Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Ion Munteanu, Elena Starodub and Nicolae A Enaki*
Received: April 24, 2023; Published: May 05, 2023
*Corresponding author: Nicolae A Enaki, Quantum Optics and Kinetic Processes Lab, Institute of Applied Physics of Moldova, Chisinau, MD 2028, Republic of Moldova
DOI: 10.26717/BJSTR.2023.50.007920
The rate of disinfection or decontamination using ultraviolet (UV) radiation needs quick information about the dose of applied radiation and the level of inactivation of pathogens. By using spectroscopy analysis methods, we observe the strong correlation between the decontamination rate and absorption spectra of yeast solution was find. This relationship between the spectrum and the pathogen decontamination rate can be successfully used in diagnostics. The application of this method can be used in the diagnosis of pathogens such as Candida Albinus, which is more resistant than a class of viruses and bacteria, including COVID-19. The effects of UV radiation with a 254 nm wavelength on the molecular weight of yeast fungi are observed and analyzed in this communication.
Keywords: UV-C Radiation; Biomolecules; Pathogen; Absorbance Spectrum; Decontamination
The interaction of ultraviolet radiation with viruses is a theme already known, but as a result of the pandemic period, the problem of obtaining new methods of inactivation of viruses such as SARS-COV2 (Covid-19) has been raised. One major concern in these pandemic times is the implementation of new opportunities to increase the efficiency of the decontamination rate of fluids. Numerous studies were recently dedicated to the application under an appropriate distribution of UV-C radiation for the decontamination of fluids and surfaces in order to destroy pathogens (microbes and viruses), such as SARSCov2 [1-4]. Ultraviolet radiation has a shorter wavelength than visible light, and the wavelength range of ultraviolet radiation is typically defined as 100-400 nm, and it is further divided into three subcategories: UV-A (315-400 nm), UV-B (280- 315 nm), and UV-C (100-280 nm). UVC radiation, which has the shortest wavelength (UV-C) and the highest energy, is the most effective at killing or inactivating microorganisms, making it a popular choice for disinfection and decontamination Ref. [5-7]. The research in this domain demonstrates the direct action of DNA denaturation induced by UV-C photons on the nucleic acid base that inactivates the virus Ref. [8-11]. In this paper, we decided to use the yeast colonies for study due to their eukaryotic cellular constructions, which ensure better resistance to UV-C radiation, compared to pathogenic procaryotic colonies. Indeed, the remnant populations are, in fact, a major form of microbial existence in natural settings that provide cells with the ability to defend effectively against environmental attacks and to adapt/survive long periods and other stresses.
Yeast fungi are single-celled microorganisms, eukaryotic, and can be found in various environments including soil, water, and on the surfaces of fruits and vegetables. Some species of yeast can cause infections in humans, particularly in people with weakened immune systems, such as candidiasis. Can form biofilms: Yeasts such as Candida albicans can form biofilms, which are communities of microorganisms that are bound together by a matrix of extracellular polymeric substances (EPS). Biofilms can be difficult to remove and may act as a reservoir of infectious organisms. These infectious organisms can be killed or inactivated by various methods: such as heat, chemical agents, and ultraviolet (UV) radiation, but the use of UV radiation for yeast disinfection is not common. In this research, we specifically used these yeast fungi, because, molecularly, they are very similar to the molecular structure of viruses with an increased risk of infection. The aim of this study was to investigate the effect of UV- irradiation (254 nm) on the molecular sizes of yeast fungi and on their surface properties. The changes in chemical structure were studied using UVVIS spectroscopy. The sample morphology was observed in optical microscopy.
