Abstract
Introduction: Candida is a normal flora of the oral cavity, gastrointestinal tract (GIT) and the vagina but has been reported to become opportunistic pathogen (switch) in certain disease conditions. An investigation into this switch in the GIT will give a clear insight into their progression from commensal to opportunistic pathogens. Therefore, this study aimed to isolate and evaluate the presence of Candida species and associated microbes causing GIT infections among patients accessing care at a tertiary healthcare facility in Lagos, southwest Nigeria
Methodology: A total of 150 consenting participants (M=82; F=68) age ranged 1-60 years were recruited into this study between July and December 2019 at the Lagos University Teaching Hospital, Idi-araba using standard methods. Demographic details were taken and characte¬ristics of the stool specimens were noted. Samples were examined macroscopically and microscopically, then cultured on Sabouraud dextrose agar with corn meal extract along with other bacteria culture media and incubated appropriately. CHROMagar TM for Candida was used for presumptive identification of isolates and was further confirmed with the API32C system for Candida.
Results: An overall rate for Candidiasis in this study was 26.7% (40/150) with female having higher rate 18.7% (28/150) than 14.7% in male (22/150). Twenty-five (16.7%) of the participants’ stool culture yielded pure fungi isolates of C. albicans. As for C. albicans, the female had higher rate (10%) than their male (6.7%) counterparts. However, by gender, this was not statistically significant (p=0.012). Pathogenic stool bacteria isolates were found in 16 (10.7%) and 84 (56%) of participants with normal flora of the GIT. The remaining 25 (16.7%) were reported as mixed culture of Candida spp. Also, C. albicans and C. dubliniensis were found as a pair in seven stool samples from participants with cases of peptic ulcer disease 1(14.3%), gastrointestinal bleeding 4(57.1%)), immune suppression 2(28.6%) while others occurred in combinations of threes and fours of different species. Obvious clinical manifestations when compared with the study findings revealed that C. albicans co-existed as the causative fungi causing gastroenteritis in 17% of the participants whose stool culture yielded pure growth.
Conclusion: This study found comparable high burden of Candida isolates and associated GIT microbes. Although the rates were higher in female than their male counterparts, these findings were however not statistically significant, this therefore calls for better hygiene practice through educative advocacy. Effective management of opportunistic mycoses depends on identification of the specific organism causing the infection otherwise, a systemic infection of this type can be fatal.
Keywords: Candida Species; GIT infections; CHROMagar TM; Sabouraud Dextrose Agar
Introduction
Candida species reside in the human gastrointestinal tract as part of the body’s microbiota. Due to changes in host environment as a consequence of disease conditions (such as immunosuppression, metabolic imbalances and dysbiosis), they can proliferate as opportunistic pathogens [1,2]. They are the predominant cause of opportunistic human mycoses, and are capable of causing superficial as well as invasive mycoses [3] Candida proliferation, especially with heavy growth, within the gut may result in and abdominal discomfort [4,5]. There are many Candida species harvesting the GIT and any of them can cause gastrointestinal candidiasis among disease patients; however, Candida albicans is the most common [6].
Studies reporting prevalence of gastrointestinal candidiasis at the global and continental levels are scarce, and studies in various countries have yielded varying results [2,7,8]. Previous studies carried out in different centres in African continent reported a non-uniform prevalence of gastrointestinal candidiasis with a varying rate of 12.4% but with a slightly higher prevalence in sub- Saharan Africa at 12.8% [9]. Both C. albicans and non-albicans Candida species revealed high rates of intrinsic and acquired forms of antifungal resistance [5,10-12]. Other microscopic fungi that may become opportunistic pathogens apart from Candida species include Aspergillus, Mucor, and Cryptococcus [11]. When specimens from infected patients are cultured, Candida spp are considered as pathogens when isolated either in pure or mixed culture and counts ≤ 103, 104 and ≥105 cfu/ml are considered to be significant [13].
Successful treatment of opportunistic mycoses depends on identification of the specific organism causing the infection. Without effective therapy a systemic infection of this type can be fatal [14]. Furthermore, the yeast is also common commensal of the gastrointestinal tract (GIT). Most Candida infections are opportunistic, occurring in debilitated persons and also in persons with prolonged broad-spectrum antibiotics therapy. Roberth et al. [15], Edwards, [16] noted that even when the digestive flora remained balanced in otherwise healthy persons who lack beneficial bacteria in their gut (due to bad life style or bad feeding habits, probably on antibiotic, chemotherapy or on other strong chemicals); C. albicans was seen to over-proliferate and cause ill health.
