Analysis of Physicochemical Properties
Physical properties such as moisture content, refractive index, density, specific gravity and viscosity of the single and blended oils were determined by the method described by Garba, et al. [10] and AOAC [11]. Acid value, saponification value, iodine value, peroxide value, unsaponifiable matter and free fatty acid were estimated according to the AOAC and AOCS Official Methods [12,13]. Viscosity of all oils were determined by Fungilab digital Rotational viscometer. They measure viscosity by sensing the torque required to rotate a spindle at constant speed while immersed in fluid.
Determination of Fatty Acid Composition
Preparation of Fatty Acid Methyl Ester: 200 mg of oil sample was taken in a 10 mL Pyrex test tube. 3.5 mL 0.5M Sodium methoxide was added to the test tube and heated using burner until removing the bubbles. Then, 1.5 mL n-hexane was added to the mixture and mixing by vortex mixture. Then around 5 mL deionized water was added to test tube slowly and waited for settle down. Organic upper layer was taken into the GC vial for GCMS analysis.
GCMS Condition: The gas chromatographic analyses of the oils were performed on SHIMADZU GC-2010. GC was equipped with auto-sampler (AOC- 20s), auto-injector (AOC-20i) and SH Rxi 5MS Sill capillary column with 30m×0.25mm×0.25 μm film. Helium was used as carrier gas at flow rate 2.0 mL/min. Initial oven temperature of GC was 1400C for 10.0 min, then increased at 70C/min to a final temperature of 250 0C for 10.0 min. The injector temperature was 250 0C and injection volume was 1.0 μL with 75:1 split ratio. Solvent cut time was 3.40 min and total run time was 35.71 min. The detector used was SHIMADZU GCMS-QP-2020 and temperature was 2550C.
Results and Discussion
Moisture is the amount of water present in edible oils. Moisture
should be as low as possible. The maximum allowed moisture
content in edible oils is 0.2% [14]. Previous studies have shown
that fungus species such as Aspergillus niger and Mucor species
survive and reproduce when the moisture content value is higher
than 0.2% / [15]. Moisture content of all oils and their blends were
determined presented in Table 1. All values were very low than
specified limit. Density, specific gravity and refractive index of all
single oils and their blends were determined presented in Table 1.
Results were within the WHO/FAO permissible limits. Viscosity of
oil is a measure of the oils resistance to flow. In the present study,
it can be observed from Table 1 that viscosity decreased in the
blended oils than single vegetable oils, this is because blending
oil with different ratios decreases saturation as well as increases
unsaturation fatty acids of oil. This phenomenon also verified by
other researcher since oil viscosity depends on molecular structure
and decreases with unsaturation of fatty acids [7]. It may due to
double bonds that make bonding more rigid and rotation between
c-c bonds become more strenuous.
The Free Fatty Acid (FFA) and Acid Value (AV) are most
important quality parameters of edible oil. The results showed
in Table 1 that the FFA value of all oils were very low and the
percentages of AV for all oils were within the permissible level (0.6
mg KOH/g) [16]. All oils were of good quality because of the low percentage of FFA and acid value. Theoretically, AV is known to be
high when FFA content is high. FFA content differs from AV, which
can be due to the fact that acid phosphates as well as amino acids
contribute to the acid level [17]. Mean peroxide value of the oil
samples are shown in Table 1. Highest level of peroxide value was
observed for mustard oil (2.84 mEq/kg oil) and lowest for RBO:MO,
90:10 blended oil (1.44 mEq/kg oil). But levels of peroxide value
obtained for the all oil samples were below the acceptable limit
(10 mEq/kg oil) recommended by Codex Standards [18]. The low
values of peroxide are indication of low levels of oxidative rancidity
of the oils. A rancid taste often begins to be noticeable when the
peroxide value is above 20 meq/kg [19]. The iodine value is a
measurement of the total unsaturation of vegetable oils, as well as
an indicator of their susceptibility to oxidation [19]. In the present
study shown in Table 1, the blends of mustard oil with rice bran oil
shows increase in iodine value. Hence, the observed higher iodine
value in the oils indicated that they are likely to be healthier for
consumption. Studies have recommended to switch from saturated
to unsaturated fats because of the risk of cardiovascular disease
associated with high consumption of saturated fatty acids [20-23].
The unsaponifiable matters for all oils were within 0.35-0.62 %
presented in Table 1 and the values were in good agreement with
the reported result of Tan, et al. [24].
Table 1: Physicochemical properties of Rice ban oil, Mustard oil and their blends.
Note: All the experiments were performed in triplicate and the results were expressed as mean ± SD (standard deviation).
