ABSTRACT
Background/Objective: Patients with thrombocytopenia accompanied with positive Ro/SS-A and/or La/SS-B autoantibodies have possible diagnosis of Sjögren’s syndrome (SS). Because of its low prevalence, large-sample controlled studies on thrombocytopenia in primary SS (pSS) are scarce. Thus, this study aimed to investigate the clinical and laboratory characteristics of pSS complicated with mild to severe thrombocytopenia and compared them with pSS patients without thrombocytopenia.
Methods: This medical records review study analyzed demographic data, clinical manifestations, laboratory examinations, and other results of 88 patients diagnosed with pSS between March 2007 and March 2018 in the Department of Rheumatology of the First Affiliated Hospital of Soochow University. Platelet count of peripheral blood below 50×109/L (≤50×109/L) was regarded as mild to severe thrombocytopenia. Results: Of the 88 pSS patients, 43 developed mild to severe thrombocytopenia (thrombocytopenia group) and 45 had no thrombocytopenia (control group). No significant difference was found in the levels of autoantibodies and inflammatory markers between the thrombocytopenia group and the control group. Dry mouth (P < .01) and dry eyes (P < .01) were not frequently observed in the thrombocytopenia group, but the level of complement C4 dropped significantly (P < .05). In contrast, the thrombocytopenia group more likely had leukopenia (P = .01) and interstitial lung disease (P < .01). Conclusion: In pSS patients with mild to severe thrombocytopenia, the incidence of xerostomia, xerophthalmia, and lung involvement reduced markedly. Knowledge on the features of pSS associated with thrombocytopenia will lead to early and better diagnosis and treatment.Abbreviations: SS: Sicca syndrome; EULAR: European League Against Rheumatism; CT: Computed Tomography; RF: Rheumatoid factor; ACA: Anticentromere Antibodies; IgA: Immunoglobulin A; ITP: Immune Thrombocytopenic Purpura
Introduction
Sjögren’s syndrome (SS; also known as Sicca syndrome)
is a chronic systemic autoimmune disease characterized by
xerophthalmia (dry eyes) and/or xerostomia (dry mouth). In
addition to the impaired function of exocrine glands, other organs
may be also involved, such as the skin, joints, lungs, liver, kidneys,
and hematological system. Hematological system involvement is
common in SS patients, and a third of patients with SS have cytopenias
such as leukopenia, anemia, and thrombocytopenia [1]. In primary SS (pSS), one of the most common complications of the hematological
system is leukopenia, while a few cases were complicated with
autoimmune hemolytic anemia and thrombocytopenia [2]. A study
analyzed 99 pSS patients and reported that 61 (61.5 %) developed
lymphopenia and leukopenia and 7 (7.1%) had thrombocytopenia
[3]. Although thrombocytopenia is not common in pSS patients
[4], severe thrombocytopenia can increase the occurrence of
adverse events, which are sometimes fatal. Some patients were
misdiagnosed because they lack the typical signs in their eyes and
mouths [5].
Thus, in 2015, the European League Against Rheumatism
(EULAR) has promoted and supported an international collaborative
study group (EULAR-SS Task Force) aimed at developing
consensual recommendations to provide a homogeneous
approach to pSS patients presenting with systemic involvement.
The guideline mentioned that patients with thrombocytopenia
accompanied with positive Ro/SS-A and/or La/SS-B autoantibodies
have possible diagnosis of SS [6]. Because of its low prevalence,
large-sample controlled studies on thrombocytopenia in pSS are
scarce. Thus, in this study, we aimed to investigate the clinical and
laboratory characteristics of pSS complicated with mild to severe
thrombocytopenia and compared them with those of pSS patients
without thrombocytopenia.
Methods
Design
In this medical records review study, we identified 841pSS patients hospitalized in the Department of Rheumatology of the First Affiliated Hospital of Soochow University between March 2007 and March 2018. Of these pSS patients, 43 developed mild to severe thrombocytopenia (thrombocytopenia group), and 45 patients who did not developed thrombocytopenia were included in the analysis as the control group. The age and sex of the control group were matched to those of the thrombocytopenia group. Patients’ demographic data, clinical manifestations, laboratory examinations, and other results were analyzed retrospectively.
Patients
PSS diagnosis was confirmed either by pathological or clinical method based on the 2002 American College of Rheumatology Classification Criteria [7]. Patients with other diseases such as chronic hepatitis C, human immunodeficiency virus infection, previous lymphoproliferative processes, or other autoimmune diseases were excluded. The study was approved by the Institutional Ethics Board of Medical College, Soochow University (No. 2018- 012).