Opportunistic fungal infections are increasing around the world with limited information on their prevalence in gastrointestinal (GIT) infections [3]. Different clinical forms of Candidiasis are known involving primarily the mucosal surfaces (thrush), GIT or urogenital tract, and deep-seated infections such as Candidaemia or Meningitis [17]. It is therefore pertinent to determine the level of pathogenicity of intestinal fungal infections in the study environment. In Nigeria the problem is compounded due to laboratory challenges and inadequate facilities in many healthcare centres, which hinders the timely diagnosis and treatment of such infections. This study therefore aimed to isolate and evaluate the presence of Candida species and associated microbes causing GIT infections among patients accessing care at a tertiary health facility in Lagos, southwest Nigeria
Methodology
Methods Ethical Considerations
This study was approved by the Ethical consent and clearance from the Management of the Lagos University Teaching Hospital (LUTH), Idi-araba with Reference approval number No: NHRECO4/04/2008. All participants were provided with written informed consent before the enrolment, and for participants younger than age 18 years, written informed accent was provided by a parent or legal guardian.
Study Design
This was a cross-sectional study carried out at the General Outpatient Department of the Lagos University Teaching Hospital. A total of 150 consenting participants (M=82; F=68) age ranged 1-60 years were recruited into this study between July and December 2019 at the Lagos University Teaching Hospital, Idi-araba along with the controls at random from both out-patient and in-patients clinics. The 80 control participants were the apparently healthy participants recruited from schools and offices at random within the ages of 1 and 60 years. Subsequently, informed consent of the participants was duly obtained in writing where they are adults and informed assent of guardians in the case of minor.
Inclusion Criteria
Only participants presenting with provisional diagnosis of GIT related diseases, clinical history of being immunocompromised, on chemotherapy, admission and between the ages of 1and 60 years were considered.
Exclusion Criteria
Participants with no GIT related illness or clinical history of being recently hospitalized or immunocompromised was not recruited for this study. Participants were randomly selected and informed consent was obtained. Eligible patients who picked cards with numbers that were multiples of three were enrolled in the study. The cards were reshuffled each time a card was picked and picked cards were not replaced. Where repeated numbers were generated, only one card with such numbers was left in the box.
Data and Sample Collection
Clinical data were obtained from the consenting participants’ medical records for demographic characteristics such as age, sex and antifungal medication. Stool specimens and blood samples were collected from the recruited participants. Participants were advised to look out for blood stained or mucoid portions of their freshly voided early morning stool sample and aseptically put directly into sterile wide mouth universal transparent container. The stool specimens were received and properly labeled with the participant’s name, date and time of collection. Briefly, a sterile, wide-mouthed spoon fitted with a graduated stool container was labeled for each participant and participants were instructed to produce about 10 mL of stool. The specimens were immediately delivered to the laboratory for subsequent analysis. One 4-mL fasting blood specimen was collected from each participant into fluoride/oxalate tubes and the plasma separated from the cells within 30 min after collection.
Sample Laboratory Analyses
Stool samples were examined macroscopically for presence of blood, mucus, texture, colour, present or absence of adult worm. Wet preparation of each stool sample including control was done on clean grease free slides using physiological saline, 40% dimethyl sulphide (DMSO) KOH and Lacto phenol cotton blue then allowed to stand for 10minutes. Slides were then viewed with X10 objective of the microscope for presence of yeast cells measuring 2 – 4μ in diameter and fungi hyphae. Gram stained smears were also examined for yeast cells and fungi hyphae under the microscope using the X100 objective of the microscope. The yeasts may often be seen attached to pseudohyphae that stain Gram positive [18].
Culture Media: Sabouraud dextrose agar (pH 5.6) containing corn meal extract was prepared to give a final pH (at 25°C) 6.0 which will enhance growth and preserve the chlamydiospores according to the manufacturer’s instructions. Bacteria stool culture media (Selenite F, MacConkey, DCA, SS agar) was prepared according to manufacturer’s instruction and used to detect or exclude bacterial isolates [18]. As for inoculation, with the use of sterile wire loop and streaking method to obtain discreet colonies on culture plates, all stool samples including control were inoculated into two different sterile freshly prepared Sabouraud dextrose agar plates containing corn meal extracts and antibiotics (chloramphenicol, streptomycin, gentamycin at a concentration of 100mg/liter of medium respectively) because of the bacteria load in stool specimens and also cultured on the different routine bacteria stool culture media mentioned above to identify and then exclude gastroenteritis of bacteria origin. All inoculated media including the Selenite F broth was incubated at 35°C – 37°C respectively. One of the Sabouraud agar plate were incubated at room temperature (18°C – 25°C).
