Abstract
Aim: In this study, we aimed to investigate cardiac involvement in patients diagnosed with the new Coronavirus Disease 2019 (COVID-19).
Methods: Patients diagnosed with COVID-19 pneumonia in the emergency department were included in this retrospective study. Patients diagnosed with COVID-19 pneumonia were included in Group 1 and healthy individuals (as control group) were included in Group 2.CCT images were transferred to the Intrasense Myrian® software. Total cardiac volume (TCV) was measured in three-dimensional images. Obtained parameters were compared between groups
Results: Total cardiac volume was 525.35±29.96 cm3 (min:244cm3 - max:1545cm3) in Group 1 and 369.64±10.41cm3 (min:229cm3 - max:522cm3) in Group 2 (P=0.000). In Group 1 patients, levels of D-dimer, C-reactive protein (CRP) and high sensitivity troponin I (hs-TnI) were 391.00 (6.00-6450) μg/L, 3.50 (0.10-30.40) mg/dL and 4.41 (1.00- 3627.00) ng/mL, respectively, whereas those in Group 2 were 112.50 (3.00-767.00) μg/L, 0.20 (0.10-19.30) mg/dL and 2.50 (2.30-36.02) ng/mL, respectively (P<0.05).
Conclusion: Total cardiac volume increases in patients that develop pneumonia due to COVID-19 infection. D-dimer, CRP and hs-TnI levels also increase with TCV.
Keywords: COVID-19; Chest Computed Tomography; Total Cardiac Volume; C-Reactive Protein; D-Dimer; Highly Sensitive Troponin I
Introduction
The Coronavirus Disease 2019 (COVID-19) is an acute disease that is transmitted mainly via droplets and primarily affects the respiratory system. Symptoms in most patients are mild, however the disease can lead to acute respiratory distress syndrome, shock and multiple organ failure in some patients. After the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) enters the body, it becomes active by binding to ACE2 receptors on the cell membrane. The ACE2 receptor is expressed the most in type I and type II pneumocytes and constitutes the main route of entry of the virus. In addition to pneumocytes, ACE2 receptors are found on heart, intestine, kidney, epithelium and vascular endothelium cells [1,2].
As a part of the systemic inflammatory response in severe COVID-19 infections, damage may arise in multiple organs, such as heart, kidney, brain and liver. Research has found that proinflammatory cytokine levels increase in patients with severe COVID-19 infections, which has been regarded as the cause of such organ damage. Furthermore, it has been shown that patients with histories of hypertension (HT), coronary artery disease, heart failure and other cardiovascular diseases are more vulnerable to COVID-19 infections and that disease progression is more severe in these patients. Moreover, some studies have indicated that SARSCoV- 2 causes myocardial cell damage directly [1,4].
In COVID-19 cases, laboratory tests may indicate hypoxemia, anemia, lymphopenia, thrombocytopenia, abnormal kidney and liver function, and high levels of creatine kinase, lactate dehydrogenase and D-dimer. Increased levels of inflammatory markers such as serum ferritin and C-reactive protein (CRP) are observed. COVID-19 patients with cardiac involvement may have high levels of troponin and brain natriuretic peptide (BNP). Therefore, these parameters should be investigated in cases of suspected cardiac involvement [5-7].
Chest X-ray and chest computed tomography (CCT) scans are used for imaging COVID-19 pneumonia. The most frequent findings on CCT scans are ground-glass opacities with typical peripheral distribution in lungs [8]. In addition to laboratory tests, transthoracic echocardiography (TTE), transoesophageal echocardiography (TEE), point-of-care ultrasonography (POCUS) and magnetic resonance imaging (MRI) are used for identifying cardiac involvement. However, especially TTE and MRI involve physical contact between patients and healthcare personnel, and contact times are long. Therefore, unnecessary imaging should be avoided, as these cause undesired exposure of healthcare personnel, which may augment disease burden. Alternative imaging methods, such as TTE or CCT, are recommended for investigating cardiac involvement [9-12]. In cases of various diseases and in healthy individuals, volumes of organs and/or masses are routinely estimated from computed tomography (CT) scans using software tools. Volume measurements can also be made by utilizing other imaging methods, such as MRI and ultrasonography [13-16]. In this study, we aimed to investigate cardiac involvement and changes in patients diagnosed with COVID-19.
