ABSTRACT
Introduction: MDR-TB is a disease that is caused by M. tuberculosis that is resistant to at least the two most effective and important TB drugs, Isoniazid (INH) and Rifampicin (RFM (. Multidrug and extensively drug-resistant tuberculosis (M/XDR-TB) is a manmade phenomenon that emerges as a result of a major challenge toward TB control and prevention. Approximately 650.000 MDR-TB cases worldwide were estimated by (WHO) in 2011. The objective of the study was to assess multidrug resistance tuberculosis (MDR-TB) pattern of M. tuberculosis and risk factors for drug resistant TB in Yemen.
Methods: A retrospective cross-sectional study was used. Data collected from January 2014 to December 2014 in National Tuberculosis Institute (NTI) in Sana’a. For TB drug resistant drug susceptibility testing on M. tuberculosis (MTB) isolates with first-line anti-tuberculosis drugs of culture positive TB cases was done. The MDR-TB was investigated and the proportion method for frequency was calculated. Multiple logistic regression analysis was performed to assess the risk factors. Total sample was 414. There were 6 variables analysed as risk factors.
Result: 0f 414 isolates examined for DST; among them 397 were new cases and 17 were previously treated. The mean age for patients was 32.60 ± 14.571 years (SD). The MDR-TB prevalence was 11 cases (2.7%) of all cases, 8 cases (2%) among new cases and 3 cases (17.4%) among the previously treated patients, respectively. Treatment history was only the significant risk factor, adjOR=10.42 (95% CI: 2.493-43.541, P =0.002).
Conclusion: The burden of multidrug-resistant TB cases was low among new cases but moderately high among previously treated cases. The need to reinforce control, detection and rigorous measures for treatment strategies for drug resistant TB is necessary. Periodic survey to detect MDR-TB and availability of second-line drug is an urgent need.
Keywords: Multidrug Resistant Tuberculosis; Risk Factors M Tuberculosis; Yemen
Abbreviations: NTI: National Tuberculosis Institute; RFM: Rifampicin; INH: Isoniazid; WHO: World Health Organization; DST: Drug Susceptibility
Introduction
Multidrug-resistant tuberculosis (MDR-TB) is defined as a tuberculosis that is resistant to at least isoniazid (INH) and rifampicin (RFM) which are the backbone of short-course chemotherapy for tuberculosis [1]. An inadequate drug treatment of an individual with TB will kill the majority of bacteria but will allow a small number of resistant bacteria to grow within those bacterial populations which are arising by spontaneous mutation [2-4]. The emergence of multidrug-resistant for M. tuberculosis isolation has increased worldwide and reached epidemic rates in some countries [5]. That is often attributed to inappropriately management of tuberculosis cases and inadequate control programs for disease [6]. Therefore, the resistance of M. tuberculosis for drugs reflects the poor quality of control programs and is a direct result of poor treatment practices concerning the use of anti-TB drugs7. An inappropriate treatment plays a dual role with respect to MDR-TB where it can be either a risk factor of resistance or a result of MDRTB7.
These relationships are so widely recognized that MDR-TB is classified as being due to previous treatment (acquired resistance) if the patient has been treated before and attributed to an infection with an MDR strain of M. tuberculosis (primary resistance) if detected in newly diagnosed patients [7]. Patients with MDR-TB provide a greater opportunity for transmission this type of tuberculosis due to the longer period during which these patients remain infectious [8]. Lack of the second line for treatment will lead to a frequent incidence for MDR-TB in population. Between 1999 and 2002, the World Health Organization (WHO) has documented the globally prevalence of MDR-TB by 1.7% in all cases, 1.1% in new cases, and 7% in previously treated cases respectively [9]. However, in the latest report, WHO (2013) estimated the globally MDR prevalence by 3.6% (95% CI: 2.1-5.1%) of new cases and 20.2% (95% CI: 13.3– 27.2%) of previously treated cases. This remarkable increasing in MDR-TB forms strong challenges and has complicated the control of the disease [10-12]. Therefore, immediate identification of these resistant isolates is very important for adjustments in treatment [13].
In Yemen; the most important challenges which face the health authorities are the lack of the second-line for treatment, the longterm for the treatment of MDR-TB, costly and failure to react by scaling‐up the country response to multidrug resistance; and thus may lead to an epidemic of M/XDR-TB in the future. Also; limited national resources are insufficient to fulfil all requirements in providing comprehensive service for MDR-TB management [14]. The objective of this study was to determine the prevalence of resistant to the tow major anti-tuberculosis drugs, i.e., isoniazid (INH), rifampicin (RFM) to assess the current performance of the NTP in Yemen.