Plate Reading: Presumptive fungi colonies were read based on their colonial appearance: size, shape, elevation, consistency (mucoid, dry or semi-mucoid), colour or pigmentation, and distinctive yeast (bread) smell. The number of colonies on plates showing pure fungal growth was counted. Those with counts ranging from ≤103, 104 and ≥105 cfu/g after 24 - 72hrs incubation were regarded as significant [13]. Plates that showed no growth after 7days incubation on the Sabouraud dextrose corn meal extract agar plates were disregarded and discarded appropriately. Bacterial isolates from the routine bacteriology culture plates were identified and their pathogenicity determined appropriately.
Identification Test Methods
These are test procedures that were used for the identification and characterization of the various fungal species isolated. The tests include Gram’s reaction. Smears of isolates were made on grease free slides, stained by Gram’s Method and examined under the x100 objective of the microscope. Yeast cells stained purple blue indicating that they were Gram positive. Gram negative organisms stain red. As for Germ tube test, suspension of pasty colonies of white or cream color on fungal culture plates were made in khan tubes containing 0.5 ml of human serum, incubated for 2 to 3hours at 37°C and examined microscopically (a drop of lacto phenol cotton blue was added to the drop of the suspension on the slide to stains the yeast cells) for the detection of yeast cells with sprouting /tubelike outgrowth from the cells (germ tubes). C. albicans is germ tube positive [18].
Preliminary Speciation of Candida using Special Selective Fungal Agar Media CHROMagarTM:
Suspected isolates were subcultured on BBL™ CHROMagarTM. The inoculated plates were incubated aerobically at 37°C for 24 hours. After incubation, the colours of the isolates were noted and identified on the colour chat.
Sugar Fermentation: Tests API 32C System: With the use of API 32C system, sugar fermentation test was carried out. Isolates were first purified by sub culturing on Sabouraud dextrose agar at least 2 to 3 times. Intermittent staining was done with Lacto phenol cotton blue and viewed under the X100 objective lens to be sure the isolate was yeast. A suspension from the culture medium was made in Sabouraud dextrose broth with a turbidity equivalent to 2 McFarland. From each of the suspension, 250μl was then transferred into an ampoule of the API 32C System medium. A volume of 135μl of prepared yeast basal medium suspension was then pipetted into wells containing 32 different freeze-dried sugars and incubated at 30°C for 24 to 48 hours. After incubation, growth was seen as turbidity in the well containing the sugars (substrate) in each cupule and was read using the ATBTM ExpressionTM or mini API instrument, or visually. Identification was obtained using the identification software into which the positive substrate code combinations are imputed and printed out [19]. Software identification was done at the Chemistry Department at FIIRO (Federal Institute of Industrial Research, Oshodi) Lagos state.
Growth at 45°CI: Suspected fungi colonies were inoculated into Sabouraud dextrose broth and incubated at 45°C and has been monitored for turbidity at different days for 7 to 10days. The ability of C. dubilinensis to grow at 45°C helps to differentiate it from C. albicans [20].
Chlamydiospore Formation: Suspected culture isolates were inoculated onto Sabouraud dextrose agar plates using the Dalmau technique i.e., on a microscopic glass slide and a light-tomoderate inoculum of the greisen tube test was transferred by loop and streaked lightly over an area slightly greater than that to be covered by the subsequently placed, sterile, 22mm cover glass which was then incubated at 37°C for up to 3hours. Periodically, this begins between 1- 1.5hours. Cultures were observed through the cover glass for germ tube production. The low-power objective was used to scan the inoculated area; the high-dry objective was used to confirm the presence of germ tubes. After germ tube detection of after 3hours at 37°C, the plates were incubated at room temperature (18°C – 25°C) for chlamydiospore formation, a drop of Lacto phenol cotton blue was added to improve clarity and staining of chlamydiospore [21].
Statistical Analysis
Descriptive statistics was used to describe the study characteristics of the participants. Continuous variables were summarized as means, standard deviations (SD) for normally distributed or medians, interquartile ranges [IQR] in the case of skewed data and categorical variables were presented as proportions. Data was double-entered into Microsoft Excel and Microsoft Access spread sheet to ensure accuracy of data placement into variables. The validated data was then transferred into SPSS version 20.0 for analysis. Data was summarized as number and percentages. The percentages of the different types of Candida species generated from the result of the study were compared between different disease conditions using Chi-square and ANOVA test. Statistical outcomes with P-value < 0.05 was considered to be significant.
