SUMMARY
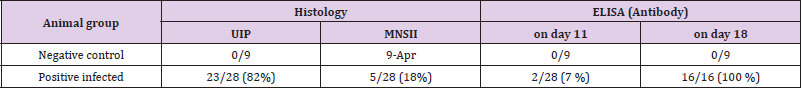
Together 28 piglets (aged over 2 months) were infected with 105 TCID50 of porcine respiratory and reproductive syndrome virus (PRRSV) into both nasal nostrils using a volume of 2x150 (i.e. 300) microliter inoculum. In addition, 9 piglets served as uninfected controls. On day 11 post-infection, tissue samples from tonsillar area, each lung lobe, spleen and liver were taken from 12 sacrificed animals. By day 18, another 16 piglets have been authopsied for tissue sampling. At both intervals, also blood samples were taken for serological examination. For histological examination, the organ samples were fixed in neutral formalin, processed, and embedded into paraffin. Sections were stained either for standard histology or treated with immunohistochemical staining reagents. A commercial monospecific antiserum against the N protein of PRRSV was applied in the first layer and then combined with an alkaline phosphatase labelled second antibody in second layer. An additional slide was treated with the second antibody only for staining control. In 4 out of 9 uninfected (negative control) piglets a slight focal thickening of the peri-bronchial connective tissue and/or of the inter-alveolar septa was noted (a finding referred to as a mild nonspecific interstitial infiltrate, MNSII). This could be clearly distinguished from the usual interstitial pneumonia (UIP) detected in the lungs of 23 out of 28 infected animals (82 %). In the latter, the inter-alveolar septa revealed more widespread mononuclear cell (mainly lymphocyte) infiltration occasionally reaching an extensive intensity. As a rule, the N-protein was found in the bronchial ciliary epithelium cells of nearly all the piglets who developed UIP (21 out of 28, 75 %), along with less frequently positive squamous epithelium at pharyngeal and/or tonsillar areas (13/28, 46 %).
Keywords: Porcine Respiratory and Reproductive System Virus (PRRSV); Usual Interstitial Pneumonia (UIP) [1]; N-Antigen Detection
Introduction
The porcine respiratory and reproduction syndrome virus
(PRRSV) forms small, enveloped particles (50-65 nm in diameter)
harboring a relatively long (approximately 15 kb in size) single
strand RNA genome [2]. The viral RNA (vRNA) is a positive-sense
molecule with terminal cap at 5´-end and a poly-A repeat at 3´-
end [3]. In the course of virus replication, the vRNA is copied as
whole, when synthesized via a full length negative-strand RNA
intermediate. The vRNA sequence begins with 2 (two) long
open reading frames (called ORF1a and ORF1b), which together comprise about 75% of the total genome sequence [4]. This portion
of the genome specifies 14 non-structural proteins (nsps) which
are formed by cleavage of both translated polyproteins. Of special
importance are, for example, two non-structural proteins (nsp9
and nsp12), which function as vRNA replicase, also termed RNAdependent
RNA polymerase (RdRp) [5]. The rest of the genome
encodes 7 structural proteins, out of which 5 are glycoproteins
(designated GP2a/Gp2, GP2b/E, GP3, GP4 and GP5) along with the
M (membrane) protein and the N nucleoprotein [6].
Regarding to the structure of the vRNA, the PRRSV has been
classified as a member of family Arteriviridae (order Nidovirales),
along with the equine arteriitis virus and the lactate dehydrogenase
elevating virus of mice [7]. In the course of vRNA replication, a
total length (genomic) minus strand is generated, which serves
as template for the synthesis of new vRNA molecules. During
viral mRNA synthesis, the negative sense RNA sequence is being
formed first; then a set of positive sense nested sub genomic (sg)
RNA molecules is transcribed. Finally, the full set of minus sense
sub genomic (sg) RNAs is formed, which becomes a template for
the synthesis of functional positive sense sg mRNAs [7]. Both
strands are complementary to each other; the coterminal 3´-ends
are equipped with a common leader sequence at their 5´-ends
[8,9]. The viral genome reveals several (but at least two) conserved
transcription regulatory sequences (TRS), which are located either
in the front of ORF1a (encoding the structural protein GP2a) or
before ORF2a (encoding the envelope glycoprotein Gp2b/E).
