Introduction
In recent years several new single-use bone marrow core biopsy needles have been introduced for obtaining bone marrow biopsy samples for histologic evaluation from patients with hematological and non-hematological disorders [1-5]. However, obtaining samples of bone marrow during autopsy still retains traditional methodology and involves an old-fashioned way of chiseling or slicing vertebral bodies with a saw. The process is cumbersome and most importantly it often does not provide samples deeply placed for examination where bone marrow abnormalities may sometimes be found. Also, when a saw is used to obtain bone marrow samples at postmortem, the sawing process can damage the tissue adjacent to the cut and result in a sampling attributable artifact. To overcome the inadequacy of the conventional methods of obtaining bone marrow samples for histological evaluation at post-mortem and to improve diagnostic efficacy, we have used a newly introduced core retention bone marrow biopsy needle [5]. Unlike conventional bone marrow biopsy needles this new needle has a unique internal core retention device and, as a result, no additional components or parts, such as the sleeve [1] or marrow acquisition cradle [2] required by other needles to capture the biopsy specimen is necessary and the needle can be withdrawn with a straight pull. No rocking, sculling, or gyratory movements, or change in the direction of the tip of the needle to capture the biopsy specimen is necessary. As a result, the needles are not bent or damaged and can be used over and over again after adequate cleaning and disinfection. As the needles are supplied non-sterile, they are less expensive and can be used as a single-use (disposable) needle to avoid contamination with HIV or hepatitis.
Materials and Methods
The Instrument* (Figure 1) Consists of Five Parts
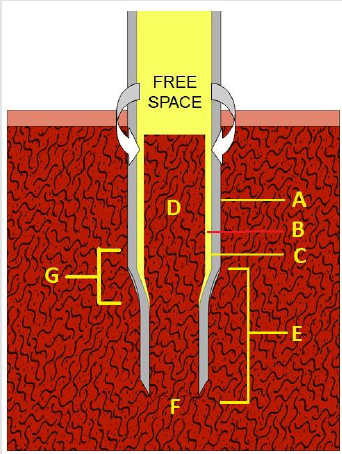
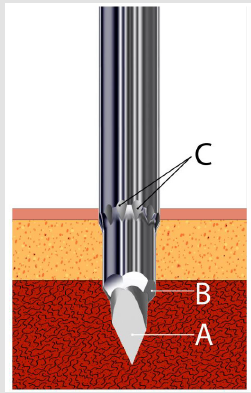
a) The needle (Figure 1A) which has an overall length of 100 mm, an uniform external diameter of 3.25 mm, and a constant internal diameter of 2.5 mm except for the 3.5 mm distal portion where it is narrowed and has multiple surface serrations/ flutes (Figure 1 inset). The terminus of the needle has six sharp cutting facets (Figure 1 inset). The internal diameter of the needle’s distal portion is less than the overall internal diameter of the needle and begins with a short, slanted step of 0.2 mm. This specially designed distal portion (Figure 1 inset) cuts the trabecular connections of bone which might keep the biopsy specimen anchored to its base and also holds on to the biopsy sample so that it does not slip out of the needle during the process of its withdrawal. The larger internal diameter of the needle at its proximal segment provides free space (Figure 2B) within the interior of the instrument thus avoiding crushing and compression of the tissue as well as plugging the lumen of the needle. It also facilitates easy delivery of the core sample through the proximal end of the needle. The introduction of the needle through the dense cortical bone can be mechanically challenging. To assist in easy, smooth entry multiple surface serrations/flutes (Figure 3C) are provided at the junction of the distal narrower portion with the adjoining wider proximal portion of the needle. These surface serrations/flutes provide a means of drilling as the needle is being advanced through the dense cortical bone while rotating the needle by clockwise and counter clockwise rotary motions. The proximal end of the needle is fitted with a large plastic T- bar handle (Figure 1B) for a firm grip.
