Abstract
To evaluate the mechanisms of Melatonin (MT)-alleviating Heat Stress (HS) in porcine oocytes in vitro, the concentration of Reactive Oxygen Species (ROS), mitochondrial distribution, the first polar body expulsion, as well as the mRNA expressions of BCL-2, BAX, CAT, and ER in the oocytes were determined, along with the estimation of the developmental potential and the total number of blastocysts after the oocytes in the control (No HS), HS, and HSMT (HS+MT) groups had been cultured to maturation in vitro. Our results of the rates of first polar body expulsion, cleavage, blastocyst count, the total number of blastocysts, the proportion of oocytes with better mitochondrial distribution, and the CAT and BCL-2 expressions had significantly improved, but the ROS concentration and BAX expression were significantly decreased in the HSMT group in comparison with those in the HS group. However, the blastocyst rate, the total number of blastocysts, the ratio of oocytes with better mitochondrial distribution, and the CAT expression remained significantly lower, while the ROS concentration was significantly higher than the corresponding control values (P<0.05). As for the ER expression, no significant difference was noted between the HS and HSMT groups, albeit both showed significantly lower values than those in the control group (P < 0.05). Thus, the characteristic of CAT and ER with high sensitivity to high temperature and low sensitivity to MT was found to be associated with poor ooplasmic (bad mitochondrial maturation and higher ROS concentration), and not to the nuclear maturation in vitro.
Keywords: Heat Stress; Melatonin; Gene Expression; Ooplasmic Maturation; Porcine Oocyte
Abbreviations: MT: Melatonin; HS: Heat Stress; ROS: Reactive Oxygen Species; GV: Germinal Vesicle; COCs: Cumulus and Oocyte Complexes; PBS: Phosphate-Buffered Saline
Introduction
High temperature not only reduces the oocytes maturation
quality [1-4] but also has a tremendous adverse impact on the
animal ovarian function and embryo attachment [5,6]. Heat
stress (HS) damage of oocyte can inhibit cumulus cells expansion
[7], which lead to the abnormal distribution of organelles [8,9],
decreased antiapoptotic and estrogen receptor gene expressions,
enhanced apoptosis gene expression [10], as well as increased
Reactive Oxygen Species (ROS) concentration [11,12] in the
oocytes. In fact, excessive ROS can induce DNA damage and lipid
peroxidation, disrupt the mitochondrial function [13,14], and induce abnormal gene expression and protein synthesis [15]. Reportedly,
mitochondrial maturation distribution is an important indicator
of the oocyte quality. Oocytes with poor maturation quality have
a non-uniform mitochondrial distribution, whereas mitochondria
are uniformly distributed in the ooplasm [16]. In order to protect
oocytes from HS damage and improve their maturation quality,
several materials are supplemented in the maturation medium
during in vitro oocyte maturation, such as insulin-like growth
factor, β-mercaptoethanol, astaxanthin, anthocyanins, Melatonin
(MT), and coagulated proteins [17-20].
It has been reported that MT is involved in regulating several
different physiological processes; it can promote the expression
of antioxidant-related genes and improve the oocyte maturation
quality and embryo developmental potential [21]. In addition,
MT concentration of 10-9 M has been proven to be effective in
promoting porcine oocyte maturation and development [22].
Oocyte maturation involves several complex events that coordinates
nuclear and cytoplasmic maturation processes. Cytoplasmic
maturation events following meiotic maturation is much more
difficult to assess microscopically, such as the abnormal distribution
of the mitochondria, lipid droplets [23], and Glutathione (GSH)
concentration detection. However, the nuclear maturation process
involves the Germinal Vesicle (GV) breakdown, chromosomal
arrangement, and completion of Metaphase 1 (MI) by extruding
the first polar body into the perivitelline space of the oocytes, all
of which were observed by stereomicroscopy in a previous study
[24]. It has been reported that HS can increase the expression of
proapoptotic genes, enhance the activity of caspase proteins, and
trigger the apoptosis pathway [25,26]. The members of the BCL-
2 family play a key role in regulating apoptosis, among which the
expressions of the proapoptotic gene BAX and antiapoptotic gene
BCL-2 as well as the ratio of these two gene expression levels are
generally deemed as indicators for predicting the oocyte maturation
quality and the embryo developmental potential.