Clinical Features
The clinical characteristics of pSS are as follows: fever, axillary temperature >37.5℃; Raynaud phenomenon, cool skin and cutaneous color changes of the fingers and toes exposed to cold and/or stress; articular feature, arthralgia or non-erosive arthritis involving two or more peripheral joints; pulmonary complications, chronic and persistent cough, dyspnea, or both, with alveolitis or fibrosis in computed tomography (CT) scans; nephropathy, permanent proteinuria (>0.5 g/day), continuously increasing serum creatinine level (>111 μmol/L), renal tubular acidosis, or glomerular nephritis; liver damage, altered plasma liver function (aminotransferase, alkaline phosphatase, gamma glutamyl transferase, and bilirubin) and/or altered bile ducts in ultrasonography, CT, or magnetic resonance imaging; hemorrhagic manifestations, skin bleeding (skin purpura or bruises), oral bleeding (oral blood blister or gingival bleeding), nasal hemorrhage, gastrointestinal bleeding (visible bleeding or fecal occult blood), urinary bleeding (urinating blood or microscopic hematuria), vaginal bleeding (massive bleeding or prolonged menstrual period), conjunctival bleeding, and intracerebral hemorrhage. Bleeding seriousness was assessed using the immune thrombocytopenic purpura (ITP) bleeding scoring system [8].
Laboratory Features
The laboratory features were as follows: mild to severe thrombocytopenia manifested by platelet count <50×109/L, leukopenia as leucocyte count <4.0×109/L, anemia as hemoglobin <120 g/L, and hypocomplementemia with complement C3 <0.79 g/L and/or complement C4 <0.16 g/L. Rheumatoid factor (RF) (latex test positive at a value >20 IU/mL) was analyzed by enzymelinked immunosorbent assay. Antinuclear antibodies (ANAs; positive at a titer 1:100 by indirect immunofluorescence) and 60- kDa and 52-kDa forms of anti-Ro/SSA and anti-La/SSB antibodies were tested independently.
Statistical Analysis
SPSS software version 21.0 (IBM Corp., Armonk, NY, USA) was used for statistical analysis. Continuous variables were compared using the Wilcoxon test, and proportions were analyzed using chisquare and Fisher’s exact test. A value of p<0.05 indicated statistical significance.
Results
Patient Characteristics
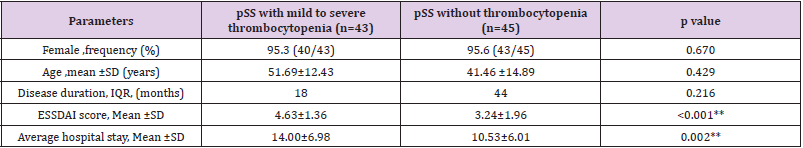
In this study, the total incidence rate of mild to severe thrombocytopenia in pSS patients was 5.1%. Among 43 patients with mild to severe thrombocytopenia, only 3 (6.9%) were men. pSS patients with mild to severe thrombocytopenia were 17–70 years old, with a mean age of 51.69±12.43 years, while control patients were 41.46 ± 14.89 years old. The median disease duration in the thrombocytopenia group was 18 months versus 44 months in the control group (Table 1).
Table 1: Characteristics of pSS patients with or without mild to severe thrombocytopenia.
Note: *p<0.05; **p<0.01
Clinical Features in pSS Patients with Mild to Severe Thrombocytopenia
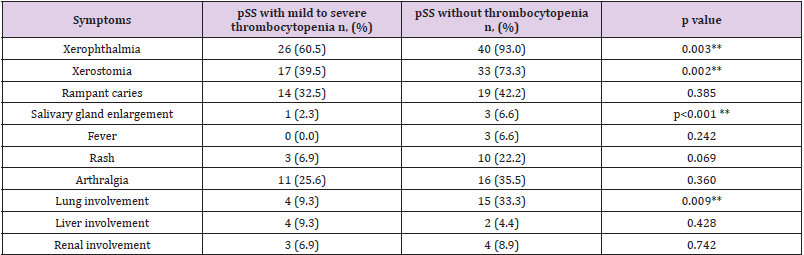
Table 2 presents the clinical features of the patients in the thrombocytopenia group. The classic manifestations of SS, such as dryness of the mouth and eyes, rampant caries, and other clinical features like fatigue, fever, and weight loss were observed. The incidence rates of xerostomia and xerophthalmia were significantly lower in the thrombocytopenia group than in the control group (P = .0028, and P = .0024, respectively). Of the 43 patients in the thrombocytopenia group, 17 sought admission because of thrombocytopenia and/or hemorrhagic manifestations, without exocrine gland features. While pulmonary interstitial disorders were more common in the control group (P = .0088). The frequency of fever, rashes, arthritis, and Raynaud phenomenon did not show statistically significant difference between the two groups. As expected, the thrombocytopenia group had significantly higher EULAR Sjögren’s syndrome disease activity index than the control group (ESSDAI scores) (P < .001).