Figure 1:The needle (A), plastic T-bar handle (B), stilette (C) with its plastic head (D), white plastic cap (E), insertion aid (F), and the pusher rod (G).
b) The stilette (Figure 1C) is a solid steel shaft of 2.0 mm in diameter. It ends with a 3.0 mm long three-faceted, sharply pointed tip (Figure 3A) which projects beyond the cutting edge of the needle. It also provides a means of easy penetration of the cortical bone. The proximal end of the stilette is fitted with a round knurled plastic knob (head) (Figure 1D) which fits into the proximal (top) end of the needle.
c) The large white plastic cap (Figure 1E) fits over and covers the top of the plastic T-bar handle (Figure 1B). It also fits snugly in the palm of the operator’s hand and fosters comfortable manipulation during the biopsy procedure.
d) The insertion aid (Figure 1F) is a tubular segment of plastic 30 mm long and 5 mm wide except at the end that fits the mouth of the needle where it is flat and 10 mm in diameter. It is centrally hollowed out to receive and permit the passage of the pusher during the removal of the biopsy specimen through the proximal end of the needle.
e) The pusher (Figure 1G) is a solid steel rod of 2.0 mm in diameter attached to a knurled, flat plastic disk. The pusher rod is used to expel the biopsy specimen from within the lumen of the biopsy needle with the help of the insertion aid which guides the pusher rod into the mouth needle.
*The non-sterile (as well as the sterile) needles are available from Cardinal Health. Cardinalhealth.com/Core Retention Bone Marrow Biopsy Needle.
Figure 2:Schematic representation of the biopsy technique. (A) the needle, (B) free space between the marrow core (D) and the internal wall of the needle (C). (E) Specially tooled distal cutting end of the needle which cuts the trabecular connections of the core at its base (F) and holds the core sample during the process of its extraction so that it does not slip out of the needle. (G) shows the area of the distal cutting end of the needle where it joins the main body of the needle which is wider.
Figure 3:Schematic representation of the distal penetrating and cutting end of the needle showing the elongated, trocar pointed stilette (A), sharp cutting facets (B) and the external surface serrations/flutes (C).
Operative Procedure (Figure 4)
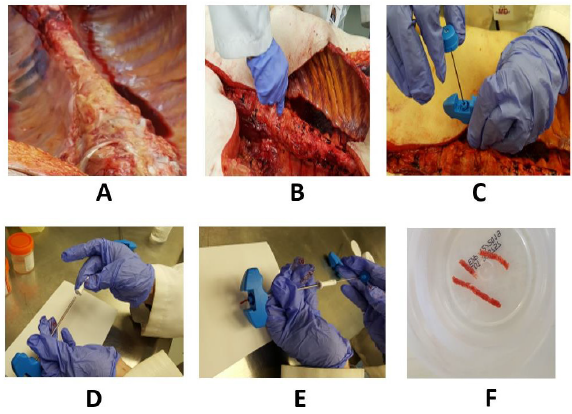
During post-mortem, once the viscera are removed the bodies of the vertebral column are exposed (Figure 4A). The biopsy needle with the stilette and large white plastic cap in place is advanced slowly pointing towards the spinous process of a lumbar vertebra (Figure 4B). The cortical bone is then penetrated by gentle rotary motions of the needle. Once the cortex is penetrated the stilette is removed by twisting the knob of the stilette anti-clockwise with one hand while holding the needle in place with the other (Figure 4C). The biopsy needle is then advanced with slow, steady, and controlled clockwise-counter-clockwise rotary motions until an adequate depth is reached (about 20-25 mm). The biopsy needle is then rotated completely several times about its long axis to sever all the trabecular connections at its base and sever the core sample from the surrounding spongy bone. It is then slowly withdrawn with a straight pull with gentle alternating rotary motions. No rocking, sculling, or gyratory movements or changes in the direction of the tip of the needle are necessary. Once the needle is completely withdrawn from the vertebral body the insertion aid is attached to the distal cutting end (mouth) of the needle (Figure 4D). The biopsy specimen is then pushed out of the needle with the pusher rod which is introduced through the opposite end of the insertion aid (Figure 4E).