In this section, we propose to experimentally study the bacterial decontamination rate as a function of time, and at each time interval, the UV action spectrum affecting the infection process will be determined. By using spectroscopy analysis methods, it observes the strong correlation between the decontamination rate and absorption spectra of yeast solution. The fungal solution of yeast has many analogies with different dangerous fungi such as Candida Albinus and is more resistant than a class of viruses and bacteria, including COVID-19. It is known that DNA is a long polymer made from repeating units called nucleotides (cytosine [C], guanine [G], adenine [A], or thymine [T]), a sugar called deoxyribose, and a phosphate group. (Figure 1A) The structure of the DNA double helix. (B) Each of the four nucleotide bases has a slightly different absorption spectrum, and the spectrum DNA is the average of them (Source from literature). Wavelengths in the UVC range are especially damaging to cells because they are absorbed by nucleic acids, see (Figure 1). Many pathogens (viruses and bacteria) are known to be more sensitive to UVC radiation than eukaryotic cells due to the double DNA protection through the cell membrane and the latter’s nucleic acid. UV radiation inactivates microorganisms by causing cross-links between constituent nucleic acids. The absorption of UV can result in the formation of intrastrain cyclobutyl-pyrimidine dimers in DNA, which can lead to mutations or cell death [12]. The predominant role played by pyrimidine dimers is shown by the C→T transversion at TC sites and CC→TT tandem mutations, considered as the specific signatures of UV-induced DNA damage (Figure 2). (Figure 2) Thymine dimers are caused by UV absorption in adjacent nucleotides (thymine doublets). There are many papers in the literature that demonstrated that thymine dimers produced by UV exposure result in the inactivation of bacteria and DNA viruses. A UV dose of 10 J/m2 produces 1.279×1019 photons per m2. A virus of 0.1-micron diameter has a cross-sectional area of 3.14×10-14 m and will be subject to the passage of about 401,000 photons when exposed to 10 J/m2, which is sufficient to highly inactivate most viruses. For example, a dose of 4.5 J/m2 is reported to cause 50,000 pyrimidine dimers per cell [13]. (Figure 3). UV excitation populates the singlet state ππ*, which decays into the singlet nπ* state.In the realization of the proposed objectives, we chose yeast colonies for study because of their eukaryotic cellular constructions, which ensure better resistance to UVC radiation as compared to pathogen prokaryotic cellular colonies. As this, for the proposed experiments we use Baker’s yeast solutions instead of dangerous contaminated fluids. In fact, yeast has stronger resistance to UVC radiation incomparison with many viruses or bacteria. The yeast solutions used by us belong to Saccharomyces cerevisiae species with the property to form colonies, according to existing investigation [7- 10]. The method of preparing the yeast solution is shown in (Figure 4) and described below. We will dissolve 10 g fungal yeast in 200g water, and to improve the development of fungi we will add 10 g sugar. At the initial stage of the development of fungi, we observed the appearance of various numbers of yeast fungus colonies with aleatory dimensions. To have a better effect, the water used to prepare the solution must be around 30 degrees Celsius. In some time, approximately 10-20 minutes, the first yeast fungi can be observed under the microscope. For the experiment, we left the yeast cells to multiply for 24 hours, so that we could observe some changes after UVC irradiation. Such an experiment with the inactivation of fungal colonies in the earlier time development interval of about 15 min was the subject of our Ref. [13-15]. (Figures 4A & 4B) At the bottom of the container are pieces of fresh Baker’s yeast with sugar. (B) Baker’s yeast solution done to be used. The experimental design of laboratory tests was constructed to investigate the UVC dose dependence of the inactivation rate for yeast colonies, which is illustrated in (Figure 5).
(Figure 5). The experimental design of laboratory tests. The 254 nm pulsed UVC radiation penetrates directly through one of the holes in the cuvette holders to Baker’s yeast solutions in 5-25 min time intervals. Every five minutes, the ultraviolet radiation is stopped to make spectral measurements to see the dynamics of the decontamination of the yeast solution in the absorption spectra. Also, in these time intervals, pictures will be taken using an electron microscope. The images obtained under the microscope and the absorption spectra of the five spectra obtained at different time intervals are shown in (Figure 6) and the relationship between the spectral analysis obtained in this experiment and the number of bacteria on each image will be described in more detail in the results and discussions. (Figure 6). The decontamination rate of the yeast solution under the action of short pulses of UVC radiation as a function of time. In the current section, we determined the spectrum of UV action on the infection process and analyzed the dependence of the bacteria’s decontamination speed on time.
Figure 1 A. The structure of the DNA double helix. B. Each of the four nucleotide bases has a slightly different absorption spectrum, and the spectrum DNA is the average of them (Source from literature).