Past studies in cattle have reported that the expressions of
genes related to oocyte maturation quality and developmental
potential were greatly reduced after the Cumulus and Oocyte
Complexes (COCs) were subjected to HS at 41℃ for 12h. The present
study discusses the sensitivity of BCL-2, BAX, CAT, and ER to high
temperature and MT and analyzes the relationship among their
sensitive differences and porcine oocyte maturation quality and
developmental potential in vitro. We first discovered that the low
sensitivity of CAT and ER to MT possibly contributes to the strong
relationship with the poor porcine oocyte maturation quality and
the developmental capacity of the embryos in vitro. We believe
that the present results would be helpful in enhancing oocytes
utilization and would provide a practical guide toward improving
pig fertility during the high-temperature season.
Materials & Methods
All reagents used in this experiment were purchased from Sigma Chemicals (St. Louis, MO, USA), unless otherwise specified.
Oocytes Collection and Culturing In Vitro
The ovaries were acquired from a slaughterhouse and dispatched to our laboratory within 2 h of collection in a thermos flask containing sterile saline at 35-37℃. The COCs were extracted from follicles (of 2-6-mm diameter) with a disposable syringe (10 mL; No. 18 needle), and only COCs with uniform ooplasm and compact cumulus cells were maturation cultured in an incubator with 5% CO2 and 95% humidified air atmosphere, as follows: some COCs were cultured at 38.5℃ for 42 h in a maturation medium (Control, No HS); some COCs were cultured at 41.5℃ for 4 h and then transferred for continuous culturing at 38.5℃ for 38 h in the maturation medium (HS group); and the other COCs were cultured at 41.5℃ for 4 h in a medium supplemented with 10-9 M MT, after which it was subjected to continuous cultured for 38 h in a maturation medium at 38.5℃ (HSMT group). The maturation medium consisted of TCM199 (with Earle’s Salts; Gibco, Grand Island, NY, USA) supplemented with 10% porcine follicular fluid (PFF), 0.1 mg/mL cysteine, 0.065 mg/mL penicillin, 10 ng/ mL epidermal growth factor (EGF), 10 IU/mL equine chorionic gonadotropin (eCG; Intervet Pty. Ltd., Boxrneer, Australia), and 10 IU/mL human chorionic gonadotrophin (hCG; Intervet Pty. Ltd.). All the experiments were repeated thrice.
Assessment of the First Polar Body Expulsion Rate
The COCs from different groups were respectively stripped off cumulus cells by gentle pipetting in Phosphate-Buffered Saline (PBS) supplemented with 0.1% hyaluronidase, and the first polar body expulsion rate was determined under a stereomicroscope. A total of 150 denuded oocytes from each group were used for determining the rate of the first polar body expulsion. All oocytes used in the subsequent experiments had their first polar body expulsed.
Ooplasmic ROS Detection
ROS was detected using the Reactive Oxygen Species Assay Kit (S0033; Beyotime®, Haimen, Jiangsu, China) as per the manufacturer’s instructions. Briefly, 50 matured oocytes from each group were rinsed in the PBS solution thrice, followed by dying with 10× M ROS dye in the dark for 10 min. Next, photographs were taken under a fluorescence microscope (TE2000-s; Nikon, Japan). The fluorescence intensity analysis was performed with the Image J (Version 1.8.0) software, and the experiment was repeated thrice.
Ooplasmic Mitochondrial Distribution Analysis
Mitochondrial staining was performed with the Mito-Tracker Red CMXROS (C1049; Beyotime®) as per the manufacturer’s instructions. Briefly, 50 oocytes from each group were stained with 200-nM mitochondrial dye in the PBS solution for 25 min at 37℃ in the dark after washing in PBS thrice. Then, the stained oocytes were observed under the fluorescence microscope. The mitochondrial distribution pattern of porcine oocytes was then characterized based on two main distribution features: uniform distribution throughout the ooplasm or non-uniform distribution throughout the ooplasm.
Parthenote Production and Culture In Vitro
A total of 100 matured oocytes from each group were transferred to the activation medium (composed of 1.0 mM CaCl2, 0.1 mM Mg Cl2, 0.3 M mannitol, and 0.5 mM HEPES). Matured oocytes were activated with two pulses of 120 V/mm DC for 60 ms with the Electro-Cell Manipulator BTX 2001 (BTX Inc., USA). After activation, the parthenotes were subsequently cultured in 2 mM 6-dymethylaminopurine (6-DAMP) for 6 h and then the parthenotes were transferred into the PZM-3 medium for 7 days in an incubator at 39℃ under 5% CO2 atmosphere in humidified air. The rates of cleavage and blastocyst transfer were observed respectively on days 2 and 7 after oocytes activation.