Figure 4:(A) demonstrates the vertebral bodies following removal of the viscera. (B) The needle with the stilette in place is inserted into the bone. Once the cortex is penetrated the stilette is withdrawn (C). (D) The photograph shows the attachment of the insertion aid to the distal cutting end (mouth) of the needle. (E) The photograph shows the introduction of the pusher through the insertion aid to push the core sample out of the needle. (F) Shows several bone marrow core samples in a fixative. Note the long and uniform core of marrow tissue obtained with this needle.
Results
The needle has been extensively tested on multiple cadavers obtaining at least 6-8 samples from each cadaver. The sites used for obtaining the core samples were slightly to the right or slightly to the left of the anterior midline of thoracic #12 and lumber # 1-5 vertebrae. An excellent sample was obtained with each attempt (Figure 4F). Bony trabeculae were clean-cut and crush artifact was almost negligible even at the edges of the biopsy specimen. Detailed histologic analysis of bone marrow specimens obtained using this new needle and other conventional bone marrow biopsy needles, including the Jamshidi [2] and the on control needle [4] since this is the downstream intent of any bone marrow core biopsy needle has already been reported [6].
Discussion
Since the introduction of the bone marrow biopsy procedure by Ghedini in 1900 [7], bone marrow core biopsy has become a routine procedure for diagnostic and prognostic evaluation of patients with hematological and non-hematological disorders [8-12]. Although the method and instrumentation of procuring such samples from the human body have become easier due to the sophistication and development of new and improved needles, tissue sampling of bone and bone marrow at post-mortem has not changed and remains crude and cumbersome. Using the conventional method of chiseling or sawing a bone segment from the vertebral bodies is still considered the norm. Although the above approach generally yields a rather large but relatively superficial area of bone and marrow this method is likely to miss a potentially desired tissue mass that is residing in the deeper, distal regions of marrow mass.
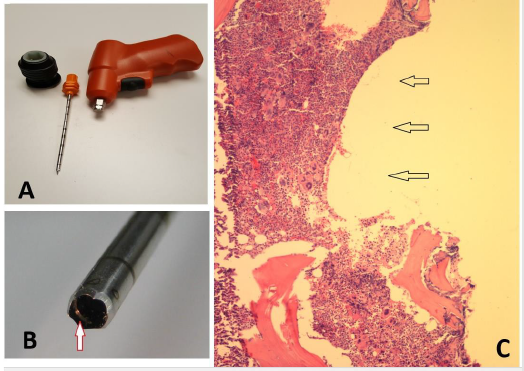
In the United States, however, the technique of obtaining core samples at post-mortem is not new, as there are several types of needles commercially available, such as Core-lok [1] or Jamshidi [2] needles. These needles are normally collected from the hematology department following bone marrow procedure, washed, cleaned, and subsequently used by the pathologists to obtain marrow and core samples at post mortem. In practice, the samples are taken from iliac crests, though vertebral bodies are not uncommon. Unfortunately, none of the existing bone marrow biopsy needles except the one described here has internal core retention devices. As a result, to capture a core sample, the newer needles (i.e., Core-lock, Jamshidi) require additional components such as the sleeve (a tweezer-like component) or a drain-spade-shovel like component called marrow acquisition cradle. They also require multiple steps and several maneuvers to complete the procedure. This often results in fragmentation of the core sample. Unlike the needle described in this article, none of the commercially available needles are sold in a reusable (non-sterile) form. There are also powered devices such as the On Control needle [4], which makes bone cutting easy. But the high-speed drilling and the associated sickle-shaped hook present near the distal cutting end of the needle (Figure 5B) which is used to capture the biopsy specimen causes considerable damage to the biopsy, particularly near the edge (5C) of the biopsy specimen as the hook passes over the core and excises the bone and marrow. On Control needles are also very expensive and are not sold in disposable forms.