The classical PRRSV strains which were isolated in the
US (VR2332) and/or in Europe (Lelystadt) differ at both, by
serological as well as genome examinations [10]. Experimental
infection with the PRRSV isolates can be lethal in newborn and/
or 3-week-old piglets. A key event of the infection process is the
involvement of porcine alveolar macrophages, which are the most
important virus target also mediating virus spread [11]. To date,
at least two macrophage surface molecules are known as entry
mediators: the siglec sialoadhesin and a scavenger receptor CD163
[12]. The PRRSV induced pneumonia is characterized by thickening
of inter-alveolar septa due to infiltration with macrophages and by
the presence of occasional inflammation and cell debris within the
alveoli itself [13]. Also, alveolar pneumocytes of type II may be found
PRRSV antigen positive along with the hyperplasia of peribronchial
lymphatic tissue [14]. The severity of lung lesions may vary from
relatively mild to quite extensive. The viral genotypes can differ
in their pathogenicity, namely the Type 2 North American PRRSV
induces more severe respiratory disease than type 1 European
virus. Nevertheless, mild thickening of interalveolar septum
can be mistaken with focal thickening of inter-alveolar septa in
combination with slight infiltration of peri-bronchial connective
tissue (referred to as mild non-specific interstitial infiltrate, MNSII),
was occasionally seen in a proportion of non-infected control
piglets and interpreted as unrelated to PRRSV infection [15]. In
this paper we describe the correlation of the lung lesions as seen at
histological examination slides stained with, HE on comparison to
the immunohistochemical detection of viral N-protein along with
the results of serological tests for N-protein antibodies.
Materials and Methods
Virus
A North American strain was cultured on the MARC-145 cell line; its titer end point (TCID50) was evaluated using a 96-well plate as described by Zhao, et al. [16] and/or Ramakrishnan (2016) [17].
Animals
Pigs (28 infected animals) were inoculated into both nostrils with 105 TCID50 of above mentioned PRRSV strain administered in a volume of 300μl culture supernatant. The negative control (9) animals were inoculated with a virus free culture medium; these animals were kept under conditions of careful isolation avoiding any contact with the virus-inoculated piglets.
Specimen Sampling and Histological Examination
At given intervals post-infection, the animals were succumbed. Blood was drawn for obtaining serum, whole the tissue samples (coming from each lung lobe, from both tonsils including adjacent paryngeal area, from spleen and liver) were removed and immediately immersed into in 10% neutral formalin for 24hr. Fixed tissue samples were rinsed in phosphate buffer, dehydrated in a series of corresponding reagents and embedded into paraffin as described Szeredi, et al. [18] and/or Stipkovits, et al. [19]. Next to sectioning, the sections were stained either by classical hematoxylin and eosin (HE) and/or treated by immunohistochemical reagents, namely using a commercial anti-PRRSV N-protein antibody mixture SDOW-17 and SR-30 in the first layer. This has been purchased from 4rtilab (SDOW17-A and SR30-A, respectively) and mixed for use in an equal 1:1 ratio.
ELISA Titer Measurements
The specific serum class IgG antibody levels against the PRRSV N antigen were determined using the INgezim PRRS 2.0 ELISA kit (purchaed from Eurofins) strictly following the procedure recommended by the manufacturer.
Saliva Collection
The pooled oral fluid was collected from each animal separately using the Civtest suis oral fluid rope IDEXX. Obtained saliva samples we examined for the presence of class IgA specific antibody to the N-antigen of PRRSV. The antibody test was performed with the Oral Fluids (IDEXX PRRS OF) kit as recommended in the manufacturer’ s manual.