Table 2: Clinical features in pSS patients with or without mild to severe thrombocytopenia.
Note: *p<0.05; **p<0.01
Difference in Serologic Features
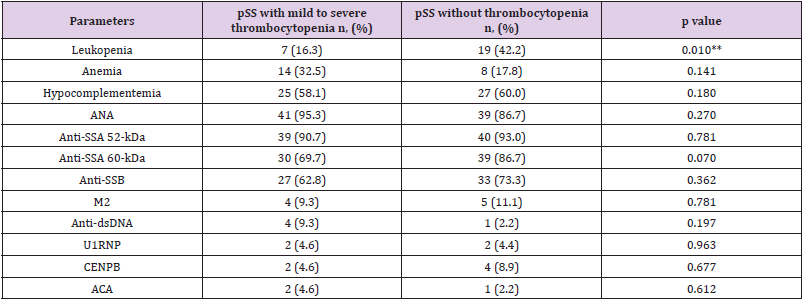
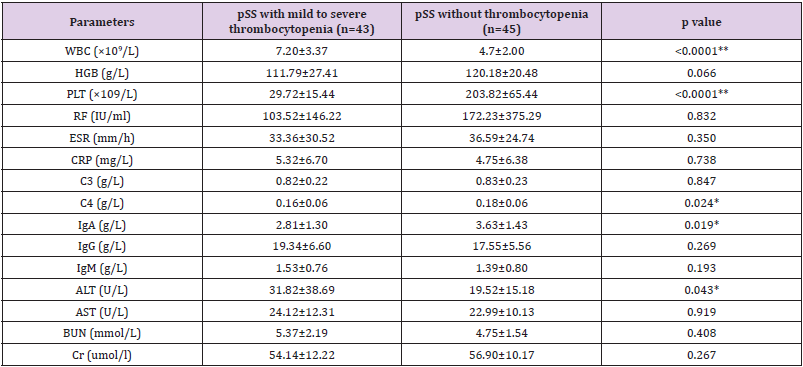
Not surprisingly, the platelet count in the thrombocytopenia group was significantly lower than that in the control group (P < .001). The thrombocytopenia group had significant hypocomplementemia (C4 level) than the control group (P = .0024). However, the existence of ANA, anti-SSA 60-kDa and 52- kDa, anti-SSB, M2, anti-dsDNA, and anticentromere antibodies (ACAs) was not obviously different between the two groups (Table 3). Levels of erythrocyte sedimentation rate, C-reactive protein, and RF were not markedly different between the two groups. The levels of immunoglobulin A (IgA) were significantly lower in the thrombocytopenia group than in the control group (P = .0019); however, no difference was found in the levels of IgG and IgM between the two groups. Among the regular chemical parameters, the level of glutamic-pyruvic transaminase (alanine transaminase) were significantly increased in the thrombocytopenia group (P = .043) (Table 4).
Table 3: Serologic features in pSS patients with or without mild to severe thrombocytopenia.
Note: *p<0.05; **p<0.01
Table 4: Serologic features in pSS patients with or without mild to severe thrombocytopenia.
Note: *p<0.05; **p<0.01
Disease and Prognosis in pSS Patients with Mild to Severe Thrombocytopenia
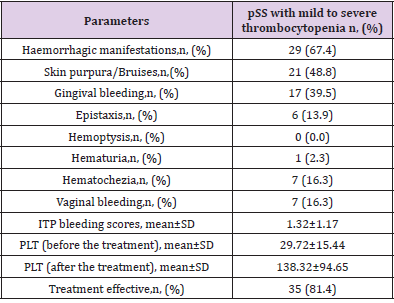
In the thrombocytopenia group, the most common hemorrhagic manifestations were skin bleeding, such as purpura/bruises (21 of 43, 48.8%), and gingival bleeding (17 of 43, 39.5%). There were no hemorrhagic manifestations in 14 of 43 patients (32.6%) (Table 4). At baseline, the ITP bleeding score was 1.32±1.17, and no correlation was found between hemorrhagic manifestations and treatment effect (Table 5). No patient died during the study period. Mild to severe thrombocytopenia was treated with highdose corticosteroids and/or intravenous gamma-immunoglobulin, and hydroxychloroquine and cyclosporine were also used. During hospitalization, among 43 patients, 35 were responsive and 8 were not responsive to the treatment. One patient had undergone splenectomy because of refractory thrombocytopenia.