Results
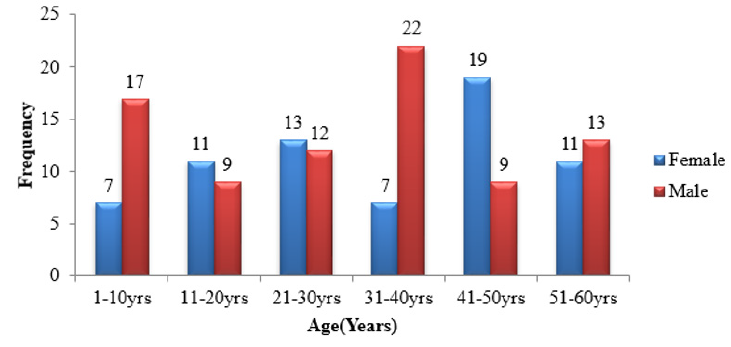
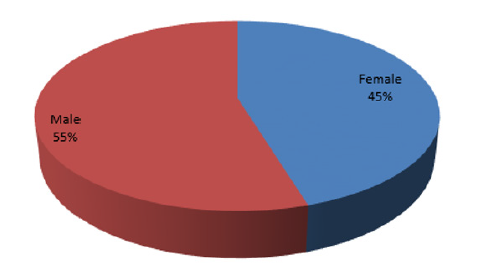
Stool samples were collected from 150 consenting participants, 82 (55%) of them were males and 68 (45%) were females (Figures 1 & 2). An overall rate of 26.7% (40/150) for Candidiasis was found in this study with female accounting for 18.7% as against 14.7% for the male counterparts. Age group (31– 40 years) had the highest population of 29 (19%) and the least came from the age group 11- 20 years with only 20 (13%) participants (Table 1).
Table 1: Gender Distribution of Isolates among Participants.
Chi-square= 28 Chi-square=21.0
P-value= 0.531 P-value =0.693
KEY: PBI => Pathogenic bacteria isolate. GIT (NF) => gastrointestinal tract (Normal flora)
Total Candida Spp=28 i.e Female (28/150=18.7%)
Total Candida Spp=22 i.e Male (22/150=14.7%); Total Candida Spp=40/150=26.7%.
Sixteen (10.7%) of the 150 participants’ stool culture yielded Pathogenic Bacteria Isolates (PBI) in both female and male participants. Seventy-six (56%) of the stool specimens yielded growth of normal flora of the GIT; the stool cultures comprised mainly of mixed bacteria colonies and scanty colonies of fungi. The microscopic examination of these stool specimens had very scanty or no yeast cells. Out of the 50 participants whose stool samples where positive for Candida spp, 25 of them yielded pure isolates of C. albicans and the remaining 25 occurred as mixed Candida isolates. Fifteen of the 25 pure C. albicans isolates were found among the female participants and the remaining 10 were from male participants (Table 1). Eighty-two (82) participants were males but only ten (10) were positive for fungal isolate (C. albicans) and 11 for pathogenic bacteria. Fifteen (15) female participants were positive for fungal isolate (C. albicans) and five (5) pathogenic bacteria were recorded among 68 female participants. A total of 84 (35 females and 49 males) participants had normal flora of the GIT in their stool cultures (Table 1).
Distribution of Isolates by Disease Conditions
Twenty-six of the participants were clinically diagnosed to have PUD, six of their stool cultures yielded Candida as isolates, 2 in pure cultures and 4 in mixed cultures. No pathogenic bacterium was isolated in this group, but 20 participants had isolates of normal GIT flora. From the 40 participants with diarrhea, there were 4 cases with fungi isolates: one in pure culture and 3 in mixed culture. Samples from six participants in the group yielded pathogenic bacteria isolates, while samples from 30 participants yielded GIT normal flora. Among participants with gastrointestinal bleeding, there were 12 cases of fungal isolates, 4 of these were pure isolates while 8 were mixed isolates. Samples from 2 and 6 participants in this group yielded pathogenic bacteria isolates and gastrointestinal normal flora respectively. Participants with immune suppression were 42. Samples from 14 of them yielded fungal isolates; 6 as pure isolates and 18 as mixed isolates. Culture of 8 participants’ samples yielded pathogenic bacterial isolates, while from 20 participants the isolates were bacteria of the normal gastrointestinal flora.
There were 21 HIV participants in the study. Thirteen of them had fungal isolates; 11 in pure culture and 2 in mixed culture. Samples from 8 participants yielded normal flora of the gastrointestinal tract. The sole participant on chemotherapy in this study had only fungal isolate in pure culture. Fifty of the 150 participants across all groups tested had fungal isolates in their cultures. Twenty five were in pure culture and another 25 in mixed culture. The microscopic examination of stool specimens yielding pure isolates revealed predominantly yeast cells with hyphae. While for those yielding mixed fungi isolates the microscopic examination revealed predominantly yeast cells only (Table 2).