Results
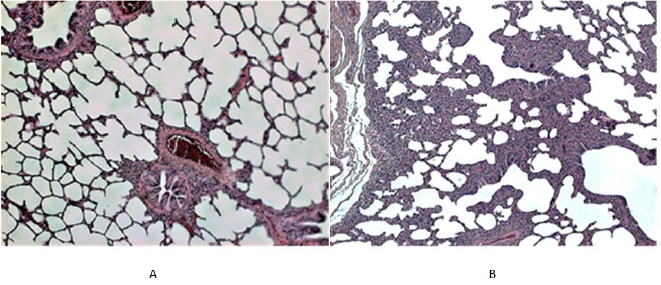
As documented in Figure 1A the interalveolar septi in normal lung tissue are very thin in order to ensure the diffusion of oxygen into blood capillaries, where the erythrocytes circulate. Occasionally, a few mononuclear cells (mainly lymphocytes) might be seen in the peribronchial connective tissue. Surprisingly, in 4 out of 9 uninfected (control) piglets, a slight focal thickening of interalveolar septi was noted along with the accumulation of relatively few interstitial infiltrates consisting of mononuclear cells, mainly lymphocytes (Figure 1B). As expected, staining for the N-antigen of PRRSV in the control lung tissue was negative by all control animals, including the above-mentioned areas in which the above mentioned mild interstitial infiltrate (MNSII) has been detected. The non-extensive MNSII (Figure 2A) has been also found in a few infected piglets (5 out of 28, 18 %), especially when the N-protein could not be detected (Table 1). As expected, in the majority of infected animals (23 out of 28) the lung tissue revealed a typical picture of usual interstitial pneumonia (UIP). In the latter, the interalveolar septi were thicker due to the presence of a rich mononuclear cell infiltrate. In UIP cases, the capillaries were widened along with occasional focal bleeding in result to the injury of endothelial cells (Figures 2B & 2C). High power view of such areas showed that the interstitial infiltrate in question consisted mainly of lymphocytes (Figure 2D). Occasionally (for example in piglet no. 40) the infiltrate was so extensive that it altered the original lung structure (Figure 3).
Figure 1: Histological picture of the lung tissue in uninfected (control) piglets.
A. In the left (piglet no. 5). The normal lung structure at low power view shows thin interalveolar septa devoid of any infiltrate;
in the peribronchial (and/or perivascular) connective tissue a few mononuclear cells (mainly lymphocytes) can be seen.
B. In the right (piglet no. 2). Unlike to 1A, this Figure shows areas of thickened interalveolar septa due to the accumulation of
monocellular cells (mainly of lymphocytes). Such focal mild non-specific interstitial infiltrate (MNSII) was found in the lungs
of 5 out of 9 uninfected controls (Table 1).
Table 1: Survey of histological lesions and the N-antigen presence in infected piglets.
Note: *Mild non-specific interstitial infiltrate (in the peribronchial area and/or interalveolar septa); ** Severe interstitial infiltrate corresponding to the diagnosis of “Usual Interstitial Pneumonia” [1].
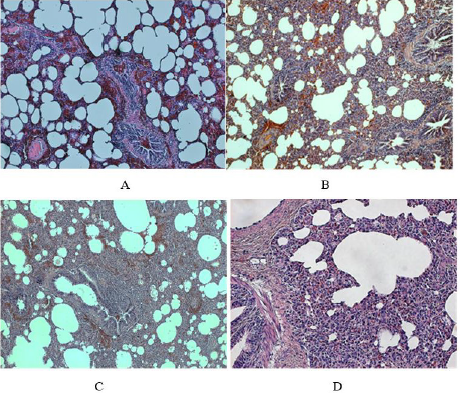
Figure 2: Histological findings in the lungs of PRRSV infected piglets.