Methods
This retrospective study was conducted after the approval of Alanya Alaaddin Keykubat University Faculty of Medicine Ethics Committee (2020/19-31). Patients who visited the emergency department between 01.05.2020 and 15.08.2020 with complaints of cough, fever and shortness of breath, who were diagnosed with COVID-19, whose CCT scans were taken and who were 18 or older were included the study. Diagnoses of COVID-19 of the patients were confirmed with reverse transcriptase polymerase chain reaction (PCR) tests, which were performed on nasopharyngeal swab samples. The control group was formed from patients that were referred to CCT imaging from the cardiovascular surgery outpatient clinic for any reason, whose CCT scans were interpreted as normal. Patient information was obtained from hospital’s automation system and patient files. A standard data collection form was created for the study. File number, age (in years), gender, initial complaints, examination findings, laboratory results, CCT reports, ward of hospital stay and clinical outcomes of the patients were recorded on these forms. Formal reports in the hospital automation system were used for interpretation of the CCT images of the patients that were included the study. Patients diagnosed with COVID-19 pneumonia were included in Group 1, while patients in the control group were included in Group 2. Patients whose CCT scans revealed findings other than viral pneumonia (cist, mass, edema, bacterial pneumonia, pneumothorax, hydrothorax, pleural effusion, pericardial effusion, etc.) were excluded from the study. Also, patients whose CCT scans could not be taken or were taken at another facility, those that were not tested with PCR, those younger than 18, and those with unavailable information were excluded from the study.
Biochemical Analysis
D-dimer levels were measured using an ACL TOP 500 (Instrumentation Laboratory, Munich, Germany). CRP levels were measured using an Abbott Architect C800. Troponin I (hs-TnI) were measured using a Siemens Advia Centaur XPT. Hemograms were performed using a Sysmex XN100. The reference ranges reported in healthy adults were as follows: 0-0.5 mg/dL for CRP, 0-240μg/ dL for D-dimer, 1.26-3.35 10^3/μL for lymphocyte count and 0-46.47ng/mL for high sensitivity troponin I (hs-TnI).
Chest Computed Tomography Imaging
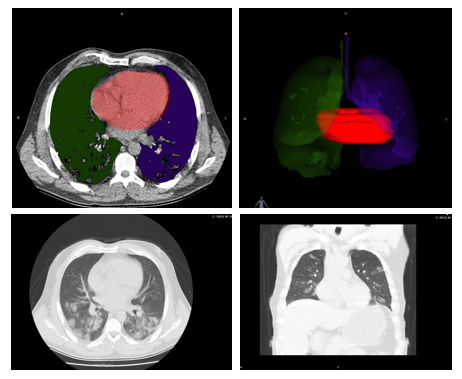
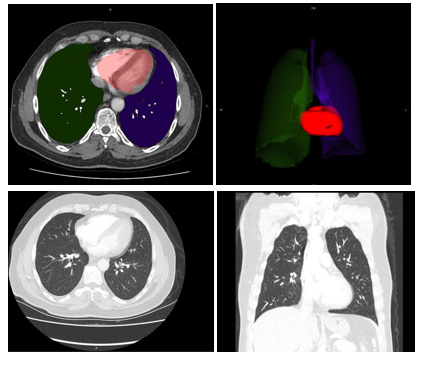
CCT scans were obtained from hospital’s automation system. A multi-slice computed tomography scanner (TOSHIBA Alexion 16 slice) was used for CCT imaging in the emergency service. CCT images on the hospital automation system were transferred to the Intrasense Myrian® (Myrian; Intrasense, Montpellier, France) software and 3D models of the data were recreated. Anatomical features of patients were defined on this application. Cardiac sections were colored to form 3D images and Total Cardiac Volume (TCV) were measured afterwards, which were carried out by an academic of anatomy. The resulting data were recorded on the data collection form (Figures 1&2).