Methods
Study Design
A retrospective cross-sectional study was used.
Study Population
All reported-positive tuberculosis cases enrolled in Sana’a NTCP institute in the period from January to December 2014 and which were examined for Drug susceptibility testing (DST) were considered the Study Population.
Drug Susceptibility Testing (DST)
All positive sputum specimens were identified, and Drug susceptibility testing (DST) was done in the NTI. For isolation of culture, each specimen was treated with 4% NaOH to one volume of sputum and then homogenized by vigorous stirring. An aliquot of 0.1 ml of the specimen thus treated was inoculated into two tubes of 2% Kudoh media and incubated at 370C. The culture was read in week 1 and after 4 weeks to detect rapid and slow growing, following which observations were made every week for 8 weeks until the result was recorded as negative [15]. M. tuberculosis growing on media was differentiated from other mycobacteria by growth characteristics, colony morphology, the niacin test by a paper strip and the P-Nitrobenzoicacid susceptibility test. DST was performed both at the National TB Reference Laboratory at the NTI, Sana’a city for confirmation. The proportion method was carried out for DST using Löwenstein-Jensen (L-J) medium at the NTI or Ogawa medium at the RIT.
The critical proportion of resistance was 1% for all drugs; the critical concentration was as follows: 0.2 μg/ml for INH, 40 μg/ml for REP, 4.0 μg/ml for SM with (L-J) medium and 10μg/ml with Ogawa medium and 2.0 μg/ml for ETB with L-J medium and 2.5 μg/ml with Ogawa medium. A preliminary reading for DST was made at week 2, 3 and finally after 4weeks [16]. Resistance was expressed as the percentage of colonies that grew on the drugcontaining medium compared to those on control medium [17].
After that MDR-TB was identified and thereafter, the patients were classified into two groups: MDR-TB and non-MDR-TB. Information was collected and analyzed. All culture-confirmed cases by Drug Susceptibility Test (DST) were taken as a sample size for the study after satisfying inclusion and exclusion criteria. Exclusion criteria were non-Yemeni people, cases with missed information and contaminated culture.
Sample Size
The sample size was determined according to Kish, L. (1965, 1995) where the sample size for a simple random sample without replacement was calculated as following formula [18]:
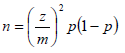
Where Z=1.96 for 95% confidence level, M =0.05 (5%) margin of error and P=50%was the expected value for the proportion of a sample that will respond a given way to a study question. For solving the equation: using my selected factors
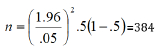
However, operationally all culture-confirmed cases which were examined for Drug Susceptibility Test (DST) were taken as a sample for the study rather than the calculated sample size.
Data Collection
Data was collected from laboratory isolates during January 2014 to December 2014 and matched to the cases registry in the tuberculosis system. All information about patients were taken from the registers where these registers contain all information such as; demographic information (age, sex, place of live) clinical information (new cases, previous treatment cases) for TB and socioeconomic information by calling the patients by their phone numbers which were presented in cases register to ask them about their missed information such as (education level, smoking) in order to study and analysis them.
Study Variables Definition
There were 7 variables analyzed. Dependent variable was multidrug-resistant TB isolates for the drugs used in first line treatment (rifampicin, isoniazid, ethambitol and streptomycin) or non-multidrug resistant. MDR-TB means resistant of M. tuberculosis for at least rifampicin and isoniazid together, and with or without the other drugs (streptomycin and ethambitol). Any other form of resistant was considered NON MDR-TB. Independent variables were age (<40 years old, >40 years old), type of patient (new, previous treatment), sex (male, female), place of live (rural, urban), level of education (literate, illiterate) and smoking (smoker, non-smoker).
Statistical Analysis
Collected data was coded and transferred to SPSS version 21 packages. Calculation of mean and SD for age by t-test and for comparison of categorical variables, significance testing was done by χ2 test with by 2-sided correlation test as appropriate. Associations between selected factors were estimated by computing odds ratios (ORs) and their 95% confidence intervals (CIs) from a logistic regression model. The criterion for significance was set at P < 0.05 based on a two-sided test. Analyses were conducted with SPSS version 21.0 (SPSS Inc., Chicago, IL, USA).
Ethics Statement
The research proposal was approved by the ethical committee of University of Science and Technology. All participants were verbally informed about the objectives of the study (oral consent), and forms of data collections were filled depending on principle of confidentiality using non linked information (no names will be written on the questionnaire). As well as official letter for permission of conducting the study to the national tuberculosis institute was provided.