Blastocyst Cells Staining
A total of 10 blastocysts were randomly selected from each group and fixed in 4% paraformaldehyde prepared in PBS containing 0.1% polyvinyl alcohol (PVA) for 1 h after washing thrice, and the blastocysts were then permeabilized in PBS-0.1% PVA solution containing 0.3% Triton for 30 min. After washing, the blastocysts were transferred to a solution supplemented with 10 μg/mL DAPI dye for 1 min, and then the blastocysts were mounted on a slide and covered with a coverslip. The total blastocyst count in each group was counted under the fluorescence microscope.
Gene mRNA Expression
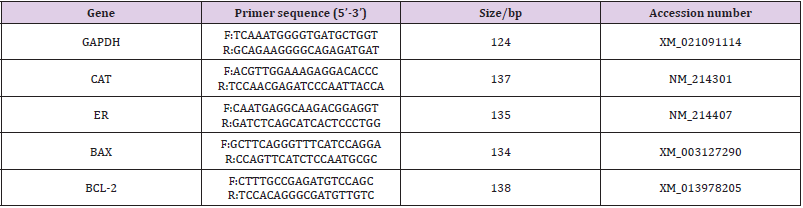
The expressions of BCL-2, BAX, CAT, and ER were analyzed by reverse transcription-polymerase chain reaction (RT-PCR). Total RNA of 100 oocytes from each group was extracted by using the Micro RNA Extraction Kit (160027349; Qiagen, CA, USA) as per the manufacturer’s instruction. After washing in PBS, the oocytes were transferred to a 200-μL centrifuge tube for pre-cooling and then stored at -80℃ for further processing. After total RNA extraction, cDNA synthesis was performed for 30 min at 55°C using the Omniscript Reverse Transcription Kit (Invitrogen) with oligodT primer. PCR was performed by using the Maxime PCR Premix with SYBR Green (TaKaRa Bio Inc., Otsu, Japan) supplemented with each primers and cDNA samples under the following conditions: predenaturation at 95°C for 3 min, denaturation at 95°C for 15 s, annealing at 56°C for 30 s, elongation at 72°C for 30 s, and a final extension at 72°C for 5 min for 40 cycles using the Eppendorf Mastercycler (Eppendorf, Hamburg, Germany). According to the mRNA sequences of Sus scrofa genes published on Gen Bank, we designed primers with the Primer 5.0 software and synthesized by Shanghai Bioengineering Co., Ltd. (Shanghai, China). The primers used in the present study were verified for their availability by RTPCR. Real-time quantitative PCR was performed by the comparative Ct (2 -△△Ct ) method, and the results obtained for each gene in each cDNA pool were normalized based on the GAPDH ratio. The primers and Genebank source accessions for each gene are listed in Table 1.
Statistics
The percent values were subjected to log transformation before analysis, and the quantitative data were analyzed by least-square analysis of variance (ANOVA) using the General Linear Models (GLM) procedures of the Statistical Analysis System (SAS Institute, Cary, NC, USA). We corrected the real-time PCR data by using the GAPDH data as a covariate for the analysis of differences. All data were expressed as mean ± SEM, with P < 0.05 deemed as statistically significant. All experiments were repeated thrice.
Results
Assessment of the First Polar Body Expulsion Rates of Oocytes in Different Groups
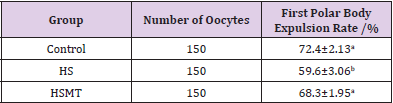
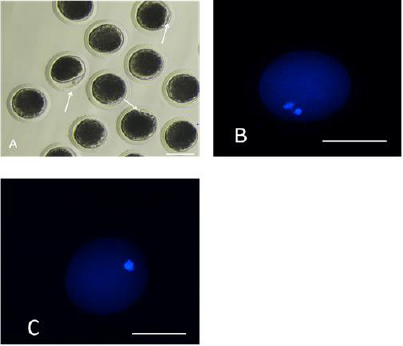
The first polar body expulsion rates of porcine oocytes are given in Table 2. Although the first polar body expulsion rate greatly decreased in the HS group in comparison with that in the control group, the corresponding rate in the HSMT group significantly increased again but exhibited no significant difference with that in the control group (P > 0.05). The first polar body of porcine oocytes was observed under a stereomicroscope (Figure 1A), and the observation was confirmed under a fluorescence microscope (Figure 1B). Figure 1C represents oocytes without the expulsed first polar body, indicating that fluorescence occurred only at the nuclear sites observed under the fluorescence microscope.