A. In the left above (piglet no. 16). At low power view some areas of the lung tissue even in the infected animal showed rather
less extensive thickening of interalveolar septa (infiltration by mononuclear cells referred to as mild non-specific interstitial
infiltrate, MNSII); note the dilatation of small vessels (magn x100).
B. In the right above (piglet no. 16). In contrast to the area shown above, another lung area of the same reveals typical UIP with
more widespread thickening of interalveolar septa and their abundant mononuclear cell infiltration (along with hyperemia, i.e.
dilatation of capillaries and small blood vessels).
C. In the left below (piglet no. 25). The lung tissue of an animal who developed typical UIP shows widespread mononuclear
infiltration of interalveolar septa and peribronchial connective tissue (a lymphatic follicle like structure can be seen, magn.
x100).
D. In the right below, the same piglet as above (no. 25). The mononuclear infiltrate in the peribronchial area consists mainly of
lymphocytes along causes thickening of interalveolar septa (magn. x240).
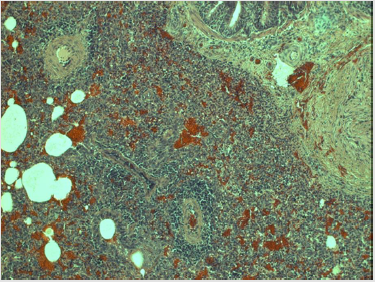
Figure 3: Extensive interstitial pneumonia in PRRSV infection.
Note: Lungs of the piglet no. 40 show severe interstial pneumonitis: the infiltration of interalveolar septa by mononuclear
cells (mainly lymphocytes) is so widespread that the original lung structure can be hardly seen. In addition, the abundant
hyperemia along with extensive proliferation of connective tissue is clearly visible (magn. 220x).
Staining with the anti-N antibody showed the presence of PRRSV antigen predominantly in the columnar ciliary epithelium lining the bronchial tree (Figure 4A). Details from such areas also demonstrated the presence of viral antigen in the cytoplasm of the small acinary mucous glands situated below the ciliary epithelium lining, namely in the connective tissue of bronchial wall (Figure 4B). The alveolar lining was rarely positive, though occasionally the type II alveolar cells could harbor the N-protein, mainly present in alveolar macrophages moving from the alveolar space across the thickened interalveolar septa into local lymphatic capillaries and/ or to the sinuses of regional lymph nodes (Figure 4C), where virus was finally deposited. Nevertheless, in some piglets the local lymph nodes did not stop the virus spread, which then might reach the spleen and/or liver via blood stream. The reticular cells of regional sinuses in spleen were found positive as well, and occasionally the antigen could be seen also in lymphatic follicles (Figure 4D).
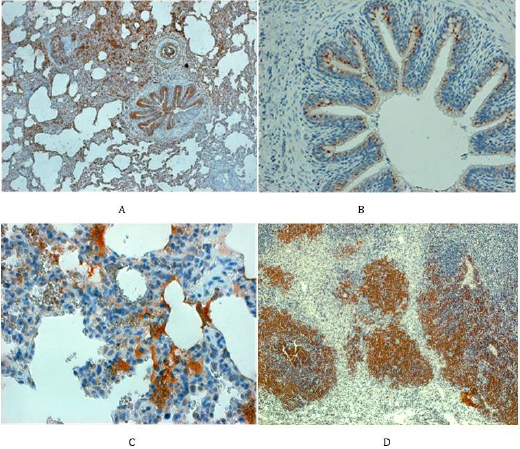
Figure 4: Staining for N-antigen in the respiratory pathway and spleen
A. In the left above (piglet no. 34). The lungs of infected animals reveal overwhelming positive staining for N-protein, namely
in the bronchial epithelium, in parabronchial mucinous glands and occasionally in the flat epithelium cells lining the aveoli
(magn. 80x).