Results
Characteristic of Patients
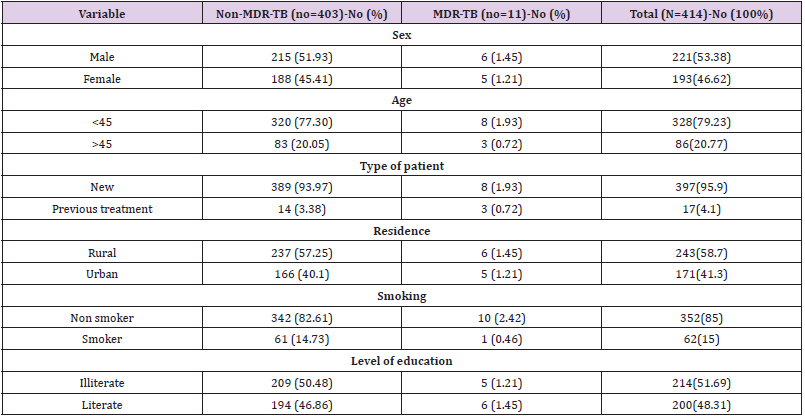
Of 650 cases of tuberculosis identified by primary culture between January to December 2014 in National Tuberculosis Institute in Sana’a; 445 isolates were examined for Drug susceptibility Test (DST). Of these isolates 4 cases were not Yemeni people and 6 isolates reported as contaminated and 21 of cases we cannot contact to complete the missing information. The rest of isolates (414) were taken for this study. The prevalence of MDR-TB was 2.7 % (11 cases) and NON-MDR TB was 97.3% (403). Of 414 cases, the males were 221(53.38%). Among them 6 cases (1.45%) were MDR-TB comparing of 5 cases MDR-TB in females which were193 (46.62%). The result was not statistically significant; adjOR=1.24(95% CI: 0.297-5.184, P>0.05). The main age for patients was 32.60 ± 14.571 years (SD). As in the Table 1; MDRTB was more frequent in patients less than 45 years old; where of 328(79.23%) cases less than 45 years old; MDR TB prevalence was 1.93 % (8 cases). In patients more than 45 years old; MDR TB prevalence was 0.72% (3 cases). The result was not statistically significant; adjOR=1.2 (95% CI: 0.974-1.081, P> 0.05).
Table 1: Characteristics of Patients with Culture Confirmed Pulmonary Tuberculosis: comparison of patients with MDR-TB and NON MDR-TB in Yemen in 2014.
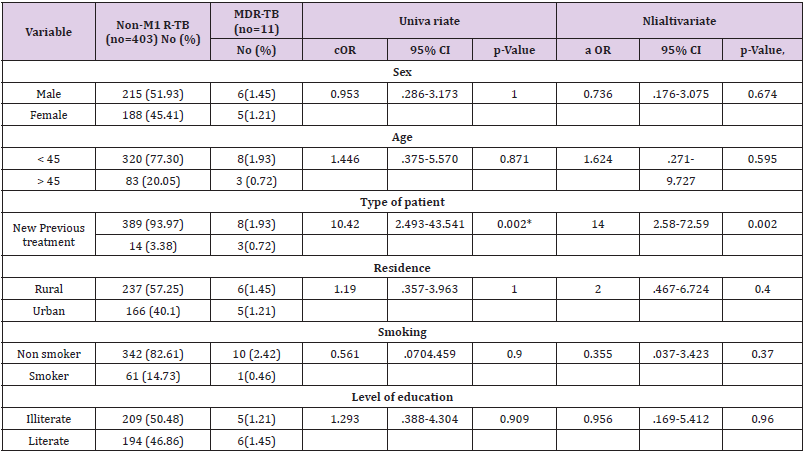
Of 414 patients; the new patients were 397 cases (95.9%); where the prevalence in this new patient group was 2% (prevalence= 8 ∕ 397) and in the previous treatment patients was 17.6 % (prevalence = 3 ∕17). The MDR TB in this group was more frequent than the new patient group. This result was statistically significant; adjOR=10.42 (95% CI: 2.493-43.541, P =0.002). Of 414 patients; non smoker patients were 352(85%) of them 10cases (2.42) were MDR TB. Smoker patients were 62(15%) of them 1 case (0.46%) was MDR TB. This result was not statistically significant; adjOR=2.17 (95% CI: 0.29-22.44, P > 0.05). Of 414 patients; 243 cases (58.7%) were lived in rural area. Of them 6 cases (1.45%) were MDR-TB comparing of 5 MDR-TB cases (1.21%) of 171 cases (41.3%) were lived in urban area. The result was not significant, adjOR=1.975 (95% CI: 0.513-7.598, P > 0.05). According to education, the illiterate patients were 214 cases (51.69%) and MDR-TB prevalence among them was 5 cases (1.21%) comparing of 6 cases among literate patients. The result was not significant; adjOR= (0.9: 95% CI 0.834-1.212, P > 0.05).