Figure 1: Porcine oocytes with or without the first polar body expulsed
A. The stereomicroscopic examination of the matured oocytes with the first polar body expulsed, as pointed by the arrows.
B. The matured oocytes were stained with fluorescent dye Hoechst 33342, where both the polar body and the nucleus
exhibited fluorescence.
C. Oocytes with no polar body expulsed were stained with fluorescent dye Hoechst 33342, where only the nucleus exhibited
fluorescence as pointed by the arrows. Bar=100 μm.
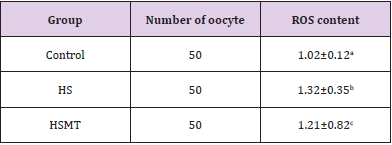
ROS Concentration in Oocytes of Different Groups
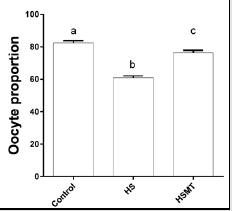
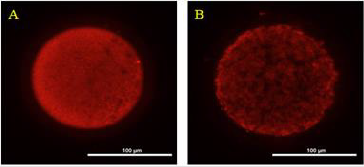
The ROS concentrations in the HS group was significantly increased relative to those in the control group. In addition, although the ROS concentration in the HSMT group decreased significantly, it remained significantly higher than that in the control group as showed in Table 3 (P < 0.05). The proportion of oocytes with uniform mitochondrial distribution in porcine oocytes of different groups. Figure 2 supports that the proportion of oocytes with a uniform mitochondrial distribution in the HS group was significantly lower than that in the control group (P < 0.05). Although the proportion of oocytes with uniform mitochondrial distribution was greatly enhanced in the HSMT group, it remained significantly lower than that in the control group (P < 0.05). Figure 3 represents the uniform and non-uniform mitochondrial maturation distribution in the ooplasm.
Figure 2: The proportion of oocytes with better mitochondrial maturation distribution in different groups. Different letters (a-c) over a bar means significant difference (P < 0.05). Each experiment was repeated three times.
Figure 3: The condition of mitochondrial maturation distribution in ooplasm Oocyts were fluorescent stained by Mito-Tracker Red, and A. Represents the oocyte of better maturation quality with even distribution of mitochondria in ooplasm. B. Represents the oocyte of poor maturation quality with uneven distribution of mitochondria in ooplasm. Bar = 100 μm.
Assessment of Porcine Oocytes Developmental Potential In Vitro
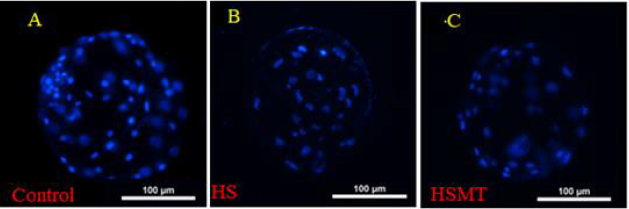
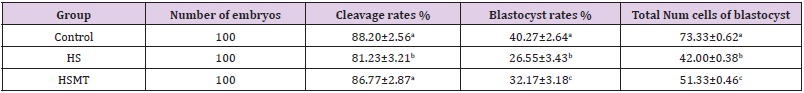
As can be seen in Table 4, the cleavage rate, blastocyst rate, and the total number of blastocysts were significantly lower in the HS group than in the control group (P < 0.05). As compared with those in the HS group, the rates of cleavage, blastocysts, and the total number of blastocysts in the HSMT group were significantly increased (P < 0.05), with no significant difference in the cleavage rate relative to that in the control group (P > 0.05). Nevertheless, the blastocyst rate and the total number of blastocysts remained significantly lower than the respective control values (P < 0.05). The results of blastocyst cells staining are depicted in Figure 4.