Core loss has been a real problem using the conventional bone marrow biopsy needles, such as the older Jamshidi or similar type of needles, which do not have a trapping device to capture a core sample, thus requiring a second or third attempt to secure a core sample. To overcome this problem and to secure a biopsy specimen, these needles do require rocking, sculling or gyratory movements or change in the direction of the tip of the needle to capture a core sample. As a result, the needles are often bent or damaged (Figure 6) and cannot be used over and over again.
Unlike the conventional bone marrow biopsy needles the needle described here (5) does not require any rocking, sculling or gyratory movements or change in the direction of the tip of the needle to capture the biopsy specimen. As a result, the needles are not bent or damaged and can be used over and over again. This needle has an internal core retention design and as a result, no additional components or parts such as a sleeve (1) or marrow acquisition cradle (2) to capture the biopsy specimen is necessary and the needle can be withdrawn with a straight pull. When additional components or parts are used to capture the biopsy specimen, they may not only cause fragmentation of the biopsy specimen but also can cause considerable damage to the biopsy and significant crush artifact particularly near the edge of the biopsy specimen. This proposed needle does not require any additional components and avoids these limitations.
In a series of histopathological bone marrow investigations in forensic medicine 35% of the cases (n=78), abnormal histopathologic changes were identified [13]. Forty of them were neoplastic, predominantly hematological diseases which were unknown during the patient’s lifetime. Referring to these findings the authors suggested that extraction of post-mortem bone marrow specimens for histopathological investigation should become an essential procedure for both the forensic and clinical pathologists’ investigations [13].
The use of the needle described herein would make the pathologist’s efforts simpler and more productive. It can be used to obtain multiple samples from different bony regions. Because the needle is supplied in a non-sterile state it is modest in cost. It can be used multiple times provided the cutting facets are not damaged. We believe the availability of this needle will give pathologists further impetus to sample bone marrow at post-mortem and perhaps resolve questions that would otherwise not be recognized or addressed.
Conflict of Interest
None.
References
- Core-LockTM bone marrow biopsy system by Worldwide Medical technologies/Argon Medical
- Jamshidi CrownTM bone marrow biopsy needle and marrow acquisition cradle by Care Fusion/BD.
- Goldenberg AS, Tiesinga JJ (2001) Clinical experience with a new specimen capturing bone marrow biopsy needle. Am J Hematol 68: 189-193.
- Vidacare Corporation, 4350 Lockhill Selma Road, Suite 150, Shavano Park, TX 78249, USA.
- Islam A (2019) A New Single-Use Bone Marrow Biopsy Needle with Core Retention Design. Biomed J Sci & Tech Res 19(3).
- Islam A (2017) Bone Marrow Solid Core Biopsy Needle: A Critical Assessment of the Utility, Benefits and Limitations of the Instruments Employed in Current Day Haematology and Oncology. J Clin Pathol:1-8.
- Ghedini G (1908) Studi sulla patologia del midello osseo umano vivente. I. Punctura explorative tecnica. Clin Med Ital 47: 724-727.
- McFarland W, Dameshek W (1958) Biopsy of bone marrow with the Vim-Silverman needle. JAMA 166: 1464.
- Ellman L (1976) Bone marrow biopsy in the evaluation of lymphoma, carcinoma and granulomatous disorders. Am J Med 60: 1.
- Burke JS (1978) The value of the bone marrow biopsy in the diagnosis of hairy cell leukaemia. Am J Clin Pathol 70: 876.
- Brunning RD, Bloomfield CD, McKenna RW, Peterson L (1975) Bilateral trephine bone marrow biopsies in lymphoma and other neoplastic diseases. Ann Int Med 82: 365.
- Jamshidi K, Swaim WR (1971) Bone marrow biopsy with unaltered architecture: a new biopsy device. J lab Clin Med 77: 335.
- Roll P, Beham A, Beham-Schmid Ch (2008) Post-mortem Histopathological Investigations of the Bone Marrow in Forensic Medicine: An Important Issue for Both the Forensic and Clinical Pathologist.

 Research Article
Research Article