B. In the right above (piglet no. 12). The N-protein can be seen in the cytoplasm of ciliary epithelium cells lining the bronchi
along with the negative goblet cells (magn. 120x).
C. In the left below (piglet no. 45). The N-protein can be seen in the cytoplasm of cells lining the alveolar wall and in mononuclear
phagocytes which infiltrate the interalveolar septi (magn. 400x).
D. In the right below (animal no. 44): the spleen showing lymphatic follicles consisting mainly of lymphocytes positive for the
N-protein (magn.x120).
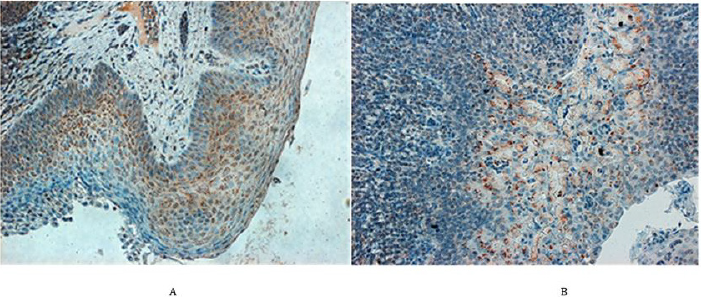
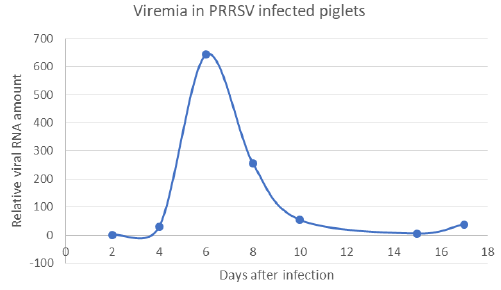
Outside of lung tissue, the N-protein of PRRSV was found especially in the non-hornified squamous epithelium of the pharyngeal area including that over tonsil (Figure 5A). Here the virus antigen occupied the deeper layers of stratified epithelium, namely the multiplying parabasal cells as well as those in the medium layer. The virus was also found in the salivary glands as documented in submandibular gland, which acinar cells harbored the N-antigen in their cytoplasm (Figure 5B). While the tonsils and/ or pharyngeal epithelium were involved relatively frequently, the presence of the virus in salivary glands acinar cell was relatively rare. Nevertheless, the real incidence of given antigen in the salivary glands was difficult to assess, since such tissue has appeared in the sections examined just by chance. The development of antibody response in comparison with lung lesions as detected by ELISA in serum samples is documented on Table 2. This shows that the virusspecific antibodies were rarely detected on day 11 post-infection but were frequently found on day 18. This may not be surprising and can be explained by the viremia (Figure 6), which peaked on day 6 post-infection, but was absent by day 10. Interestingly enough, on next day 11, the ELISA could not detect free serum antibodies, probably because they were bound to virus particles. However, the specific antibodies were clearly detected at later intervals (i.e. by day 18), when their levels in the serum increased.
Figure 5: N-protein in the pharyngeal area of PRRSV infected animals.
A. In the left (piglet no. 26). In the tonsillar squamous epithelium, the N-protein is expressed mainly wthin cytoplasm of
actively growing cells of the suprabasal and intermedial layers including a few basal epithelium cells (magn. 220x)
B. In the right (pig no. 13). N-protein can be seen in the acini of a submandibular salivary gland as well as in the marginal sinus
of adjacent lymph node (magn. 220x).
Figure 6: Presence of vRNA in the serum of infected pigs (viremia has been detected on days 6 and 8 post-infection).
Table 2: The comparison of UIP with serological response.
Discussion
The PRR syndrome in piglets is characterized with high
mortality, reproductive failure (late-term abortions and stillbirths,
premature farrowing, mummified pigs in pregnant sows) and a
severe respiratory disease (interstitial pneumonia). The disease
occurring in the nursery and among growing/finishing piglets
causes significant economic losses to the swine industry worldwide.