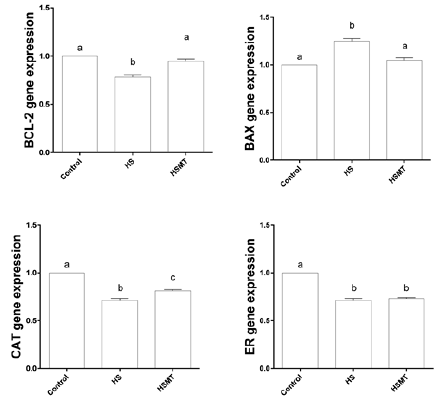
The mRNA Expressions of CAT, BCL-2, BAX, and ER in Oocytes of Different Groups
As shown in Figure 5, the mRNA expressions of CAT, ER, and BCL- 2 were decreased, whereas the BAX expression was significantly increased in the HS group in comparison with those in the control group. However, in the HSMT group, the mRNA expression levels of BCL-2 and BAX were restored to the control levels, while the CAT and ER expressions remained significantly lower than the control values (P < 0.05). Moreover, no significant difference was noted in the mRNA expression of ER between the HS and HSMT groups (P > 0.05).
Figure 4: The fluorescence staining of blastocyst cells. The blastocyst cells was fluorescent stained with 10μg/ml DAPI dye and
a number of 10 blastocysts randomly selected from each group.
A. The blastocyst comes from the Control
B. The blastocyst comes from the HS group
C. The blastocyst comes from the HSMT group. Bar = 100 μm.
Figure 5: The mRNA expressions of CAT, BCL-2, BAX and ER genes in oocytes of different groups. Different letters (a-c) over a bar means significant difference (P < 0.05). Each experiment was repeated three times.
Discussion
Previous studies have shown that the effects of MT exposure
on protecting oocytes from HS damage varied among different
animals. Negrón-Pérez reviewed that the cleavage rate and the
total number of blastocysts in dairy cows increased when MT was
supplemented in the HS system [27], while another bovine study
demonstrated that MT has no significant effect on the cleavage rate
and on the total number of blastocysts [28]. The present study on
porcine suggested that, although 10-9 M concentration of MT could
significantly improve the cleavage rate, the blastocyst rate and
total cell of blastocysts remained significantly lower in the HSMT
group than in the control group (P < 0.05). Indeed, neither previous
bovine studies nor the present porcine study indicated whether
high-concentration MT supplementation could increase the total
cell of blastocysts. With regards to porcine oocyte maturation,
the ooplasmic maturation is usually evaluated by some molecular
events, bioreaction, or organelle distribution [29], and the nuclear
maturation is estimated by the first polar body expulsed into the
perivitelline space of oocytes [30].
The results of the present study indicated that the first
polar body expulsion rate was greatly increased in the HSMT
group in comparison with that in the HS group, exhibiting no
significant difference relative to that in the control group. We
thus speculated that supplementation with 10 IU/mL Equine
Chorionic Gonadotropin (eCG) and 10 IU/mL human Chorionic
Gonadotrophin (hCG) to the maturation medium was sufficient
for porcine oocyte nuclear maturation in the present experiments,
considering that gonadotropins are responsible for the resumption
of oocyte meiosis and for the promotion of nuclear maturation
by the cAMP/PKA/MAP kinase pathway [31]. Although MT
supplementation could significantly increase the ratio of oocytes
with a uniform mitochondrial distribution, it remained significantly
lower in the HSMT group than in the control group. Several
other researches have demonstrated that HS could influence
abnormal mitochondrial distribution, which is detrimental to
oocyte maturation and development in vitro [32-37]. The data
from production also suggests that the ooplasmic mitochondrial
distribution was poor during the summers, but it improved during
the autumn season [38].
The poor mitochondrial maturation in the HSMT group oocytes
may attribute to the higher intracellular ROS concentration,
considering that ROS is one of the main factors that result in diverse
damages to oocyte maturation and development [39-42]. The
present experiments demonstrated that 10-9 M MT concentration
is insufficient to eliminate the excessive ROS in the oocytes of the
HSMT group. Our study is the first to indicate that the BAX and
BCL-2 were sensitive to both high temperature and MT. As for the
CAT, 10-9 M concentration of MT could not restore its expression to
the control level, which may explain the higher ROS concentrations
in the HSMT group. Estrogen receptor gene is closely associated
with the fertility of female animals and follicular development
and oocyte maturation [43-45]. In our study, we found that the
ER was sensitive to high temperature, but extremely insensitive
to MT exposure. The reduction in ER expression induced the lack
of binding sites for estrogen, which lowers the fertility of female
animals under hot conditions [46]. Recent data in production also
confirmed that the fertility of sow was lower in the summer season
than in other seasons [47]; moreover, past studies in cows have
reported that subcutaneous implantation with 18 mg MT could
only partially alleviate the adverse effects of HS on the reproductive
performance of cows in the hot season [48-50].