Table 2: Distribution of Isolates by Disease Conditions.
Chi-square= 66.2. p-value = 0.000
KEY- PUD= Peptic Ulcer Disease.
HIV= Human immune-deficiency virus.
Age and Sex Distribution of Pure Candida Albicans Isolates Among Participants
Out of the 150 participants tested, 50 were positive for fungi culture but only 25 participants stool culture yielded pure Candida albicans isolates. The age group 51 - 60 years with 24 (11 females and 13 males) participants had pathogenic C. albicans infection in 25% of the participants in the group as the highest. As for C. albicans, the female had higher rate (10%) than their male (6.7%) counterparts. However, by gender, this was not statistically significant (p=0.012). The age group 1- 2 years with 24 participants comprising 7 females and 17 males, had the least rate (12.5%) of pure C. albicans infection. Among the age groups 11 -20 years and 21 – 30 years, the C. albicans infection isolation rates were 15% and 16% respectively. For the age ranges 31 – 40 years and 41 – 50 years, the rates of isolates were 13.8% and 17.9% respectively. Age group (31 – 40) years recorded the highest number of participants [29] in this study with 4 participants being infected and a frequency of 3% of the total population (Table 3).
Table 3: Age and sex distribution of Pure Candida albicans Isolates among Participants.
Chi-square= 14.7, p-value=0.012
NB: C. albicans, female had higher rate of 10% (15/150) than their male of 6.7% (10/150) counterparts.
The total number of mixed Candida isolates from the stool culture of 25 participants was 66, from 9 different Candida species in all the disease conditions. There were six different combinations of 2 species, seven different combinations of 3 species, and two different combinations of 4 species. Thus, the different groups of mixed candida isolates were 15 in number. Candida tropicalis, C, albicans, C. dubilensis, and C. lusitaniae were present in 12, 10, 5 and 4 mixed Candida isolates respectively. Candida parapsilosis was present in 3 different mixtures, while C membranaefasciens, C. kruzei and C. galbrata were each present in varied combinations of two species. Candida kefyr was present with C. tropicalis and C. dubilinensis, in only one combination of three mixed species isolated from a participant with peptic ulcer disease (Table 4). The mixed isolates of C. albicans and C. dubilensis were isolated from seven participants: 1 with peptic ulcer disease; 4 with gastrointestinal bleeding; and 2 with immune suppression. Each of the other five pairs of mixed candida isolates was isolated from one participant. No pair of Candida isolates was obtained from participants with immune suppression and those on chemotherapy. The combination of C. albicans, C. tropicalis and C. dubilinensis was isolated in three participants: 1 with peptic ulcer disease and 2 with immune suppression. While the combination of C. albicans, C. tropicalis and C. parepsilosis was isolated in two participants: 1 with diarrhoeal disease and 1 with gastrointestinal bleeding. Three other combinations of 3 candida species were each obtained from one participant in the group with immune suppression. The same combination of four candida species was isolated from two participants with gastrointestinal bleeding. While a different combination of four mixed candida isolates was obtained from a participant with immune suppression. There was no mixed candida isolates obtained from the stool cultures of participants on chemotherapy.
Candida albicans was part of the mixed isolates from 20 (80%) of the 25 participants. Candida tropicalis, C. dubliniensis and C. parapsilosis were present in the mixed isolates from 16 (64%), 13 (52%) and 5 (20%) of the participants. Candida lusitaniae, C. kruzei, C. membranaefaciens, C. galbrata, were also identified as parts of the mixed isolates in 4 (16%), 3 (12%), 2 (8%) and 2 (8%) respectively, in the participants. Candida Kefyr was identified in only 1 (2%) of the 25 participants (Table 4). The total number of control participants recruited for this study was eighty, thirty-six (45%) of them were females and forty-four (55%) were male participants. All the stool samples collected from the participants in the control group yielded growth of normal flora (bacteria and fungi) of the gastrointestinal tract (GIT). Their stool microscopy revealed very scanty or no yeast cells (Table 5).
Table 4: Distribution of Mixed Isolates of Candida species by Disease Conditions.
Chi-square= 72.2, p-value=0.000
*Values too small for statistics
KEY
PUD – Peptic Ulcer Disease
DD - Diarrhoea Disease
GIT BD – Gastrointestinal Bleeding
IMM SU – Immune suppression
HIV – Human Immune Virus
CHEMO – Chemotherapy
Table 5: Frequency table for control participants.