Discussion
Information on anti-tuberculosis drug resistance levels is an essential management tool for evaluating the performance of national TB control programmers (NTPs). Resistance in previously treated cases is an indicator of current treatment practices in the community. Drug resistance in new cases reflects transmission of disease with resistant bacilli [19]. This present study shows that the prevalence of MDR-TB was 2.7% (11 cases) for all cases and the prevalence between the new patient group was 2% (prevalence = 8 ∕ 397) and among previously treated patients’ group was more frequent than the new patient group where the prevalence was 17.6% (prevalence= 3 ∕17). This result was statistically significant; (adjOR=10.24 (95% CI 2.41- 43.541, P =0.002) and consistent with most studies done in analyzing MDR-TB. Compared with the national survey that has been conducted in 2010-2011, which estimated the rates for MDR-TB in new and previously treated cases by 1.4% and 14.4%, respectively 14, the MDR-TB prevalence among previously treated cases and among new cases was modestly higher than the national average but this rates were slightly down from the globally estimation reported by WHO in (2013) which estimated the MDR-TB prevalence by 3.6% (95% CI: 2.1–5.1%) of new cases and 20.2% (95% CI: 13.3–27.2%) of previously treated cases 10.
Compared with neighboring countries; Saudi Arabia multidrugresistant TB were 3.4% [20]. Rates in the United Arab Emirates and Kuwait are among new 2.2% (95% CI: 0.0-10.8) and 10.8% (95% CI: 0.0-34.5) among previously treated patients. In morocco the prevalence was 0.5% among newly treated (95% CI: 0.2-1.1) and 12·2 % (95% CI: 8.2-17.7) among previously treated patients. In Iran the prevalence was 5% among new patients (95% CI: 3.5- 6.9) and 48.2% (95% CI: 35.7-61.0) among patients. In Oman the prevalence of MDR-TB among new was 2.2% (95% CI: 0.7-6.2) and 8.3% (95% CI: 0.4-35.4) among previously treated cases. In Jordon 5.4 % (95% CI: 2.5-11.3) among new patients and 40% (95% CI: 24.6-57.7) among previously treated [21]. In Somalia MDR-TB was detected in 5.2% (95% CI: 2.8–7.5) of newly diagnosed TB patients and in 40.8% (95% CI: 24.7–57.0) of previously treated TB [22]. Resistant to the first-line drugs were usually difficult to be detected because culturing and drug susceptibility testing occurs only in two central laboratories; Aden and Sana’a and are not routinely performed at the other centers and districts tuberculosis clinics. The use of standard first-line drugs in the treatment of these patients may increase the risk of relapse, treatment failure and acquired drug resistance [19]. As in the Table 2; this present study shows that there were not significant association between the following variables, age (adjOR=01.62: 95% CI 0.271-9.727, P=0.871), sex (adjOR=1.24: 95% CI .297-5.184, P> 0.05), smoking (adjOR=2.17: 95% CI: 0.29-22.444, P >0.05), place of live (adjOR=1.773: 95% CI .467-6.724, P>0.05) and level of education (adjOR= 0.9: 95% CI: 0.834-1.212 p=0.909). These results were consistent with many previous studies.
Study Limitations
Patients’ related information was retrieved from medical records. Missing and wrong data was possible to affect the result and interpretation. The other main limitation is that I could not include other important variables associated with MDR-TB such as HIV infection; diabetes mellitus, cavitary chest lesion, level of habitant and socioeconomic factors.
Conclusion
This study conducted under difficult circumstances in the country with unstable social conditions where the security situation is most disturbance and disruption of care more frequent. In additional to Yemen also is most affected by recent food shortages and has the most internally displaced persons, these factors are expected to exacerbate disease progression and transmission of M. tuberculosis [23]. Thus, MDR-TB is expected to be a serious under-detected and widespread public health problem in Yemen. Despite this, the study could determine the burden of multidrugresistant TB cases that was low among new cases but moderately high among previously treated cases. This study recommends the need to reinforcement control, detection and rigorous measures for treatment strategies for drug resistant TB are necessary. Periodic survey for detect MDR-TB and availability of second-line drug is an urgent need.
Acknowledgment
Acknowledge with deep appreciation and gratitude to my supervisor; Associated Professor Dr. Azmi Mohammed Tamil for his expertise and support. I would like to thank everyone in National Institute of TB who supported me to achieve this study.