Discussion
To the best of our knowledge, this is the first study to show a
negative relation between mild to severe thrombocytopenia
and lung involvement in pSS. pSS is one of the major chronic
inflammatory autoimmune diseases associated with B lymphocyte
hyper-reactivity. Recently, the reported prevalence of pSS ranges
from 0.05% to 0.23% [9,10], and in China, it is approximately
0.33% to 0.77% [11]. Although most patients have exocrine gland
involvement, such as labial gland and lacrimal gland involvement
[12], their clinical manifestation is usually quite nonspecific and
varied, resulting in the delay in the diagnosis for 3-8 years from the
onset of first symptoms [13,14]. In our study, all 88 patients had
pathological examination of lower lip biopsy. Among pSS patients
with mild to severe thrombocytopenia, 4 had no lymphocytic
foci, but in pSS patients without thrombocytopenia, 1 had no
lymphocytic foci. Thrombocytopenia is not common in pSS patients.
In our study, 5.1% of the pSS patients developed mild to severe
thrombocytopenia; among them, 95.3% were female.
Moreover, our participants were inpatients, making our
cohort similar to those in other studies. Studies reported that
5%-16% of pSS patients developed thrombocytopenia [15,11,16].
Although there was no real distinction in the median time for
diagnosis confirmation between patients with or without mild
to severe thrombocytopenia, pSS patients with mild to severe
thrombocytopenia obviously had higher ESSDAI scores than those
without, and they needed longer hospitalization. Leukopenia is the
most frequent hematologic abnormality noted in pSS, and 30%-40%
of pSS patients may have leukopenia [2]. In our study, 29.5% of the
patients have leukopenia, but among patients with mild to severe
thrombocytopenia, the prevalence of leukopenia was significantly
lower than in those without thrombocytopenia. All patients
responded remarkably well to corticosteroids. Antineutrophil
antibody may be responsible for autoimmune neutropenia [17],
but the low frequency of neutropenia in pSS patients with mild to
severe thrombocytopenia was not reported in previous studies;
thus, further research is required. In our study, decreased levels
of C4 and IgA were more common in the thrombocytopenia group,
indicating the possible important complex immune mechanism in
the pathogenesis of thrombocytopenia in pSS patients.
A large proportion of patients in both groups were positive for
ANA. The prevalence of all autoantibodies such as anti-SSA and
anti-SSB, AIM-M2, CENPB, ACA, anti-dsDNA, and U1RNP showed
no remarkable difference between the two groups. For some
antibodies, the absence of a significant difference may have resulted
from a relatively small sample size. There was lung involvement
in 10%-20% of pSS patients [18]. Among 88 patients, 21.6%
had lung involvement, which fairly accorded with the results in
previous studies [19]. However, in pSS patients with mild to severe
thrombocytopenia, the incidence of lung involvement reduced
markedly in our study. Platelet-derived growth factor was reported
to contribute directly to the migration of fibrocytes to the injured
lungs [20]. Current understanding of the pathophysiology of lung
disease in pSS suggests a similar process as those in the salivary
glands, with epithelial cells playing a critical role in the initiation
[19]. It can be assumed that pSS with lung involvement and pSS
with hematological involvement have different pathological and
clinical manifestations. However, this study has limitations that
hindered the generalization of the results: small sample size,
retrospective design, single-center setting, etc. More research is
needed on the relationship between thrombocytopenia and lung
involvement in pSS.
Conclusion
our study explored the clinical features of pSS accompanied with mild to severe thrombocytopenia. pSS patients with mild to severe thrombocytopenia could have higher ESSDAI scores than those without thrombocytopenia, but the incidence of some complications such as leukopenia and lung involvement was low. In pSS patients with mild to severe thrombocytopenia, the incidence of xerostomia, xerophthalmia, and lung involvement reduced markedly. Thrombocytopenia could be present upon the onset of pSS development, without any involvement of the exocrine glands. Thus, there is an urgent need for more detailed classification of pSS based on its clinical manifestations and pathology. Knowledge on the features of pSS associated with thrombocytopenia will help in early and better diagnosis and treatment.
Acknowledgement
We would like to thank Editage (www.editage.cn) for English language editing.
Source of Funding
This work was supported by grants from the National Natural Science Foundation of China (No. 81771782, No. 81801595, and No.81873876).
Conflicts of Interest
The authors have no conflict of interest to declare.