Statistical Analysis
Statistical analysis: In this study, normal distribution for continuous data was tested with skewness-kurtosis, histogram, Q-Q graphs, and standard deviation/mean and Kolmogorov Smirnov tests. Normally distributed continuous data were shown as mean ± standard error (MEAN±SEM), and t-test was used comparison of two independent groups. The data that were not distributed normally were shown as the median (minimum-maximum) and the Mann Whitney U test was applied in independent pairwise comparisons. Yates chi-square test was used in the frequency analysis of noncontinuous categorical variables. By utilizing receiver operating characteristic (ROC) analysis, the area under the curve (AUC) with lower and upper bound was used to calculate the cut-off value, specificity, sensitivity. In order to make COVID positive predictive analysis with serum d-dimer, CRP, LYM, troponin values, the cut-off values obtained in the ROC analysis were categorized and the variables were categorized, and multiple binary regression analysis was performed. Nagelkerke’s R2 determination coefficient was used to evaluate the predictive ability of the model. In statistical analysis, α = 0.05 was accepted and statistically significant for p <α. In this study, statistical analysis was applied with IBM SPSS 23.00 package program.
Figure 1: A 55-year-old male patient was admitted to the emergency department with complaints of cough and fever. The patient’s reverse transcriptase polymerase chain reaction (PCR) test was positive. He was hospitalized in the intensive care unit with the diagnosis of COVID-19 pneumonia. Total heart volume measured using Intrasense Myrian® software was calculated as 460cm3 (Group 1).