KEY
Nil= No pathogen was isolated from the control group.
Discussion
In this study, the intestinal stool culture yielded pure fungi isolates of Candida species with an overall prevalence of 26.7% (40 isolates found among 150 patients). This is considerably higher than other findings such as 21.5% in Cameroon [22] but lower than 41.1% in Mexico (2) and 75.47% in Poland in 2015 [23] The prevalence in this study was also higher than the overall prevalence in sub-Saharan Africa, which was estimated to be 23.4% [7]. [8] in India, however, reported a lower prevalence (9.7%). Previous authors have noted a wide variation in the prevalence of Candidiasis depending on region, population surveyed and even the research methods used [24]. However, most studies used similar culturebased methods, the quantification threshold for colony-forming units of the culture colonies have diverse threshold. We adopted a threshold range of 105 colony forming unit (CFU) considered significant for growth, whereas in Poland study according to [23] a wider range (103 CFU – 106 CFU) was considered. This may have resulted for the significantly higher prevalence in their study.
In the current study, female patients had a higher prevalence rate compared to male patients (18.7% vs 14.7%; p = 0.0439). A study conducted in Vienna, Austria, on burn patients found a female predisposition in systemic and related candidiasis [25]. The predilection of gastrointestinal Candidiasis in female patients is poorly understood and there is no proper explanation for the predisposition [26]. In species distribution of Candida isolates, C. albicans accounted for the majority (16.7%) of isolates in this study. This is consistent with other studies that identified it as the most common isolate from clinical materials, [27,28] and could be attributed to the fact that C. albicans is highly adapted to the human mucosal surfaces and possesses virulence factors such as protease production and biofilm formation [24,29]. These enhance its chances of survival in the gastrointestinal tract. Omrani et al. [30] in their review of African studies also found a predominance of C. albicans.
Furthermore, the remaining 25 (16.7%) were reported as mixed culture of Candida spp. C. albicans and C. dubliniensis were found as a pair in seven stool samples from participants with cases of peptic ulcer disease 1(14.3%), gastrointestinal bleeding 4(57.1)), immune suppression 2(28.6%); others occurred in combinations of threes and fours of different species. This is consistent with the studies in Poland [23] and India [31]. Non-albicans species in general accounted for a larger number of isolates in studies by Banerjee et al. [31] in India, as well as in Brazil and Chile [32]. The most frequent pathogens were Candida spp. (14.9%). The pathogenicity of fungal infection in this study was established in 17% of the studied population and C. albicans was confirmed to be the pathogenic fungus causing infection (gastroenteritis) in them. A sub group of participants with HIV infection in this study had a prevalence of 85% in pure culture and 15% in mixed culture respectively. The high prevalence in this subgroup could be as a result of the limited number of participants with HIV in this study. Generally, the immune system in HIV cases are suppressed hence the chances of C. albicans attaining pathogenic state were high [29,32].
In other disease conditions, the pervalence of C. albicans were significant, ranging from 25% in diarrhoea disease to 100% in the only participant with cancer and on chemotherapy included in this study. Co-existing infections is comoon when immunity is reduced and as a result, the prevalence obtained in the subgroups (HIV and Immune Supression) in this study were 85% and 43% respectively which were obtained for pure Candida albicans isolates. This was in line with the study obtained in Saudi Arabia with the overall prevalence of intestinal fungal infections of 58.7% in transplant recipients [32].
The most prevalent fungus in that study was Candida spp., which was seen in 22% of the study patients and 24.4% of the control group. Co-existing infection with two or more fungi was seen in 14.8% and 3.4% in the case and control groups. Interestingly, there was no significant difference (p-value= 0.05) in the prevalence of infection by a single organism between the two groups [33,34]. However, co-existing infection with two or more species was more prevalent in transplant recipients. There was a significant difference in the prevalence between the case and control groups in this study and this was seen in all the disease conditions especially in cases of GIT bleeding and immune suppression. Stool samples from all the participants used as control, yielded growth of gastrointestinal normal flora in this study. This may be as a result of other host factors like irritable bowel syndrome etc. that must be present for Candida to exist as a pathogen [34,35].
The pair of C. albicans and C. dubliniensis was isolated in seven stool samples from participants with cases of Peptic Ulcer Disease (1), Gastrointestinal Bleeding (4) and Immune Suppression (2) in this study; this may be attributed to the closely related genetic materials shared by these species which may have helped to increase their level of invasiveness in strain related virulence factors [36-38] though further study may be required to establish this fact. Other stool samples also yielded pairs (5) but in combination of various species. Ten (10) stool samples occurred as mixed cultures of three different species and only three of the participant’s stool reported a combination of four different species (Table 4). Generally, fungal infection (gastroenteritis) was reported more in this study than bacteria (gastroenteritis) which indicates a shift from the normal cases. The reason for this shift may be attributed to Candida overgrowth which was supported by the study of Roberth et al. [15] and is in line with results from this work.