References
- Laura HerreraLeón, Tamara Molina, Pilar Saíz, Juan Antonio Sáez Nieto, Maria Soledad Jiménez (2005) New multiplex PCR for rapid detection of isoniazid resistant Mycobacterium tuberculosis clinical isolates. Antimicrob Agents Ch 49(1): 144-147.
- Borrell S, Gagneux S (2009) Infectiousness, reproductive fitness and evolution of drug-resistant Mycobacterium tuberculosis. Int J Tuberc Lung Dis 13(12): 1456-1466.
- Surendra K Sharma, Gaurav Kaushik, Brajesh Jha, Ninoo George, SK Arora, et al. (2011) Prevalence of multidrug-resistant tuberculosis among newly diagnosed cases of sputum-positive pulmonary tuberculosis. Indian J Med Res 133(3): 308-311.
- (2013) Centers for Disease Control and Prevention. Treatment of Tuberculosis, American Thoracic Society, CDC, and Infectious Diseases Society of America. MMWR 52: 11.
- Mokrousov I, Otten T, Vyshnevskiy B, Narvskaya O (2003) Allele-specific rpoB PCR assays for detection of rifampin resistant Mycobacterium tuberculosis in sputum smear. Antimicrob Agents Ch 47: 2231-2235.
- Antônio C (2013) Multidrug-resistant tuberculosis. The brazj infectdis 17(2): 239-246.
- Faustini A, Hall AJ, Perucc CA (2006) Risk factors for multidrug resistant tuberculosis in Europe: a systematic review. Thorax 61(2): 158-163.
- Laura F Anderson, Surinder Tamne, Timothy Brown, John P Watson, Catherine Mullarkey, et al. (2014) Transmission of multidrug-resistant tuberculosis in the UK: a cross-sectional molecular and epidemiological study of clustering and contact tracing. Lancet Infect Dis 14(5): 406-415.
- (2004) World Health Organisation. Anti-tuberculosis drug resistance in the world, WHO Report No 3. Geneva: World Health Organisation.
- (2013) World Health Organization. Global tuberculosis report 2013. Geneva: World Health Organization WHO/HTM/TB/2013.11.
- (2000) WHO. Anti-tuberculosis Drug Resistance in the World. Report 2: Prevalence and Trends. WHO, Geneva.
- (2000) WHO. Drug-Resistant Isolates of TB Increasing Worldwide? WHO, Geneva?
- Abe C, Aono C, Hirano K (2001) Evaluation of the BACTEC MGIT 960 system for drug susceptibility testing of Mycobacterium tuberculosis isolates compared with the proportion method on solid media. Kekkaku 76(10): 657-662.
- (2013) National Tuberculosis Control Programme. National Strategic Plan for the National Tuberculosis Control Programme 2013-2017. Sana’a, Republic of Yemen: Ministry of Public Health & Population.
- Fujiki A (2001) TB bacteriology examination to stop TB. Tokyo, Japan: Research Institute of Tuberculosis.
- Abe C, Kobayashi I, Mitarai S (2008) Biological and molecular characteristics of Mycobacterium tuberculosis clinical isolates with low-level resistance to isoniazid in Japan. J Clin Microbiol 46(7): 2263-2268.
- AL Akhali A (2007) Nationwide survey on the prevalence of anti-tuberculosis drug resistance in the Republic of Yemen, 2004. Int J Tuberc Lung Dic 11(12): 1328-1333.
- Kish L (1965) Survey Sampling. New York: John Wiley & Sons, Inc.
- Liu Q, Zhu L, Shao Y, Song H, Li G, et al. (2013) Rates and risk factors for drug resistance tuberculosis in Northeastern China. BMC Public Health 13: 1171.
- Laila Nimri, Hala Samara, Raymond Batchoun (2011) Detection of mutations associated with multidrug-resistant Mycobacterium tuberculosis clinical isolates. Federation of European Microbiological Societies. Immunol Med Microbiol 62(3): 321-327.
- (2011) World Health Organization. TB And MDR/XDR-TB in the EMR, The Global Burden of TB 2010, Anti-Tuberculosis Drug Resistance in The World. Fourth Global Report. WHO, Geneva?
- Ireneaus Sindani, Christopher Fitzpatrick, Dennis Falzon, Bashir Suleiman, Peter Arube, et al. (2013) Multidrug-resistant Tuberculosis, Somalia, 2010-2011. CDC; Journal: Emerging Infectious Diseases 19(3): 1281-1287.
- United Nations Office for the Coordination of Humanitarian Affairs.

 Research Article
Research Article