Figure 2: A 57-year-old male patient visited the cardiovascular surgery clinic with a complaint of chest pain. Total heart volume measured using Intrasense Myrian® software was calculated as 305 cm3 (Group 2)
Results
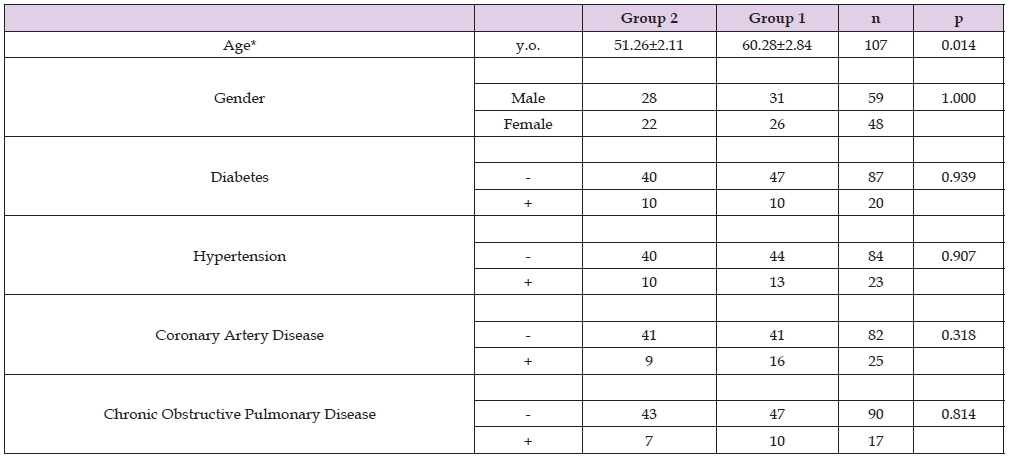
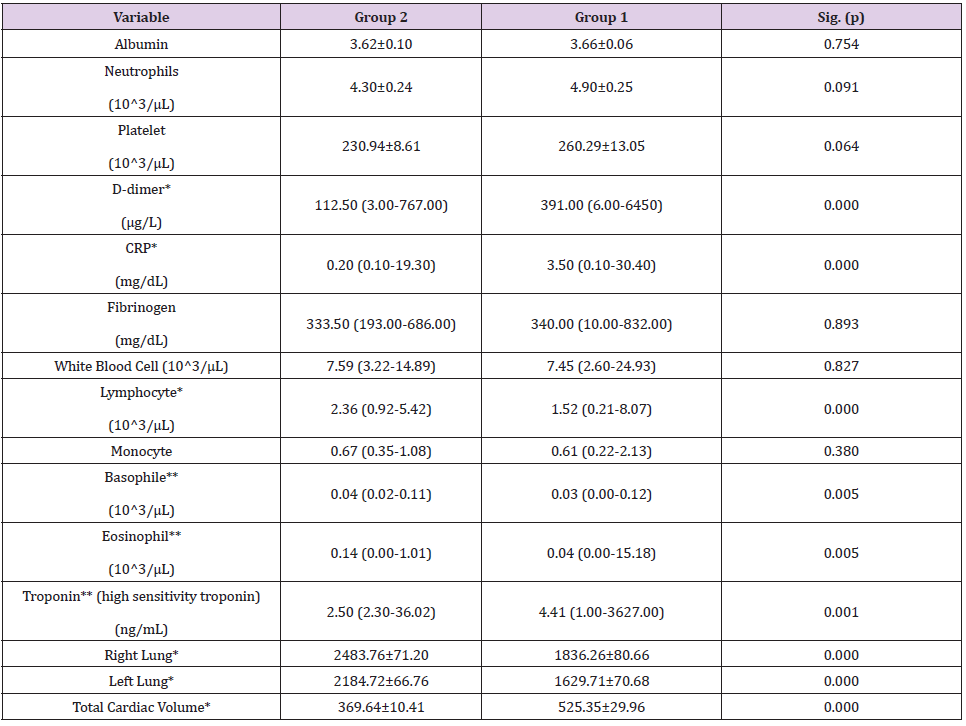
A total of 57 patients were included in Group 1. The control group (group 2) consisted of 50 healthy individuals. Patients in Group 1 and 2 were not significantly different in terms of gender, or concomitant medical conditions (Table 1). However, the average age of patients with COVID-19 was found as than healthy people. Group 1 patients had higher levels of TCV, hs-TnI, D-dimer and CRP, and lower lymphocyte counts compared to Group 2 patients. In contrast lower lung volumes were detected in patient group (group 1) when compared with healthy individuals (group 2). We found that TCV 42% higher in Group when compared with Group 2. The comparisons of parameters between two groups are summarized in Table 2.
Table 1: Characteristics of Study Patients.
* The difference between groups is statistically significant at 95% confidence interval (p<0.05).
Table 2: Comparison of Laboratory Results and Chest Computed Tomography Calculations.
Parametric data’s were shown as Mean ± Standard error (SEM) and Student T test was used for comparisons of two independent groups.
Nonparametric data’s were shown as Median (Minimum-Maximum) and Man-Whitney U test was used for comparisons of two independent groups.