Backed by our research, we propose that the reduced secretion
of MT and the insensitivity of ER to MT is most probably related
with the lower fertility of animals in high temperature season.
However, until date, the quantity of MT required to counteract
the adverse effects of HS on pig remain unreported. Based on
our experiments, we believe that much more than the 10-9 M
concentration of exogenous MT would be required. The present
experimental results imply that the lower fertility of animals in high
temperature season can be attributed to the sensitive differences
in the expressions of BCL-2, BAX, CAT, and ER to high temperature
and MT, which provides important insights to the application
of MT toward improving the fertility of animals during the high
temperature season.
Conclusion
BCL-2 and BAX were found to be sensitive to both high temperature and MT exposure; however, CAT and ER, especially the latter, were found to be sensitive only to high temperature and extremely insensitive to MT exposure. The sensitive differences in these genes contributed to poor ooplasmic transfer, lack of nuclear material transfer, and maturation, which further hampered the developmental capacity of porcine oocytes.
Conflict of Interest
The authors declare that they have no competing interests.
Acknowledgment
This work was supported by the Tianjin seed Industry major project (2019ZXZYSN00110), Tianjin “131” Innovative Talents Team (20180338).
References
- Abbott CR, Saxton AM, Rispoli LA, Payton RR, Pohler KG, et al. (2018) An in vivo model to assess the thermoregulatory response of lactating Holsteins to an acute heat stress event occurring after a pharmacologically induced LH surge. Therm Biol 78: 247-256.
- Ahmed JA, Nashiruddullah N, Dutta D, Biswas RK, Borah P (2017) Cumulus cell expansion and ultrastructural changes in in vitro matured bovine oocytes under heat stress. Iranian Journal of Veterinary Research 18(3): 203-207.
- Arias Álvarez M, García RM, López Tello J, Rebollar PG, Gutiérrez Adán A, et al. (2017) In vivo and in vitro maturation of rabbit oocytes differently affects the gene expression profile, mitochondrial distribition, apoptosis and early embryo development. Reprod Fertil Dev 29(9): 1667-1679.
- Alicia S, Athorn RZ, Grupen CG (2018) Anti-Müllerian hormone and Oestradiol as markers of future reproductive success in juvenile gilts. Animal Reproduction Science 195: 197-206.
- Bavister BD, Squirrell JM (2000) Mitochondrial distribution and function in oocytes and early embryos. Hum. Reprod 15(2): 189-198.
- Bonilla AQ, Oliveira LJ, Ozawa M, Newsom EM, Lucy MC, et al. (2011) Developmental changes in thermo protective actions of insulin-like growth factor-1 on the preimplantation bovine embryo. Mol Cell Endocrinol 332(1-2): 170-179.
- Cavallari FC, Leal CLV, Zvi R, Hansen PJ (2019) Effects of melatonin on production of reactive oxygen species and developmental competence of bovine oocytes exposed to heat shock and oxidative stress during in vitro Zygote 27(3): 180-186.
- Camargo LSA, Aguirre Lavin T, Adenot P, Araujo TD, Mendes VRA, et al. (2019) Heat shock during in vitro maturation induces chromatin modifications in the bovine embryo. Reproduction 158(4): 313-322.
- De RF, Scaramuzzi RJ (2003) Heat stress and seasonal effects on reproduction in the dairy cow-a review. Theriogenology 60(6): 1139-1151.
- Dadarwal D, Honparkhe M, Dias FC, Alce T, Lessard C, et al. (2015) Effect of super stimulation protocols on nuclear maturation and distribution of lipid droplets in bovine oocytes. Reprod Fertil Dev 27(8): 1137-1146.
- Dinopoulou V, Drakakis P, Kefala S, Kiapekou E, Bletsa R, et al. (2016) Effect of recombinant-LH and hCG in the absence of FSH on in vitro maturation (IVM) fertilization and early embryonic development of mouse germinal vesicle (GV) stage oocytes. Reprod Biol 16(2): 138-146.
- El Raey M, Geshi M, Somfai T, Kaneda M, Hirako M, et al. (2011) Evidence of melatonin synthesis in the cumulus oocyte complexes and its role in enhancing oocyte maturation in vitro in cattle. Mol Reprod Dev 78(4): 250-262.
- Eyster KM (2016) The Estrogen Receptors: An Overview from Different Perspectives. Methods Mol Biol 1366: 1-10.
