Abstract
Wild medicinal plants are an efficient source of anticancer agents. The widely distributed Launaea fragilis and Launaea nudicaulis in Egypt were investigated for their anti-proliferative activities against some cancer cell lines. Ethanol extracts of the two species were evaluated in vitro by Sulforhodamine B (SRB) assay on six cell lines; hepatocellular carcinoma (Hep G2), prostatic carcinoma (PC-3), colorectal carcinoma (HCT116), non-small cell lung carcinoma (H1299), breast adenocarcinoma (MCF7) and colon adenocarcinoma (Caco-2). The results showed that the incubation of both L. fragilis and L. nudicaulis with non-small cell lung carcinoma (H1299) inhibited significantly the cell proliferation with IC50=26.5 and 20 μg/ml, respectively. Launaea fragilis recorded the lowest inhibition values for prostatic carcinoma (PC-3) with IC50=40μg/ml. However, breast adenocarcinoma (MCF7) revealed the lowest inhibition value for L. nudicaulis with IC50=35μg/ml. Ethanol extract of L. nudicaulis was more effective in the inhibition of the cell proliferation than that of L. fragilis for the six cell lines.
Keywords: Anti-Proliferation; Breast; Colon; Colorectal; Hepatocellular; Launaea fragilis; Launaea nudicaulis; Non-Small Cell Lung; Prostatic
Abbreviations: Caco-2: Colon Adenocarcinoma Cell Line; H1299: Non-Small Cell Lung Carcinoma Cell Line; HCT116: Colorectal Carcinoma Cell Line; Hep G2: Hepatocellular Carcinoma Cell Line; MCF7: Breast Adenocarcinoma Cell Line; PC-3: Prostatic Carcinoma Cell Line; SRB: Sulforhodamine B
Background
Cancer is a rapid division of abnormal cells which grow beyond their normal limits. Similarly, it can attack adjacent parts of the body and spread to other organs. Lung and breast are the most common cancers (2.09 million cases) followed by colorectal, prostate, skin and stomach in a decreasing order. According to the World Health Organization [1], cancer is the second prominent reason of death globally and it is responsible for an estimated 9.6 million deaths in 2018 and approximately 70% of mortality occurs in the poor developing countries. Chemotherapeutic agents of cancer possessed toxic side effects on patients, as the cardiac, pulmonary, neurological and renal toxicity, besides to their limited anticancer activities which impeded the conquering of this disease [2,3]. Nowadays, there is an increasing demand to discover new anticancer agents that are more selective and less toxic than those currently used [4]. The natural metabolites acted as a repository of various bioactive compounds, which considered a valuable source of anticancer agents [3,5,6]. Launaea Cass. (Asteraceae, Cichorioideae, Cichorieae) is polymorphic and controversial genus with confused intraspecific delimitation. It comprised about 54 species and 10 subspecies distributed in different geographical regions [7]. It had economic values, ecological and ethnobotanical importance [8-10]. Despite the richness of Launaea species with different phenolic compounds such as flavonoids, coumarins and terpenoids [11-14], only limited data was available concerning the anticancer effects of its members.
This study aimed to assess the anti-proliferative activities of L. fragilis and L. nudicaulis ethanol extracts through the Sulforhodamine B (SRB) assay, in vitro, on six cell lines; hepatocellular carcinoma (Hep G2), prostatic carcinoma (PC-3), colorectal carcinoma (HCT116), non-small cell lung carcinoma (H1299), breast adenocarcinoma (MCF7) and colon adenocarcinoma (Caco-2).
Methods
Launaea fragilis and L. nudicaulis were collected from their natural habitats in flowering season 2015. The former was gathered from Ras-El-Hikma (N31.19107, E27.78305), El-Gophera village, 180 Km Alexandria-Matruh road (N31.05661, E28.08929), El-Dabaa, Alex- Matrouh International coastal road (N31.0291, E28.44343), 150 Km Alexandria-Matruh road (N31.01645, E28.6005), Burg El-Arab- El-Hammam road (N30.94875, E29.50859), Sidi Krir (N31.01733, E29.6339), Abu Qir, Alexandria (N31.32621, E30.06425), as well as Edku and Edku Lake (N31.30612, E30.29934). On the other hand, Launaea nudicaulis was collected from Wadi Halazeen, 45 km west Matrouh-Salloum road (N31.20902, E26.80541), Wadi Um-Ashtan, Matrouh (N31.27938, E27.0209), 150 Km Alexandria-Matruh road (N31.01645, E28.6005), El-Omayed (N30.83975, E29.28817), Burg El-Arab-El-Hammam road (N30.94875, E29.50859), 97 Km Alex- Cairo road (N30.49128, E30.27832) and 107 Km Alex-Cairo road (N30.4392, E30.34149). Voucher specimens were deposited at Alexandria University Herbarium (ALEX), Egypt. Specimens were investigated and morphologically identified according to Kilian [7] and Boulos [15] at the latter herbarium.