*The difference between groups is statistically significant at 99.9% confidence (p<0.001)
**The difference between groups is statistically significant at 99% confidence (p<0.01)
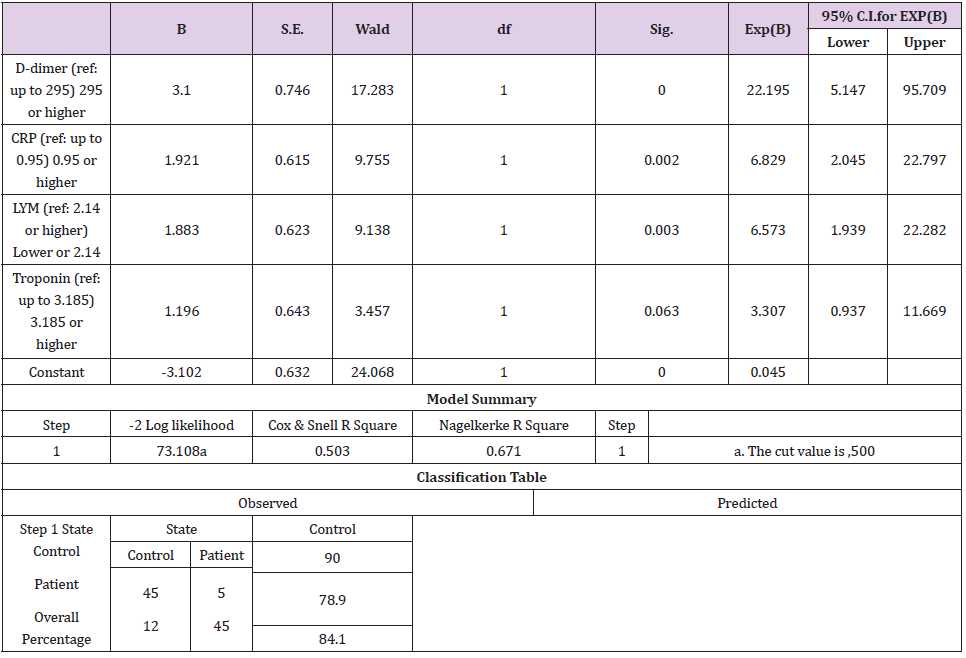
We studied D-dimer, CRP, LYM and troponin parameters binary logistic regression model. In multiple binary logistic regression analysis D-dimer is found as the most meaningful blood parameter for identifying COVID-19 patients (Table 3). Nagelkerke’s R2 determination coefficient was used to evaluate the predictive ability of the model. This model predicts of patient 78.9% and outpatient 90% correctly (Table 3). The Nagelkerke’s R2 value explains 84.1% whether the patients or not (Table 3).
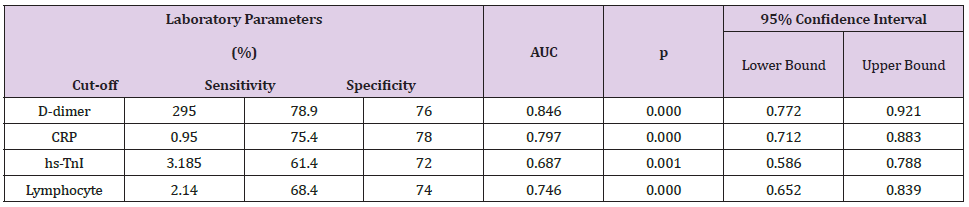
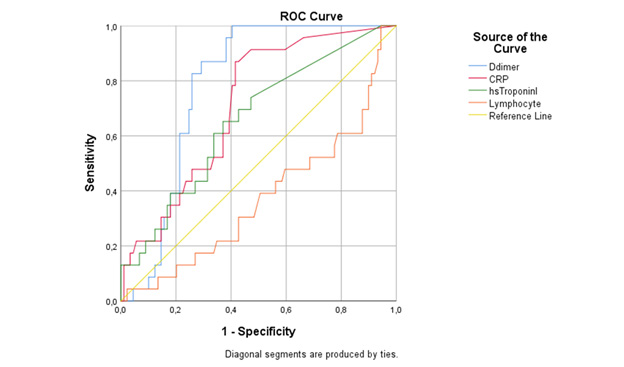
In ROC curve analyze D-dimer was found most sensitive and specific laboratory marker for COVID-19 with the 295μg/L cutoff value (78.9% sensitivity, specificity 76% specificity). The ROC curve analyze of D-dimer, CRP, LYM, and Troponin presented in Table 4, Figure 3).