- García Ispierto I, López Gatius F, Santolaria P, Yániz JL, Nogareda C, et al. (2006) Relationship between heat stress during the peri-implantation period and early fetal loss in dairy cattle. Theriogenology 65(4): 799-807.
- Gao C, Han HB, Tian XZ, Tan DX, Wang L, et al. (2012) Melatonin promotes embryonic development and reduces reactive oxygen species in vitrified mouse 2-cell embryos. J Pineal Res 52(3): 305-311.
- Gendelman M, Roth Z (2012) Incorporation of coenzyme Q10 into bovine oocytes improves mitochondrial features and alleviates the effects of summer thermal stress on developmental competence. Biol Reprod 87(5): 118.
- Giordano JO, Edwards JL, Di Croce FA, Roper D, Rohrbach NR, et al. (2013) Ovulatory follicle dysfunction in lactating dairy cows after treatment with Folltropin-V at the onset of luteolysis. Theriogenology 79(8): 1210-1217.
- Hung WT, Hong X, Christenson LK, McGinnis LK (2015) Extracellular Vesicles from Bovine Follicular Fluid Support Cumulus Expansion. Biol Reprod 93(5): 117.
- He YT, Wang W, Shen W, Sun QY, Yin S (2019) Melatonin protects against Fenoxaprop-ethyl exposure-induced meiotic defects in mouse oocytes. Toxicology 425: 152241.
- Huang W, Kang SS, Nagai K, Yanagawa Y, Takahashi Y, et al. (2016) Mitochondrial activity during pre-maturational culture in in vitro-grown bovine oocytes is related to maturational and developmental competences. Reprod Fertil Dev 28(3): 349-356.
- Ispada J, Rodrigues TA, Risolia PHB, Lima RS, Gonçalves DR, et al. (2018) Astaxanthin counteracts the effects of heat shock on the maturation of bovine oocytes. Reprod Fertil Dev 30(9): 1169-1179.
- Itami N, Shirasuna K, Kuwayama T, Iwata H (2018) Short-term heat stress induces mitochondrial degradation and biogenesis and enhances mitochondrial quality in porcine oocytes. Journal of Thermal Biology 74: 256-263.
- Khan I, Lee KL, Xu L, Mesalam A, Chowdhury MM, et al. (2017) Improvement of in vitro- produced bovine embryo treated with coagulansin-A under heat-stressed condition. Reproduction 153(4): 421-431.
- Loven DP (1988) A role for reduced oxygen species in heat induced cell killing and the induction of thermotolerance. Med Hypotheses 26(1): 39-50.
- Lord Fontaine S, Averill Bates DA (2002) Heat shock inactivates cellular antioxidant defenses against hydrogen peroxide: protection by glucose. Free Radic Biol Med 32(8): 752-765.
- Li L, Wu J, Luo M, Sun Y, Wang GL (2016) The effect of heat stress on gene expression, synthesis of steroids, and apoptosis in bovine granulosa cells. Cell Stress and Chaperones 21(3): 467-475.
- Martino NA, Marzano G, Mangiacotti M, Miedico O, Sardanelli AM, et al. (2017) Exposure to cadmium during in vitro maturation at environmental nanomolar levels impairs oocyte fertilization through oxidative damage: A large animal model study. Reprod Toxicol 69: 132-145.
- Nabenishi H, Ohta H, Nishimoto T, Morita T, Ashizawa K, et al. (2012) The effects of cysteine addition during in vitro maturation on the developmental competence, ROS, GSH and apoptosis level of bovine oocytes exposed to heat stress. Zygote 20(3): 249-259.
- Nabenishi H, Takagi S, Kamata H, Nishimoto T, Morita T, et al. (2012) The role of mitochondrial transition pores on bovine oocyte competence after heat stress, as determined by effects of cyclosporin. A Mol Reprod Dev 79(1): 31-40.
- Namekawa T, Ikeda S, Sugimoto M, Kume S (2010) Effects of astaxanthin-containing oil on development and stress-related gene expression of bovine embryos exposed to heat stress. Reprod Domest Anim 45(6): e387-391.
- Negron Perez VM, Fausnacht DW, Rhoads ML (2019) Management strategies capable of improving the reproductive performance of heat-stressed dairy cattle. J Dairy Sci 102(12): 10695-10710.
- Opiela J, Katska Ksiazkiewicz L, Lipinski D, Slomski R, Bzowska A, et al. (2008) Interactions among activity of glucose-6-phosphate dehydrogenase in immature oocytes, expression of apoptosis-related genes Bcl-2 and Bax, and developmental competence following IVP in cattle. Theriogenology 69(5): 546-555.