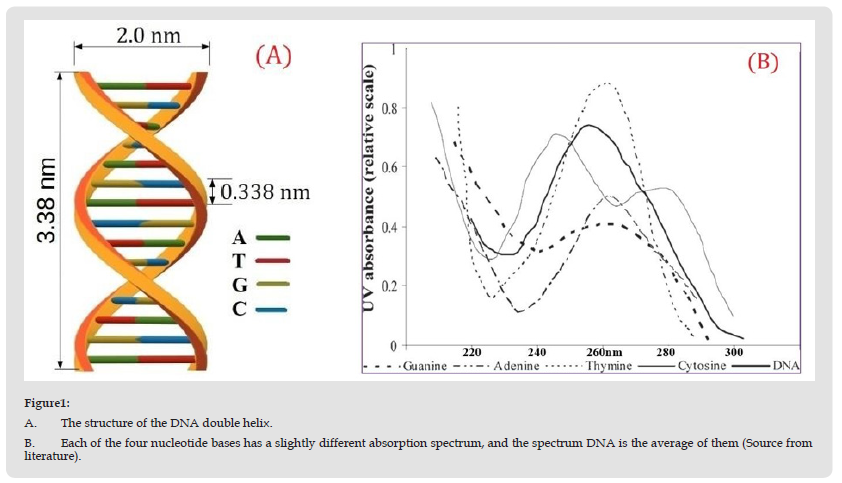
Figure 4 A. At the bottom of the container are pieces of fresh Baker’s yeast with sugar. B. Baker’s yeast solution done to be used.

Figure 6 The decontamination rate of the yeast solution under the action of short pulses of UVC radiation as a function of time.

Studying the absorption spectra for different doses of pulsed radiation, we observe equivocal evidence between the number of colonies observed under the microscope after measuring the absorption spectrum for each sample. A decrease in the absorption coefficient is observed compared to the duration of time under the influence of UVC see (Figure 6). For example, exposure to 5-minute pulses gives an insignificant decrease in the absorption spectrum in the UVC range (220-280), viewing under a microscope has the same tendency. In this short time, the number of colonies changes insubstantial. A completely different picture is observed at 25 min. Here the absorption coefficient drops substantially and is accompanied by a substantial decrease in the number of colonies (Figure 7A). (Figures 7A & 7B) The inactivation of fungus from the yeast solution in the static regime under the direct UVC exposition during the time intervals: 5 Min, 10 Min, 15 Min, 20 Min, and 25 Min. (B) Yeast decontamination rate as a function of time duration of UVC radiation application These correlations allow the use of spectroscopy to quickly highlight the degree of inactivation of the pathogenic agent, in our case yeast fungi. The repetition of such experiments on pathogens in the laboratory is not allowed, but it is possible to find correlations according to the literature. According to the literature Ref. [16-18], yeast fungi have the same resistance to UVC as Candida Albicans fungi and are much more resistant than other bacteria or viruses (including Covid-19). The correlation between the spectrum and decontamination rate may be used in diagnostics. We proposed investigating the effect of UV irradiation and obtaining the absorption spectrum in order to highlight the degree of inactivation of the pathogen.
Figure 7 A. The inactivation of fungus from the yeast solution in the static regime under the direct UVC exposition during the time intervals: 5 Min, 10 Min, 15 Min, 20 Min, and 25 Min. B. Yeast decontamination rate as a function of time duration of UVC radiation application

The dose of ultraviolet radiation applied to inactivate pathogens (viruses, bacteria, etc...) requires special attention in the last period of time. That is why in this paper the main idea was to establish a correlation between the disinfection or decontamination rate with the absorption spectrae of the yeast solution obtained at different time intervals. By using spectroscopy analysis methods in this paper, it can be said that UV irradiation can be considered a good method for modifying the structures of yeast fungi. Experiments have conclusively demonstrated that the correlation between the decontamination rate of UVC radiation and the absorbance spectrum of different infected solutions can be used in diagnostics.
The authors don’t have the financial, commercial, legal, or professional relationship with other organizations, or with the people working with them, that could influence their research.
This paper is supported by the projects: No. 20.80009.5007.01 and NATO EAP SFPP 984890.