Discussion
While COVID-19 infection primarily affects the respiratory system, major cardiac complications have been reported, especially in patients with pneumonia [7]. Fatal cardiovascular complications that develop in such patients can be acute myocardial infarction, myocarditis, arrhythmia, heart failure, cardiomyopathy, cardiogenic shock and venous thromboembolism [2,17]. Myocardial injury in these patients has been attributed to several factors. The interaction between SARS-CoV-2 and ACE2 receptors, increased cardio metabolic demand, hypoxia, side effects of medication used for treatment, electrolyte imbalance, systemic inflammation, and plaque rupture due to elevated catecholamine are among these factors [2,4,5,7,18]. The most common cardiac pathologies observed on echocardiography (ECHO) images of COVID-19 patients are reported as right-ventricular (RV) dilatation and dysfunction, left-ventricular (LV) diastolic dysfunction, LV systolic dysfunction, and regional or global wall motion abnormalities. Studies have attributed RV dilatation to pulmonary hypertension caused by pulmonary embolism or acute respiratory distress syndrome. LV dysfunction has been associated with embolism in large or small blood vessels, and ischemia resulting from prothrombotic state [9,19-22]. In our study, TCV was measured on CCT images using the Intrasense Myrian® software. A 42% increase in TCV was observed in patients with COVID-19 pneumonia compared to the control group. This increase in cardiac volume may originate from the right or left heart chambers or may be due to myocardial inflammation. However, in our study, the right and left heart chambers were not measured separately on CCT images; as such, no interpretation could be made regarding this issue.
Table 4: ROC Analysis of Laboratory Parameters Predicting High TCV.
AUC: the area under the curve, CRP: C-reactive protein,hs-TnI: high sensitivity troponin I
In patients with cardiac involvement due to COVID-19 infection, increase in troponin levels, as well as increase in levels of creatine kinase-myocardial band and N-terminal pro-B-type natriuretic peptide has been observed. In these studies, increase in troponin levels has been reported in the range of 7-21%. These studies have also shown that functions of left and right ventricles deteriorate as troponin levels rise [21-26]. A negative correlation has been found between left ventricular ejection fraction and hs-TnI levels [21]. It has been shown that CRP levels also increase in patients with cardiac involvement [20,22,27]. Studies have attributed the increase in levels of troponin and CRP to acute myocarditis [17,18]. In agreement, myocyte necrosis and mononuclear cell infiltration have been observed during autopsy of heart muscle samples from COVID-19 patients. Nevertheless, mechanism, clinical significance and prevalence of myocardial inflammation are yet to be elucidated [2]. It has been reported that in addition to inflammation, COVID-19 infection is complicated with coagulopathy and that the major cause of death is disseminated intravascular coagulation. Abnormal coagulation parameters, especially D-dimer and fibrin catabolic products, have been shown to be significantly elevated in deceased patients. In addition to the growing number of papers reporting high levels of D-dimer in the literature, gradual increase of D-dimer levels during the progression of COVID-19 has been found to be associated with poorer clinical outcomes [28-31]. While high levels of D-dimer and CRP have been shown to be related to RV systolic dysfunction, no such relation has been found with troponin I [20]. In our study, patients with COVID 19 pneumonia had higher CRP and D-dimer levels compared to control patients. On the other hand, hs- TnI levels slightly increased in the patients. Additionally, our results supported that by knowing D-dimer, CRP, LYM and troponin levels, COVID positive status can be predicted.
Even though CT, echo and MRI are being used for identifying cardiac involvement, these imaging methods are not recommended for routine use. When ordering imaging procedures for patients with cardiovascular symptoms, and suspected myocardial dysfunction, severe arrhythmia, Takotsubo cardiomyopathy or myocarditis, troponin levels are taken into consideration [32]. On the other hand, levels of cardiac markers may not increase in every patient with cardiac involvement. In our study, TCV was found to be high in patients with pneumonia. Also, D-dimer, hs-TnI, and CRP were higher as well as TCV in COVID-19 patients.
As far as our knowledge, even higher D-dimer, hs-TnI, CRP, and LYM levels were reported in previous reports, our study is the first report taken together these parameters with TCV. According to our results, the lung capacity of people with COVID-19 has decreased by 27%. Those with COVID-19 have an average of 42% enlarged heart. While D-dimer, CRP and troponin levels increased in the serum values of those patients, and reversely the LYM level decreased. There was no change in serum fibrinogen, WBC and monocyte values of those with COVID-19.