Conclusion
Prevalence of gastrointestinal candidiasis was relatively high among the participants of this study. The infection is associated with female sex having higher burden of candida isolates and other GIT microbes due to poor glycaemic control and previous use of antifungals, antibiotics and corticosteroids. Nystatin can be a drug of choice in the treatment of gastrointestinal candidiasis, if the suspension or tablet formulations can be made available. This therefore calls for better hygiene practice through educative advocacy. Effective management of opportunistic mycoses depends on identification of the specific organism causing the infection otherwise, a systemic infection of this type can be fatal.
References
- Lisboa C, Ferreira A, Resende C, Rodrigues AG (2009) Infectious balanoposthitis: management, clinical and laboratory features. International Journal of Dermatology 48(2): 121-124.
- Martinez RFF, Jaimes Aveldanez A, Hernandez Perez F, Arenas R, Miguel GFS (2013) Oral Candida spp carriers: Its prevalence in patients with type 2 diabetes mellitus. An Bras Dermatol 88(2): 222-225.
- Achkar JM, Fries BC (2010) Candida infections of the genitourinary tract. Clinical Microbiology Reviews 23(2): 253-273.
- Eckert LO, Hawes SE, Stevens CE, Koutsky LA, Eschenbach DA, et al. (1998) Vulvovaginal candidiasis: clinical manifestations, risk factors, management algorithm. Obstetrics & Gynecology 92(5): 757-765.
- Marin MG, King R, Fameni SS, Dennerstein GJ (2000) Adverse behavioral and sexual factors in chronic vulvar disease. The American Journal of Obstetrics and Gynecology 183(1): 34-38.
- Alvarez Lerma F, Nolla Salas J, Leon C, Jorda R, Carrasco N, et al. (2003) Candiduria in critically ill patients admitted to intensive care medical units. Intensive Care Medicine 29(7): 1069-1076.
- Jafar N, Edriss H, Nugent K (2016) The effect of short-term hyperglycemia on the innate immune system. Am J Med Sci 351(2): 201-211.
- Gurleen K, Savio R (2016) Prevalence of Candida in Diarrhoeal Stools. J Dent Medi Sci 15(4): 47-49.
- Kauffman CA, Vazquez JA, Sobel JD, HA Gallis, DS McKinsey, et al. (2000) Prospective multicenter surveillance study of funguria in hospitalized patients. The National Institute for Allergy and Infectious Diseases (NIAID) Mycoses Study Group. Clinical Infectious Diseases30(1): 14-18.
- Xu J, Boyd CM, Livingston E, Meyer W, Madden JF, et al. (1999) Species and genotypic diversities and similarities of pathogenic yeasts colonizing women. Journal of Clinical Microbiology 37(12): 3835-3843.
- Abu Elteen KH (2001) Increased incidence of vulvovaginal candidiasis caused by Candida glabrata in Jordan. Japanese Journal of Infectious Diseases 54(3): 103-107.
- Chong PP, Lee YL, Tan BC, Ng KP (2003) Genetic relatedness of Candida strains isolated from women with vaginal candidiasis in Malaysia. Journal of Medical Microbiology 52(8): 657-666.
- Pittet D, Monod M, Suter PM, Frenk E, Auckenthaler R (1994) Candida colonization and subsequent infections in critically ill sursgical patients. Ann Surg 220(6): 751-758.
- Saunders (2008) Saunders Comprehensive Veterinary Dictionary, 3rd (edn.). 49(9): 906.
- Roberth K, Gunter J Krejs, Wenisch C, Reisnger EC (2003) Elevated Fecal Candida Counts in Patients with Antibiotic-Associated Diarrhea (AAD). Clinical Diagnostic Laboratory Immunology10(1); 167-168.
- Edwards S (1996) Balanitis and balanoposthitis: a review. Genitourinary Medicine 72(3): 155-159.
- Christian Perez J, Alexander D Johnson, William E Goldman (2013) Regulatory Circuits That Enable Proliferation of the Fungus Candida albicansin a Mammalian Host. Public Library of Science Biology (PLoS) Pathogens 9(12): e1003780.
- Cheesbrough M (2006) Microbiological tests. District Laboratory Practice in Tropical Countries 2: 442.