The corresponding virus (PRRSV) replicates mainly in the porcine
alveolar macrophages (PAMs) and dendritic cells (DCs) [20]. The
virus also causes persistent infection eliciting antibody dependent
enhancement (ADE) and occasional immunosuppression. Being
a member of the family Arteriviridae, it belongs to the order
Nidovirales together with the Coronaviridae and Roniviridae
families [21]. PRRSV was originally divided into European type 1
and North American type 2 genotypes. Later on, the East European
PRRSV isolates have been found to be of the European genotype but
forming different subtypes. A novel virus, namely the Belarusian
strain Lena, has been recently characterized as a highly pathogenic
East European subtype 3, which differs from European subtype
1 Lelystad and North American US5 strains at genetic as well as
antigenic levels [22].
Numerous results suggest that PRRSV may utilize multiple
strategies of replication and spread in the infected pigs, including
subversion of the host innate immune response, inducing an antiapoptotic
and anti-inflammatory state as well as developing ADE.
The PRRSV induced immunosuppression might mediate apoptosis
of infected cells, which causes depletion of immune cells and induces
an anti-inflammatory cytokine response due to which the host is
unable to eradicate the primary infection. The initial antibodies
do not confer protection and can even be harmful by mediating an
antibody-dependent enhancement (ADE), since they can facilitate
the virus entry of into targets cells in vitro. To characterize the
humoral immune response direct enzyme-linked immunosorbent
assays (ELISA) can be used including different mainly recombinant
PRRSV antigens. For example, the kinetics of antibody responses
directed against nonstructural virus coded proteins (nsp) can be
analysed in pigs experimentally exposed to the virus [23]. In such
case, high antibody reactivities especially against nsp1, nsp2, and
nsp7 were noted. Among the latter, nsp7 recombinant proteinbased
ELISA showed good sensitivity and specificity most suitable
for diagnostic development especially for identification and
differentiation of type 1 and type 2 PRRSV. Several non-structural
proteins (such as nsp1, nsp2, nsp5, nsp7 nsp9, nsp10 and nsp11)
have been implicated in the induction of IFN-γ and also in the
development of the cell-mediated immune response [24]. On other
hand, the induction of neutralizing antibodies (NAs) may be delayed
and/or their levels may remain low, which is not only the problem of
early diagnostic, but is also of importance regarding effective virus
elimination. NAs may protect against disease if present in sufficient
quantities before infection, but they do not seem to be essential for
clearing virus in blood during the course of the infection. PRRSV is
able to modulate innate responses, probably through the regulation
of IFN-α and IL-10 responses [25].
As described, PRRSV replicates predominantly in the lung
alveolar macrophages, can induce prolonged viremia, and cause
persistent infections lasting for months after initial infection.
PRRSV strongly modulates the host’s immune response and
changes its gene expression. Studies showed that PRRSV inhibits
type I interferons (IFN-β). Regarding cell-mediated responses,
development of PRRSV-specific gamma interferon-secreting cells
(IFN gamma-SC) and interleukin 4-secreting cells (IL4-SC) in
PBMC was examined by ELISPOT assay. Using this technic, no IFN
gamma-SC was detected until day 14 p.i., whereas for IL4-SC, such
differences were not seen. Concurrently with the onset of viremia
and the development of clinical signs, serum haptoglobin levels
and interleukin 10 (IL10) in PRRSV-stimulated PBMC-culture
supernatants increased significantly. These results are compatible
with the model of pathogenesis in which the immune response
does not fully control the outcome of infection [26].