Conclusion
In conclusion, COVID-19 patients may suffer cardiac involvement together with pneumonia. TCV increases in patients with cardiac involvement. The increase in TCV may be due to dilatation of right or left ventricles, or myocardial inflammation. Concurrent with the TCV increase, levels of D-dimer, CRP and troponin I also raise in these patients. According to our results, it seems possible to exclude both COVID-19 and Cardiac involvement in patients without D-dimer, troponin and CRP elevations. Nonetheless, further largescale studies should be conducted using CT and ECHO imagining, as well as laboratory testing in order to investigate COVID-19-related cardiac involvement.
Conflicts of Interest
The author declares that I have no conflicts of interest.
References
- Tahir F, Arif TB, Ahmed J, Malik F, Khalid M (2020) Cardiac Manifestations of Coronavirus Disease 2019 (COVID-19): A Comprehensive Review. Cureus 12(5): e8021.
- Guzik TJ, Mohiddin SA, Dimarco A, Patel V, Savvatis K, et al. (2020) COVID-19 and the cardiovascular system: implications for risk assessment, diagnosis, and treatment options. Cardiovasc Res : cvaa106
- Inciardi RM, Lupi L, Zaccone G, Italia L, Raffo M, et al. (2020) Cardiac Involvement in a Patient With Coronavirus Disease 2019 (COVID-19). JAMA Cardiol 5(7): 819-824.
- Schiavone M, Gobbi C, Biondi-Zoccai G, D’Ascenzo F, Palazzuoli A, et al. (2020) Acute Coronary Syndromes and Covid-19: Exploring the Uncertainties. J Clin Med 9(6): 1683.
- Lazaridis C, Vlachogiannis NI, Bakogiannis C, Spyridopoulos I, Stamatelopoulos K, et al. (2020) Involvement of Cardiovascular System As The Critical Point in Coronavirus Disease 2019 (COVID-19) Prognosis and Recovery. Hellenic J Cardiol.
- Clerkin KJ, Fried JA, Raikhelkar J, Sayer G, Griffin JM, et al. (2020) COVID-19 and Cardiovascular Disease. Circulation 141(20): 1648-1655.
- Gupta R, Das MK, Mohanan PP, Deb PK, Parashar SK, et al. (2020) Cardiological society of India document on safety measure during echo evaluation of cardiovascular disease in the time of COVID-19. Indian Heart J 72(3): 145-150.
- Matsushita K, Marchandot B, Jesel L, Ohlmann P, Morel O (2020) Impact of COVID-19 on the Cardiovascular System: A Review. J Clin Med 9(5): 1407.
- Szekely Y, Lichter Y, Taieb P, Banai A, Hochstadt A, et al. (2020) The Spectrum of Cardiac Manifestations in Coronavirus Disease 2019 (COVID-19)-a Systematic Echocardiographic Study. Circulation 142: 342-353.
- Dhakal BP, Sweitzer NK, Indik JH, Acharya D, William P (2020) (SARS-CoV-2 Infection and Cardiovascular Disease: COVID-19 Heart. Heart Lung Circ 29(7): 973-987.
- Picard MH, Weiner RB (2020) Echocardiography in the Time of COVID-19. J Am Soc Echocardiogr 33(6): 674-675.
- Creel-Bulos C, Hockstein M, Amin N, Melhem S, Truong A, et al. (2020) Covid-19 Cases Acute Cor Pulmonale in Critically Ill Patients with Covid-19. N Engl J Med.
- Bozkurt B, Emek E, Arikan T, Ceyhan O, Yazici P, et al. (2019) Liver Graft Volume Estimation by Manual Volumetry and Software-Aided Interactive Volumetry: Which is Better? Transplantation Proceedings 51(7): 2387-2390.