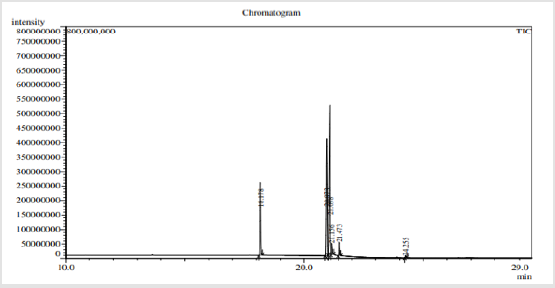
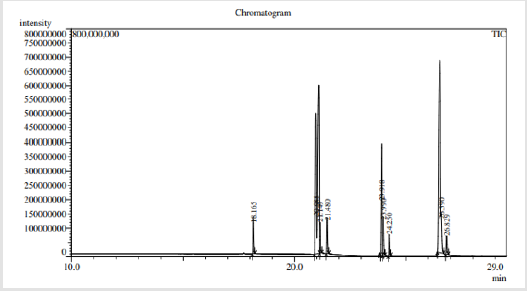
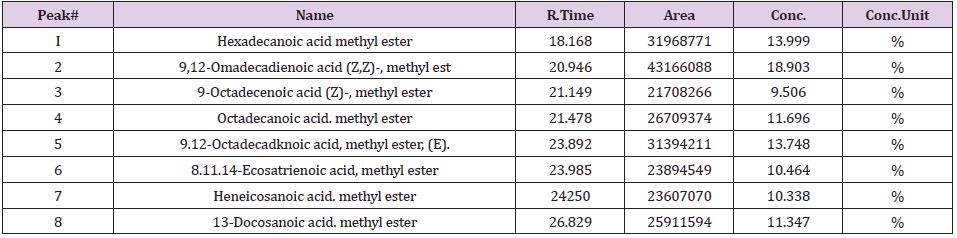
The results of GCMS analysis of rice bran oil are displayed in Figure 1 and presented in Table 2. They revealed the presence of 6 fatty acids were classified as Saturated Fatty Acids (SFA), Monounsaturated Fatty Acids (MUFA) and Polyunsaturated Fatty Acids (PUFA) in percentages 36.704, 37.624 and 25.671 respectively. 9-octadecenoic acid methyl ester as oleic acid was the major identified monounsaturated fatty acid (37.624%). Fatty acid ratio was SFA:MUFA:PUFA=1.43:1.46:1.0 shown in Table 3. GCMS analysis of mustard seed oil are shown in Figure 2 and presented in Table 4. They identified 8 fatty acids where saturated fatty acids 34.033%, monounsaturated fatty acids 20.853% and polyunsaturated fatty acids 43.115%. Fatty acid ratio was SFA:MUFA:PUFA = 1.72:1:2.06 presented in Table 3. GCMS analysis of blended oil mixing ratio of RBO:MO;90:10 are displayed in Figure 3 and presented in Table 5. They revealed the presence of 9 fatty acids. Saturated, monounsaturated and polyunsaturated fatty acids were 31.569%, 42.297% and 26.134% respectively. Fatty acid ratio was SFA:MUFA:PUFA = 1.2:1.6:1.0. Also Fatty acid ratio of blended oil in the mixing ratio of RBO:MO;60:40 and RBO:MO;70:30 were determined and presented in Table 3. SFA:MUFA:PUFA ratio was 1.24:2.1:1.0 for RBO:MO;60:40 blend and SFA:MUFA:PUFA ratio was 1.34:2.56:1.0 for RBO:MO;70:30 blend. Among three blending, SFA:MUFA:PUFA = 1.2:1.6:1.0 ratio for RBO:MO;90:10 blend was near World Health Organization guidelines. Results depicted that single rice bran and mustard seed oils have a higher unsaturated fatty-acids than saturated fatty acid. However, highly unsaturated oils goes oxidative degradation due to the presence of their double bonds [25] unless sufficient antioxidant is added [26-28]. Also high consumption of saturated fatty-acids associated with the risk of cardiovascular diseases [20-23]. So, mixing vegetable oil can be a cost-effective practice to modify their fatty acid profile and physico-chemical properties [7]. Present work can meet the desired requirements.
Conclusion
Blending mustard oil with rice bran oil in the ratio of RBO: MO, 90:10 can be encouraged to enrich fatty acid composition as well as balance fatty acid ratio of the oil, because MO is rich in PUFA, while RBO is rich in SFA and MUFA with addition to bioactive phytochemicals. This study will help the oil producing industry to find the most economically viable oil blends for cooking purposes with the maximum fatty acids, nutrition and desirable physicochemical properties.
References
- Das AB, Goud VV, Das C (2018) Extraction and characterization of phenolic content from purple and black rice (Oryza sativa L.) bran and its antioxidant activity. Food Measur 12: 332-345.
- Perez Ternero C, Sotomayor MA, Herrera MD (2017) Contribution of ferulic acid, g-oryzanol and tocotrienols to the cardiometabolic protective effects of rice bran. J Funct Food 32: 58-71.
- Choudhary M, Grover K, Kaur G (2015) Development of rice bran oil blends for quality improvement. Food Chem 173: 770-777.