The PRRSV replication and its spread in the body subverts the
host innate immune response as well when high jacking its lipid
metabolism and inducing an anti-apoptotic and anti-inflammatory
state. The latter is indicated by suppressing the expression of serine
proteinase inhibitor 2 (SPI 2), IFN-α, and down-regulation of the
expression of pro-apoptotic genes such as B-cell lymphoma 2 (BCL-2)
antagonist/killer (BAK) and the BCL-2 associated X (BAX). Whereas
BAX resides predominantly in the cytosol, BAK is constitutively
localized to the outer mitochondrial membrane; both form toxic
mitochondrial pores in response to cellular stress. Furthermore,
the APR-1, i.e., the Adenomatous polyposis coli (APC) protein which
is a Wnt signaling component along with a microtubule-associated
protein SARP3 (several ankyrin repeat protein 3), may be downregulated.
Both were shown to interact with all isoforms of PP1
(protein phosphatase 1). Infections of N-PRRSV viruses resulted in
fever and inflammatory response, as indicated by high expression of
proinflammatory cytokines and chemokines, adhesion molecules,
inflammatory enzymes and their receptors, such as IL-1β, IL8, SELL,
ICAM, CCL2, CXCL9, CXCL10, B2M, proteasomes and cathepsins.
This was compounded by cell death and elevated expression of
NFKBIA, XAF1, GADD45A, perforin, granzymes, and cytochrome C,
coupled with increased ROS-mediated oxidative stress, as indicated
by up-regulated expression of cytochrome b245. Taken together,
the N-PRRSV infection may have resulted in an excessive immune
and inflammatory response that contributed to tissue damage [27].
Conflict of Interest
No conflict of interest with any institution/organization.
References
- (1994) For the expression “Usual Interstitial Pneumonia” see “The respiratory system”, In: Rubin E, JL Farber (Eds.)., Pathology (2nd)., IP Lippincott Company, Philadelphia, pp: 556-617.
- Spilman MS, Welbon C, Nelson E, Dokland T (2009) Cryo-electron tomography of porcine reproductive and respiratory syndrome virus: organization of the nucleocapsid. J Gen Virol 90: 527-535.
- Arteriviridae (1999) In: Murphy AF, Gibbs EPJ, Horzinek MC and Studert MJ (Eds.)., Veterinary Virology (3rd)., Academic Press, pp: 509-516.
- Lalit Beura (2011) Role of porcine reproductive and respiratory syndrome virus nonstructural protein 1, in: Viral replication and pathogenesis, Dissertations & Theses in Veterinary and Biomedical Science, University of Nebraska-Lincoln, pp. 190.
- Conzelmann KKN Visser, Van Woensel P, Thiel HJ (1993) Molecular characterization of porcine reproductive and respiratory syndrome virus, a member of the arterivirus group. Virology 193: 329-339.
- Snijder EJ,Meulenberg JMM (2001) Arteriviruses. In: Knipe DM and PM Howley (Eds.)., Fields Virology vol. 1, (4th )., Lippincott Wiliam and Wilkins, Walter Kluwer Comp, Philadelphia, pp: 1205-1220.
- (2017) International Committee on Taxonomy of Viruses (ICTV).
- Snijder EJ, Meulenberg JJM (1998) The molecular biology of Arteriviruses. J gen Virol 79: 961-979.
- Pasternak AO, Van den Born E, Spaan WJ, Snijder EJ (2001) Sequence requirements for RNA strand transfer during nidovirus discontinuous subgenomic RNA synthesis. Embo J 20: 7220-7228.
- Katz JB, Shafer AL, Eernisse, KA, Landgraf, JG, Nelson EA (1995) Antigenic differences between European and American isolates of procine reproductive and respiratory syndrome virus (PRRSV) are encoded by the carboxy-terminal portion of viral open reading frame 3. Vet Microbiol 44: 65-76.
- Lawson SR, Rossow KD, Collins JE, Benfield DA, Rowland RR (1997) Porcine reproductive and respiratory syndrome virus infection of gnotobiotic pigs: sites of virus replication and co-localization with MAC-387 staining at 21 days post-infection. Virus Res 51: 105-113.