- Prodhomme O, Seguret F, Martrille L, Pidoux O, Cambonie G, et al. (2012) Organ volume measurements: comparison between MRI and autopsy findings in infants following sudden unexpected death. Arch Dis Child Fetal Neonatal Ed 97(6): F434-438.
- Treece G, Prager R, Gee A, Berman L (2001) 3D ultrasound measurement of large organ volume. Med Image Anal 5(1): 41-54.
- Cheng T, Li Y, Pang S, Wan HY, Shi GC, et al. (2019) Normal lung attenuation distribution and lung volume on computed tomography in a Chinese population. Int J Chron Obstruct Pulmon Dis 14: 1657-1668.
- Long B, Brady WJ, Koyfman A, Gottlieb M (2020) Cardiovascular complications in COVID-19. Am J Emerg Med 38(7): 1504-1507.
- Tersalvi G, Vıcenzı M, Calabretta D, Bıasco L, Pedrazzını G, et al. (2020) Elevated Troponin in Patients With Coronavirus Disease 2019: Possible Mechanisms. J Card Fail 26(6): 470-475.
- Sud K, Vogel B , Bohra C , Garg V , Talebi S , et al. (2020) Echocardiographic Findings in Patients with COVID-19 with Significant Myocardial Injury. J Am Soc Echocardiogr 33(8): 1054-1055.
- Mahmoud-Elsayed HM, Moody WE, Bradlow WM, Khan-Kheil AM, Senior J, et al. (2020) Echocardiographic Findings in Patients With COVID-19 Pneumonia. Can J Cardiol 36(8): 1203-1207.
- Jain SS, Liu Q, Raikhelkar J, Fried J, Elias P, et al. (2020) Indications and Findings on Transthoracic Echocardiogram in COVID-19. J Am Soc Echocardiogr.
- Li Y, Li H, Zhu S, Xie Y, Wang B, et al. (2020) Prognostic Value of Right Ventricular Longitudinal Strain in Patients With COVID-19. JACC Cardiovasc Imaging.
- Huang C, Wang Y, Li X, Ren L, Zhao J, et al. (2020) Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395 (10223): 497-506.
- Shi S, Qin M, Shen B, Cai Y, Liu T, et al. (2020) Association of Cardiac Injury With Mortality in Hospitalized Patients With COVID-19 in Wuhan, China. JAMA Cardiol 5(7): 802-810.
- Deng P, Ke Z, Ying B, Qiao B , Yuan L (2020) The diagnostic and prognostic role of myocardial injury biomarkers in hospitalized patients with COVID-19. Clin Chim Acta 510: 186-190.
- Wang D, Hu B, Hu C, Zhu F, Liu X, et al. (2020) Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA 323(11): 1061–1069.
- Argulian E, Sud K, Vogel B, Bohra C, Garg VP, et al. (2020) Right Ventricular Dilation in Hospitalized Patients With COVID-19 Infection. JACC: Cardiovascular Imaging.
- Tang N, Bai H, Chen X, Gong J, Li D, Sun Z (2020)Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost 18:1094-1099.
- Tang N, Li D, Wang X, Sun Z (2020) Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost 18: 844-847.
- Lippi G, Favaloro EJ (2020) D-dimer is Associated with Severity of Coronavirus Disease 2019: A Pooled Analysis. Thromb Haemost 120(5): 876-878.
- Marietta M, Ageno W, Artoni A, De Candia E, Gresele P, et al. (2020) COVID-19 and haemostasis:a position paper from Italian Society on Thrombosis and Haemostasis (SISET). Blood Transfus 18(3): 167-169.
- Skulstad H, Cosyns B, Popescu BA, Galderisi M, Salvo GD, et al. (2020) COVID-19 pandemic and cardiac imaging: EACVI recommendations on precautions, indications, prioritization, and protection for patients and healthcare personnel. Eur Heart J Cardiovasc Imaging.

 Research Article
Research Article