Anti-Proliferative Assay
A pooled sample was prepared for each species from their aerial parts. Ten grams of powdered sample was macerated in 95% ethanol (100 ml) for 24 hours at room temperature. The solution was filtered, and the filtrate was evaporated. Extracts were tested at the National Cancer Institute, Cairo, Egypt, by serial subcultures according to Skehan et al. [16]. Surviving fractions of cells throughout extract exposure was characterized graphically by IC50 values. IC50 values were estimated by a linear least-squares regression of the growth values versus the logarithm of the extract concentration; only concentrations that yielded growth values between 10% and 90% were used in the calculation. A bar parallel to the x-axis and intersecting the point 50% on the y-axis was constructed. Also, a bar was plotted parallel to the y-axis that starts from the point of intersection of the bar with the dose-response plot. Therefore, IC50 could then be determined at the point of intersection with the x-axis [16].
Growth Inhibition Percentage (GIP)
Growth Inhibition Percentage (GIP) was calculated based on Mosmann [17] equation.
GIP= [100- (Treated survival cells /control cells) × 100]
Results
Survival Percentage
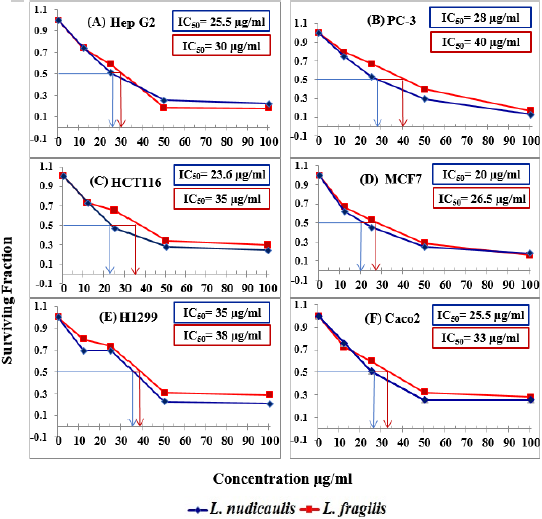
Figure 1 represents dose response curves constructed for SRB method between the ranges of 0-100 μg/ml. According to the rule that the lower the IC50 value, the stronger the inhibitor, where strong inhibitors are active at lower extract concentrations. Consequently, the obtained results showed that the incubation of L. fragilis with H1299 inhibited significantly the cell proliferation with IC50=26.5μg/ml. Subsequently, Hep G2, Caco-2 and HCT116 cell lines moderately suppressed the cell proliferation with IC50 was 30, 33 and 35μg/ml, respectively. Nevertheless, MCF7 and PC-3 showed the lowest inhibition values with IC50 were 38 and 40μg/ml, respectively. Alternatively, the incubation of L. nudicaulis with H1299 inhibited successfully the cell proliferation with IC50= 20μg/ml, followed by HCT116 with IC50= 23.6μg/ml. Both Caco- 2 and Hep G2 cell lines were suppressed with IC50=25.5μg/ml, whereas PC-3 recorded IC50= 28μg/ml. However, MCF7 revealed the lowest inhibition value with IC50= 35μg/ml.
Figure 1: Effect of Launaea fragilis and Launaea nudicaulis ethanol shoot extracts on (A)Hep G2; (B)PC-3; (C)HCT116; (D) MCF7; (E)H1299 and (F)Caco2 cell lines.