References
- Ramos-Casals M, Daniels TE, Fox RI (2009) Sjögren’s syndrome. In: Stone JH (Edt.)., A clinician’s pearls and myths in rheumatology. New York (NY): Editorial Springer.
- Schattner A, Friedman J, Klepfish A, Berrebi A (2000) Immune cytopenias as the presenting finding in primary Sjogren’s syndrome. QJM 93: 825-829.
- Aoki A, Ohno S, Ueda A, Ideguchi H, Ohkubo T, et al. (2000) Hematological abnormalities of primary Sjogren's syndrome. Nihon Rinsho Meneki Gakkai Kaishi 23: 124-128.
- Ramakrishna R, Chaudhuri K, Sturgess A, Manoharan A (1992) Haematological manifestations of primary Sjögren’s syndrome: a clinicopathological study. Q J Med 83: 547-554.
- Khattri S, Barland P (2012) Primary Sjogren’s syndrome and autoimmune cytopenias: a relation often overlooked. Bull NYU Hosp Jt Dis 70: 130-132.
- Brito-Zerón P, Theander E, Baldini C, Seror R, Retamozo S, et al. (2016)Early diagnosis of primary Sjögren’s syndrome: EULAR-SS task force clinical recommendations. Expert Rev Clin Immunol 12: 137-156.
- Vitali C, Bombardieri S, Jonsson R, Moutsopoulos HM, Alexander EL, et al. (2002) Classification criteria for Sjogren’s syndrome: a revised version of the European criteria proposed by the American-European Consensus Group. Ann Rheum Dis 61: 554-558.
- (2016) Thrombosis and Hemostasis group, Hematology Society, Chinese Medical Association. Consensus of Chinese experts on diagnosis and treatment of adult primary immune thrombocytopenia (version 2016). Zhonghua Xue Ye Xue Za Zhi 37: 89-93.
- Birlik M, Akar S, Gurler O, Sari I, Birlik B, et al. (2008)Prevalence of primary Sjogren’s syndrome in Turkey: a population-based epidemiological study. Int J Clin Pract 63: 954-961.
- Anagnostopoulos I, Zinzaras E, Alexiou I, Papathanasiou AA, Davas E, et al. (2010)The prevalence of rheumatic diseases in central Greece: a population survey. BMC Musculoskelet Disord 11: 98.
- Zhang NZ, Shi CS, Yao QP, Pan GX, Wang LL, et al. (1995) Prevalence of primary Sjogren’s syndrome in China. J Rheumatol 22: 659-661.
- Asmussen K, Andersen V, Bendixen G, K Bendtzen, JU Prause, et al. (1997) Quantitative assessment of clinical disease status in primary Sjögren’s syndrome. A cross-sectional study using a new classification model. Scand J Rheumatol 26: 197-205.
- Pavlidis NA, Karsh J, Moutsopoulos HM (1982) The clinical picture of primary Sjogren's syndrome: a retrospective study. J Rheumatol 9: 685-690.
- Markusse HM, Oudkerk M, Vroom TM, Breedveld FC (1992) Primary Sjogren’s syndrome: clinical spectrum and mode of presentation based on an analysis of 50 patients selected from a department of rheumatology. Neth J Med 40: 125-134.
- Asmussen K, Andersen V, Bendixen G, M Schiødt, P Oxholm (1996) A new model for classification of disease manifestations in primary Sjögren’s syndrome: evaluation in a retrospective long-term study. J Intern Med 239: 475-482.
- Alexander EL, Arnett FC, Provost TT, Stevens MB (1983) Sjögren’s syndrome: association of anti-Ro(SS-A) antibodies with vasculitis, hematologic abnormalities, and serologic hyperreactivity. Ann Intern Med 98: 155-159.
- Hartman KR, Mallet MK, Nath J, Wright DG (1990) Antibodies to actin in autoimmune neutropenia. Blood 75: 736-743.
- Kokosi M, Riemer EC, Highland KB (2010) Pulmonary involvement in Sjögren syndrome. Clin Chest Med 31: 489-500.
- Gupta S, Ferrada MA, Hasni SA (2019) Pulmonary manifestations of primary Sjögren's syndrome: Underlying immunological mechanisms, clinical presentation, and management. Front Immunol 10: 3389-3398.
- Aono Y, Kishi M, Yokota Y, Azuma M, Kinoshita K, et al. (2014) Role of platelet-derived growth factor/platelet- derived growth factor receptor axis in the trafficking of circulating fibrocytes in pulmonary fibrosis. Am J Respir Cell Mol Biol 51: 793-801.

 Research Article
Research Article