- Prochazka R, Nemcova L (2019) Mechanisms of FSH-and Amphiregulin-Induced MAP Kinase 3/1 Activation in Pig Cumulus-Oocyte Complexes During Maturation In Vitro. Int J Mol Sci 20(5): 1179.
- Pang YW, SunYQ, Sun WJ, Du WH, Hao HS, et al. (2016) Melatonin inhibits paraquat-induced cell death in bovine preimplantation embryos. Journal of Pineal Research 60(2): 155-166.
- Rodrigues TA, Tuna KM, Alli AA, Tribulo P, Hansen PJ, et al. (2019) Follicular fluid exosomes act on the bovine oocyte to improve oocyte competence to support development and survival to heat shock. Reproduction Fertility and Development 31(5): 888-897.
- Rosado Berrios CA, Christian V, Zayas B (2011) Mitochondrial permeability and toxicity of diethylhexyl and monoethylhexyl phthalates on TK6 human lymphoblasts cells. Toxicol In Vitro 25(8): 2010-2016.
- Roth Z (2017) Effect of heat stress on reproduction in dairy cows: Insights into the cellular and molecular responses of the oocyte. Annu Rev Anim Biosci 5: 151-170.
- Roth Z, Hansen PJ (2004) Involvement of apoptosis in disruption of developmental competence of bovine oocytes by heat shock during maturation. Biol Reprod 71(6): 1898-1906.
- Sakatani M (2017) Effects of heat stress on bovine preimplantation embryos produced in vitro. J Reprod Dev 63(4): 347-352.
- Stojkovic M, Machado SA, Stojkovic P, Zakhartchenko V, Hutzler P, et al. (2001) Mitochondrial distribution and adenosine triphosphate content of bovine oocytes before and after in vitro maturation: correlation with morphological criteria and developmental capacity after in vitro fertilization and culture. Biol Reprod 64(3): 904-909.
- Sakatani M, Yamanaka K, Kobayashi S, Takahashi M (2008) Heat shock-derived reactive oxygen species induce embryonic mortality in in vitro early stage bovine embryos. J Reprod Dev 54(6): 496-501.
- Sakatani M, Suda I, Oki T, Kobayashi S, Kobayashi S (2007) Effects of purple sweet potato anthocyanins on development and intracellular redox status of bovine preimplantation embryos exposed to heat shock. J Reprod Dev 53(3): 605-614.
- Sirard MA (2001) Resumption of meiosis: mechanism involved in meiotic progression and its relation with developmental competence. Theriogenology 55(6): 1241-1254.
- Somfai T, Hirao Y (2011) Synchronization of in vitro maturation in porcine oocytes. Methods Mol Biol 761: 211-225.
- Wang F, Tian X, Zhang L, Tan D, Reiter RJ, et al. (2013) Melatonin promotes the in vitro development of pronuclear embryos and increases the efficiency of blastocyst implantation in murine. J Pineal Res 55(3): 267-274.
- Xu Z, Lv MY, Wei HX, Zhang HW, Chen GX, et al. (2019) Effects of Variety, the Seasons of Entering into Swinery and Breeding, First Mating Time on Reproduction Performance of Gilts. China animal husbandry and veterinary 46: 3332-3340.
- Yamanaka KI, Khatun H, Egashira J, Balboula AZ, Tatemoto H, et al. (2018) Heat-shock-induced cathepsin B activity during IVF and culture compromises the developmental competence of bovine embryos. Theriogenology 114:293-300.
- Yang LJ, Zhou M, Huang LB, Yang WR, Yang ZB, et al. (2018) Zearalenone-Promoted Follicle Growth through Modulation of Wnt-1/β- Catenin Signaling Pathway and Expression of Estrogen Receptor Genes in Ovaries of Postweaning Piglets. J Agric Food Chem 66(30): 7899-7906.
- Zhang Y, Yan X, Yang S, Zhang YW, Li Y, et al. (2019) Exposure to Aristolochic Acid I Compromises the Maturational Competency of Porcine Oocytes via Oxidative Stress-Induced DNA Damage. Aging 11(8): 2241-2252.
- Zhao XM, Wang N, Hao HS, Li CY, Zhao YH, et al. (2018) Melatonin improves the fertilization capacity and developmental ability of bovine oocytes by regulating cytoplasmic maturation events. J Pineal Res 64(1): e12445.

 Research Article
Research Article