Growth Inhibition Percentage (GIP)
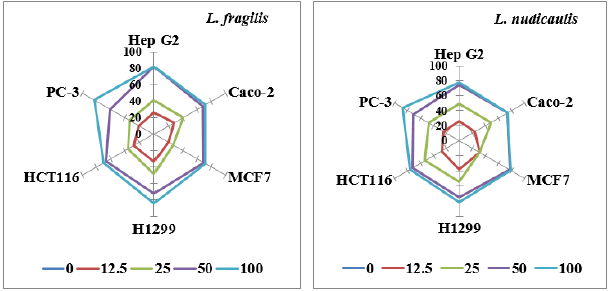
GIP is illustrated and statistically represented in Figure 2. The effect of the two extracts was examined as dose-response experiment after 48 hours, at concentrations of 0.0-100 μg/ml. It was notable that the inhibition of cancer cells increased with the increase of the extract concentration, although it was noticed that at 100% extract concentration, the number of inhibited cells is nearly the same or slightly higher than that obtained at 50% extract concentration. For L. fragilis, the highest inhibition percentage was 81.7% with Hep G2, while the lowest inhibition percentage was 60.4% observed for PC-3. Nevertheless, the percentage was in descending order; H1299> MCF7> Caco-2> HCT116 with values were 71.4, 69.4, 68 and 66.3% respectively. Launaea nudicaulis exhibited the highest inhibition percentage (77.2%) with MCF7, while, the lowest percentage (70.8%) was noticed for PC-3. Furthermore, the inhibition percentage was arranged in a decreasing order from H1299, Hep G2, Caco-2 followed by HCT116 cell lines, with values 75.2, 74.6, 74.4 and 72.6% respectively.
Figure 2: Growth Inhibition Percentage (GIP) of Launaea fragilis and Launaea nudicaulis ethanol shoot extracts against six human cell lines by using five different extract concentrations (0 “control”, 12.5%, 25%, 50% and 100%).
Discussion
Wild medicinal plants present a vast amount of new compounds, which can be used in the modern pharmaceutical research. Nowadays, up to 60% of the currently used anti-tumorigenic drugs, are derived from natural sources [18-19]. The use of natural alternative medicines becomes urgently and necessary. Previous studies of Rashid et al. [20] were carried to test extracts of L. nudicaulis and L. resedifolia (as quoted by González [21], Launaea fragilis is the correct name for the latter species) against beetles and larvae, also he stated that both attain cytotoxic activities. Launaea fragilis is common, polymorphic and possesses a wide range of tolerance [22]. It contained some phenolic compounds such as flavonoids, coumarins, saponins and phenolic acids [12,14]. The incubation of L. fragilis with H1299 resulted in a strong inhibition for the growth of cancer cells, while, the moderate effect was recorded for Hep G2, Caco-2 and HCT116. On the other hand, the lowest inhibition value is recorded for PC-3. Launaea fragilis is commonly used in the traditional medicine for the treatment of hepatic pains and gastric disorders as reported by Cheriti et al. [9]. Launaea nudicaulis is considered a polymorphic common species in the Egyptian floras [15,22]. Various extracts from aerial parts of the plant exhibited significant scavenging activity; as a result, it can protect the body from damages caused by free radical induced oxidative stress. This higher antioxidant activity was referred to the presence of natural products such as flavonoids, tannins, cardinolids alkaloids, saponins and terpenes [23-24].
Launaea nudicaulis inhibited efficiently the cell proliferation of H1299. Subsequently, HCT116, Caco-2, Hep G2 and PC-3 cell lines exhibit moderate suppression of the cell proliferation, however, MCF7 revealed the lowest inhibition value. This species was traditionally used to treat gastric burns, stomach pain and constipation. Also, it was useful for hemorrhoids, children fever, in addition to the treatment of skin itches and eczema [9]. Similarly, Khan et al. [25] tested the alcoholic extract of Launaea procumbens and reported its significance in the inhibition of the tumor with IC50= 13±0.2 μg/mL. As well, the methanol extract of Launaea mucronata was evaluated against breast (MCF7), hepatocarcinoma (Hep G2), colon adenocarcinoma (HCT116) and cervix (HeLa) cell lines with IC50= 12.70, 19.60, 19 and 12.60 μg/ml; respectively [26].
Conclusion
Anti-proliferative effect of L. fragilis and L. nudicaulis ethanol extracts can act as bioactive sources of natural anticancer agents. Ethanol extract of L. nudicaulis is more effective in the inhibition of the cell proliferation than that of L. fragilis, for the six investigated cell lines. This may be ascribed to the higher antioxidant activity of the former species relative to the latter.
References
- (2019) World Health Organization: A report about Cancer.
- Alonso-Castro AJ, Villarreal ML, Salazar-Olivo LA, Gomez-Sanchez M, Dominguez F, et al. (2011) Mexican medicinal plants used for cancer treatment: pharmacological, phytochemical and ethnobotanical studies. J. Ethnopharmacol 133(3): 945-972.
- Shin SA, Moon SY, Kim WY, Paek SM, Park HH, et al. (2018) Structure-based classification and anti-cancer effects of plant metabolites. Int J Mol Sci 19(9): 2651.