- Van Gorp H, Van Breedam W, Delputte PL, Nauwynck HJ (2008) Sialoadhesin and CD163 join forces during entry of the porcine reproductive and respiratory syndrome virus. J Gen Virol 89: 2943-2953.
- Rossow KD (1998) Porcine Reproductive Respiratory Syndrome. Review Article. Vet Pathol 35: 1-20.
- Rossow KD, Benfield DA, Goyal SM, Nerlson EA, Christopher-Hennings J, et al. (1996) Chronical immunohistochemical detection of the porcine reproductive and respiratory syndrome virus infection in gnotobiotic pigs. Vet Pathol 33: 551-556.
- Martínez-Lobo FJ, Díez-Fuertes F, Segalés J, García-Artiga C, Simarro I, et al. (2011) Comparative pathogenicity of type 1 and type 2 isolates of porcine reproductive and respiratory syndrome virus (PRRSV) in a young pig infection model. Vet Microbiol 154: 58-68.
- Zhao C, Liu S, Li C, Yang L, Zu Y (2014) In vitro evaluation of the antiviral activity of the synthetic epigallocatechin gallate analog-epigallocatechin gallate (EGCG) palmitate against porcine reproductive and respiratory syndrome virus. Viruses 6(2): 938-950.
- Ramakrishnan MA (2014) Determination of 50% endpoint titer using a simple formula. Viruses 6: 938-950. World J Virol 5(2): 85-86.
- Szeredi L, Molnár T, Glávits R, Takai S, Makrai L, et al. (2006) Two cases of equine abortion caused by Rhodococcus equi. Vet Pathol 43(2): 208-211.
- Stipkovits L, Glavits R, Palfi V, Beres A, Egyed L, et al. (2012) Pathologic lesions caused by coinfection of Mycoplasma gallisepticum and H3N8 low pathogenic avian influenza virus in chickens. Vet Pathol 49(2): 273-283.
- Shuqi Xiao, Jianyu Jia, Delin Mo, Qiwei Wang, Limei Qin, et al. (2010) Understanding PRRSV Infection in Porcine Lung Based on Genome-Wide Transcriptome Response Identified by Deep Sequencing. PLoS ONE 5 (6): 11377.
- Cavanagh D (1997) Nidovirales: a new order comprising Coronaviridae and Arteriviridae. Arch Virol 142: 629-633.
- Karniychuk Uladzimir U, Marc Geldhof, Merijn Vanhee, Jan Van Doorsselaere, Tamara A Saveleva, et al. (2010) Pathogenesis and antigenic characterization of a new East European subtype 3 porcine reproductive and respiratory syndrome virus isolate. BMC Veterinary Research 6: 30-39.
- Elizabeth Brown, Steven Lawson, Craig Welbon, Josephine Gnanandarajah, Juan Li, et al. (2009) Rowland and Ying Fang Antibody Response to Porcine Reproductive and Respiratory Syndrome Virus (PRRSV) Nonstructural Proteins and Implications for Diagnostic Detection and Differentiation of PRRSV Types I and II. Clinical And Vaccine Immunology 16(5): 628-635.
- Edgar Rascón-Castelo, Alexel Burgara-Estrella, Enric Mateu, Jesús Hernández (2015) Immunological Features of the Non-Structural Proteins of Porcine Reproductive and Respiratory Syndrome Virus. Viruses 7: 873-886.
- Mateu E, Diaz I (2008) The challenge of PRRS immunology. Vet J 177: 345-351.
- Dıaz I, L Darwich, G Pappaterra, J Pujols, Mateu E (2005) Immune responses of pigs after experimental infection with a European strain of Porcine reproductive and respiratory syndrome virus. J Gen Virol 86: 1943–1951.
- Kimman TG, Cornelissen LA, Moormann RJ, Rebel JM, Stockhofe- Zurwieden N (2009) Challenges for porcine reproductive and respiratory syndrome virus (PRRSV) vaccinology. Vaccine 27: 3704-3718.

 Research Article
Research Article