- Karasu Yalcin S, Senses Ergul S, Yesim Ozbas Z (2012) Identification and Enzymatic characterization of the Yeasts isolated from Erzincan tulum cheese. Mljekarstvo 62(1): 53-61.
- Fotedar R, Al Hedaithy SSA (2003) Candida dubliniensis at a University Hospital in Saudi Arabia. Journal of Clinical Microbiology 41(5): 1907-1911.
- Firooz B, Smith GA, Krause WG (1975) Germ Tube and Chlamydospore Formation by Candida albicans on a New Medium. Journal of Clinical Microbiology 2(4): 345-348.
- Bissong M, Azodo CC, Agbor MA, Nkuo akenji T, Fon P (2015) Oral health status of diabetes mellitus patients in Southwest Cameroon. Odontostomatol Trop 38(150): 49-57.
- Kowalewska B, Kawko M, Zorena K, Myśliwiec M (2014) Yeast-like fungi in the gastrointestinal tract in children and adolescents with Diabetes type 1. Pediatr Endocrinol Diabetes Metab 20(4): 170-177.
- Anaissie EJ, McGinnis RM, Pfaller AM (2009) Clinical mycology. 2nd St Louis, MO: Elsevier.
- Rubaihayo J, Tumwessigye NM, Konde Lule J, Wamani H, Nakku Joloba E, et al. (2016) Frequency and distribution patterns of opportunistic infections associated with HIV/AIDS in Uganda. BMC Res Notes 9(1): 501.
- Loster JE, Wieczorek A, Loster BW (2016) Correlation between age and Candida species infections of complete denture wearers: A retrospective analysis. Clin Interv Aging 2016(11): 1707-1714.
- Flevari A, Theodorakopoulou M, Velegraki A, Armaganidis A, Dimopoulos G (2013) Treatment of invasive candidiasis in the elderly: A review. Clin Interv Aging 8: 1199-1208.
- Sanguinetti M, Posteraro B, Lass Florl C (2015) Antifungal drug resistance among Candida species: Mechanisms and clinical impact. Mycoses 58(Suppl 2): 2-13.
- Arslan S, Koç AN, Sekerci AE, Tanriverdi F, Sav H, et al. (2016) Genotypes and virulence fac+tors of Candida species isolated from oral cavities of patients with type 2 diabetes mellitus. Turk J Med Sci 46(1): 18-27.
- Omrani SA, Pecen L, Rajek P, Raghubir N, Zigmond J (2014) Prevalence of invasive and superficial Candida infections in Africa and Middle East; a systematic review and meta-analysis. 114th Meeting of the American Society for Microbiology (ASM), Boston, MA, pp. 17-20.
- Banerjee P, Kaur R, Uppal B (2013) Study of fungal isolates in patients with chronic diarrhea at a tertiary care hospital in North India. J Mycol Med 23(1): 21-26.
- Sardi OCJ, Scorzoni L, Bernardi T, Fusco-Almeida MA, Mendes GSJM (2013) Candida species: Current epidemiology, pathogenicity, biofilm formation, natural antifungal products and new therapeutic options. J Med Microbiol 62(1): 10-24.
- Omalu ICJ, Yako AB, Duhlinska DD, Anyanwu GI, Pam VA, et al. (2005) First detection of intestinal microsporidia in Northern Nigeria. Online J Health Allied Science 4(3): 3-4.
- Naeini AE, Sharifi M, Shahidi S, Taheri S, Seirafian S, et al. (2012) Intestinal fungal and Parasitic infection in Kidney transplant recipients: A multi-center study. Saudi J Kidney Dis Transplant 23(4): 677-683.
- Ibrahim A, Ikeh E, Malu A, Okeke E. Damen J (2007) Intestinal Parasitosis in Human Immunodeficiency Virus (HIV) Infected Adults with Chronic Diarrhoea at Jos University Teaching Hospital, Nigeria. The Internet Journal of Parasitic Diseases 2(1).
- Julian R Naglik, Stephen J Challacombe, Bernhard Hube (2003) Candida albicans Secreted Aspartyl Proteinases in Virulence and Pathogenesis. Microbiology and Molecular Biology Review 67(3): 400-428.
- Karkowska Kuleta J, Rapala Kozik M, Kozik A (2009) Fungi Pathogenic to Humans: Molecular Bases of Virulence of Candida albicans, Cryptococcus neoformans and Aspergillus fumigatus. Acta Biochemical Polonica 56(2): 211-224.
- Lopes da Rosa J, Kaufman PD (2013) Chromatin-Mediated Candida albicans Virulence. Biochem Biophys Acta 1819(3-4): 349-355.

 Research Article
Research Article