- Zapata B, Betancur-Galvis L, Duran C, Stashenko E (2014) Cytotoxic activity of Asteraceae and Verbenaceae family essential oils. J Essent Oil Res 26(1): 50-57.
- El-Darier SM, Abdelhady EF (2017) Phytochemistry and cytotoxic activity of Annona squamosa L. fruit pulp against human carcinoma cell lines. Cancer Biol 7(3): 26-30.
- Roy A, Ahuja S, Bharadvaja N (2017) A review on medicinal plants against cancer. J Plant Sci Agric Res 2(1): 008.
- Kilian N (1997) Revision of Launaea Cass. (Compositae, Lactuceae, Sonchinae). Englera 17: 1-478.
- Al-Mahrezi JA, Al-Sabahi JN, Akhtar MS, Selim D, Weli AM (2011) Essential oil composition and antimicrobial screening of Launaea nudicaulis grown in Oman. IJPSR 2(12): 3166-3169.
- Cheriti A, Belboukhari M, Belboukhari N, Djeradi H (2012) Phytochemical and biological studies on Launaea Cass. genus (Asteraceae) from Algerian Sahara. Curr Top Phytochemistry 11: 67-80.
- Heneidy SZ, Halmy MW, Bidak LM (2017) The ethnobotanical importance and conservation value of native plants in eastern Arabian Peninsula. Feddes Repert 128(3-4): 105-128.
- Mansour RM, Ahmed AA, Saleh NA (1983) Flavone glycosides of some Launaea species. Phytochemistry 22(11): 2630-2631.
- Giner RM, Díaz J, Máñez S, Recio MC, Soriano C, et al. (1992) Phenolics of Spanish Launaea species. Biochem Syst Ecol 20(2): 187-188.
- Ahmed Z, Ali D, Malik A (2006) Structure determination of ursene‐type triterpenes by NMR techniques. Magn Reson Chem 44(7): 717-719.
- Gherraf N, El-Bassuony AA, Zellagui A, Rhouati S, Ahmed AA, et al. (2006) Isolation of coumarins and coumarin glucoside from Launaea resedifolia. Asian J Chem 18(3): 2348.
- Boulos L (2009) Flora of Egypt Checklist. Cairo: Al-Hadara Publishing, Egypt pp: 249-252.
- Skehan P, Storeng R, Scudiero D, Monks A, McMahon J, et al. (1990) New colorimetric cytotoxicity assay for anticancer-drug screening. JNCI: J Natl Cancer Inst 82(13): 1107-1112.
- Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65(1-2): 55-63.
- Da Silva AC, Do Nascimento RM, Do Nascimento Rodrigues DC, Ferreira PM, Pessoa C, et al. (2019) In vitro activity evaluation of seven Brazilian Asteraceae against cancer cells and Leishmania amazonensis. S Afr J Bot 121: 267-273.
- Hussain M, Khera RA, Iqbal J, Khalid M, Hanif MA (2019) Phytochemicals: Key to effective anticancer drugs. Mini-Rev Org Chem 16(2): 141-158.
- Rashid S, Ashraf M, Bibi S, Anjum R (2000) Insecticidal and cytotoxic activities of Launaea nudicaulis (Roxb.) and Launaea Resedifolia (Linn.). Pak J Biol Sci 3(5): 808-809.
- (1979) González GL. Launaea fragilis (Asso) Pau, nombre correcto para L. resedifolia auct. plur. non (L.) Kuntze. Anales Jard Bot Madrid 36: 135-138.
- (1974) Täckholm, V Students’ Flora of Egypt. 2nd (Eds.), Cairo University Publishing, Egypt.
- Belboukhari M, Cheriti A, Belboukhari N (2014) In vitro antioxidant activity of Launaea nudicaulis (Asteraceae) growing in Southwest of Algeria. Ann Sci Technol 6(1): 52-55.
- El-Sharkawy ER, Ed-Dra AB, Abdallah EM (2017) Phytochemical, antimicrobial and antioxidant properties of Launaea nudicaulis and Farsetia hamiltonii. J Biol Control 31(2): 102-109.
- Khan RA, Khan MR, Shah NA, Sahreen S, Elahi SN (2016) Antitumor characterization of various fractions of Launaea procumbens. Toxicol Ind Health 32(1): 188-191.
- El-Sharkawy ER, Mahmoud K (2016) Cytotoxicity of two new coumarin derivatives isolated from Launaea mucronata. Nat Prod Res 30(4): 394-398.

 Short Communication
Short Communication