- Ivanov DS, Lević JD, Sredanović SA (2010) Fatty acid composition of various soybean products. Food Feed Res 37: 65-70.
- Arslan FN, Kara H, Talpur MY, Sherazi STH (2015) A chemometric approach to assess the frying stability of cottonseed oil blends during deep-frying process: fatty acid composition and tocopherol analyses. Int J Food Prop 18: 2776-2790.
- Fernández Cedi LN, Enríquez Fernández BE, Yañez LÁC, Sosa Morales ME (2012) Performance of palm olein and soybean oil during the frying of French fries and its effect on the characteristics of the fried product. J Culin Sci Technol 10: 211-222.
- Hashempour Baltork F, Torbati M, Azadmard S Damirchi, Geoffrey P Savage (2016) Vegetable oil blending: A review of physicochemical, nutritional and health effects. Trends in Food Science & Technology 57: 52-58.
- Ali MH, Rahman MS, Ahmed GM, Hossain MA, Uddin MA (1996) The effect of packing materials on the stability of ground nut oil during storage J Sc Ind Res 31(3): 59-67.
- Pestana VR, Zambiazi, RC, Mendonça CRB, Bruscatto MH, Lerma Garcia MJ, et al. (2008) Quality changes and tocopherols and γ-oryzanol concentrations in RBO during the refining process. J Am Oil Chem Soc 85: 1013-1019.
- Garba AA, Medugu DW, Gwaski PA, Amusat RO (2015) Extraction and characterization of moringa oleifera seed oil. Applied Research Journal 1(9): 473-477.
- (2005) AOAC, Official methods of analysis (16th)., Association of Official Analytical Chemists, Washington, DC, USA.
- (1999) Gaithersburg AOAC International Official Methods of Analysis of AOAC International, (16th ).
- Mehlenbacher VC, Hopper TH, Sallee EM, Walker RO, Walker RC, et al. (2009) Official methods and recommended practices of the American Oil Chemists Society (6th)., American Oil Chemists Society, Champaign.
- Federation AO (2011) Section 1: Quality Standards, Technical Information & Typical Analysis. Australian Oilseeds Federation, Australia Square, p. 40-45.
- Commission CA (2001) Codex standard for named vegetable oils codex stan 210-1999. Report of the 17 the session of the codex committee on fats and oils, London, p. 19-23.
- Alimentarius C Codex standard for named vegetable oils.
- Wrostald RE, Decker EA, Schwartz SJ, Sporns P (2005) Handbook of food analytical chemistry: water, proteins, enzymes, lipids and carbohydrates. Wiley and Sons Inc, New Jersey, USA.
- Codex Standards (2015) Codex Standards for Edible Fats and Oils Not Covered by Individual Standards. Codex Stan 19-1981 (Amendment 2015).
- Pandurangan MK, Murugesan S, Gajivaradhan P (2014) Physico-chemical properties of groundnut oil and their blends with other vegetable oils. J Chem Pharm Res 6(8): 60-66.
- De Souza RJ, Mente A, Maroleanu A, Cozma AI, Kishibe T (2015) Intake of saturated and trans unsaturated fatty acids and risk of all cause mortality, cardiovascular disease and type 2 diabetes: systematic review and meta-analysis of observational studies. BMJ 351: h3978.
- Li Y, Hruby A, Bernstein AM, Ley SH, Wang DD, et al. (2015) Saturated fats compared with unsaturated fats and sources of carbohydrates in relation to risk of coronary heart disease: a prospective cohort study. J Am Coll Cardiol 66(14): 1538-1548.
- German JB, Dillard CJ (2004) Saturated fats: what dietary intake? Am J Clin Nutr 80(3): 550-559.
- Nettleton JA (1995) Omega-3 fatty acids and health. Omega-3 fatty acids and health. Springer, Berlin, Germany, p. 64-76.
- Tan HC, Ghazali MH, Kuntom A, Tan PC, Ariffin AA (2009) Extraction and physicochemical properties of low free fatty acid crude palm oil. Food Chemistry 113: 645-650.
- Adetola O, Alabi O, Abdulrauf I (2016) Investigating storage duration and packaging materials on quality of fresh palm oil. FUTA Journal of Research in Sciences 12(2): 252-259.
- Xiu Qin L, Chao J, Yan Yan S, Min Li Y, Xiao Gang C (2009) Analysis of synthetic antioxidants and preservatives in edible vegetable oil by HPLC/TOF-MS. Food Chem 113(2): 692-700.
- Cheung SCM, Szeto YT, Benzie IF (2007) Antioxidant protection of edible oils. Plant Foods Hum Nutr 62(1): 39-42.
- Dziedzic SZ, Hudson BJ (1983) Polyhydroxy chalcones and flavanones as antioxidants for edible oils. Food Chem 12(3): 205-212.

 Research Article
Research Article