Abstract
Objective: To investigate the mechanism of the effects of total flavonoids of Rhizoma Drynariae in different concentrations on synovial inflammation in rheumatoid arthritis, which is expected to provide experimental evidence for clinical application.
Methods: Serum carrying total flavonoids of Rhizoma Drynariae was used to treat the in vitro culture of synovial cells isolated from patients with rheumatoid arthritis. Cells were identified and passaged before the cell treatment. The experimental groups included the 20% blank serum group, the 20% high-dose total flavonoids of Rhizoma Drynariae medicated serum group, 20% low-dose total flavonoids of Rhizoma Drynariae medicated serum group, and the 20% tripterygium glucosides medicated serum group. With the serum carrying tripterygium glycosides as a positive control, the drug-carrying serum at a concentration of 20% was added to the third-generation synovial cells, and the cells were continuously cultured.
Results: Compared with the blank control serum, the sera carrying total flavonoids of Rhizoma Drynariae could significantly inhibit the expression of proliferating cell nuclear antigen (PCNA) mRNA and Bcl-2 mRNA in fibroblast-like synoviocytes (FLSs) (P<0.0001), and the effect of the treatment at a high dose was more significant.
Conclusion: Total flavonoids of Rhizoma Drynariae may play a role in inhibiting the proliferation of FLSs by regulating the expression of PCNA mRNA and Bcl-2 mRNA in FLSs.
Keywords:Rheumatoid Arthritis; Synovial Inflammation; Fibroblast; Rhizoma Drynariae
Abbreviations: RA: Rheumatoid Arthritis; FLSs: Fibroblast-Like Synoviocytes; ELISA: Enzyme-Linked Immunosorbent Assay; DMSO: Dimethyl Sulfoxide; PCNA: Proliferating Cell Nuclear Antigen
Introduction
Rheumatoid arthritis (RA) is a common chronic systemic autoimmune disease. Currently, it is generally believed that the dysfunction caused by RA, cartilage damage, and bone invasion are closely related to the lack of apoptosis of fibroblast-like synoviocytes (FLSs), and insufficient apoptosis is an important cause of most tumors and autoimmune diseases [1]. Therefore, inhibition of the apoptosis of FLSs can effectively treat RA [2]. Drynaria is the dry rhizome, which is belonged to epiphytic fern, keel subject. Drynaria is also named as monkey ginger or turmeric. The efficacy is for reinforcing kidney, strengthening bone and promoting blood circulation and removing blood stasis. Previous studies have shown that total flavonoids of Rhizoma Drynariae have anti inflammatory and osteogenesis effects [3]. However, the effect of total flavonoids of Rhizoma Drynariae on the treatment of RA is rarely reported. In this study, the effect of total flavonoids of Rhizoma Drynariae on RA was observed.
Materials and Methods
Materials
Experimental Animals and Synovial Tissues: Twenty SPFgrade female Wistar rats (200±10g) were purchased from Shanghai Experimental Animal Center of the Chinese Academy of Sciences; the license number of animal use was SYXK (Shanghai) 2008 0050. The synovial tissue was obtained from the Department of Joint Surgery of Guanghua Hospital affiliated with Shanghai University of Traditional Chinese Medicine. All patients were female, with a mean age of 48.23±5.36 years and a disease duration of 7±11.45 years. The diagnosis was in accordance with the diagnostic criteria for RA in ACR/EULAR in 2009. This study was reviewed and approved by the Ethics Committee of Guanghua Hospital affiliated with Shanghai University of Traditional Chinese Medicine.
Main Drugs: The following drugs were used in this study: total flavonoids of Rhizoma Drynariae (Qianggu Capsule, Specifications: 0.25g×12, Beijing Qihuang Pharmaceutical Co., Ltd.) and tripterygium glycoside tablets (Specifications: 10 mg/tablet, Huangshi Feiyun Pharmaceutical Co., Ltd.).
Methods
Preparation of Drug-Carrying Serum: Twenty Wistar rats were randomly divided into the blank control group (gavage with sterilized water), the high-dose total flavonoids of Rhizoma Drynariae group, the low-dose total flavonoids of Rhizoma Drynariae group, and the tripterygium glycosides group. The rats in the blank control group were gavaged with sterilized water (2mL/rat/day). For the rats in the total flavonoids of Rhizoma Drynariae groups, according to the clinical equivalent dose conversion equation proposed by the US FDA, Qianggu Capsule was administered at 300mg/kg/day (containing approximately 240mg/kg/day of total flavonoids of Rhizoma Drynariae). The total flavonoids of Rhizoma Drynariae were dissolved in sterile water to prepare a 120mg/ mL solution. The low-dose group was given 0.5mL per rat per day, diluted to 2mL for gavage, and the high-dose group was given 2mL per day per rat. The tripterygium glycoside tablets (1.2mg per rat per day) were dissolved in water for gavage. All animals were administered the tablets for three consecutive days, and blood was collected 1 hour after the last administration. The blood was placed in a sterile test tube without an anticoagulant to allow natural coagulation. All tubes were placed in a test tube rack, incubated in a 370C water bath for 1h, and centrifuged at 3000 r/min for 15min to separate the serum, which was placed in another sterile test tube and stored in a -200C refrigerator for later use. Just before use, the serum was inactivated at 560C for 30min, diluted with the serumfree medium and sterilized by filtration.
Isolation, Culture and Passage of Primary Synovial Cells: Under sterile conditions, the synovial tissue was collected, placed in a 15-mL centrifuge tube containing 5% FBS DMEM, and placed in a sterile Petri dish containing DMEM within 2 hours. Fat and blood clots on the surface were removed under sterile conditions, and the tissue was then washed 3 times. The synovial tissue was cut into small, 1mm3 pieces and placed in a 6cm2 culture dish. After adding 4mL of DMEM medium and 500μl of collagenase type I (final concentration of 1mg/mL), the tissue was digested for 4 h in an incubator at 370C and 5% CO2. The tissue block was flocculent. The sample was filtered through a 100-mesh stainless steel filter and rinsed with PBS. The filtrate was centrifuged at 1500 rpm for 5 min, and the supernatant was discarded. The cells were resuspended in 15% FBS DMEM and placed in a 10-cm2 culture dish, which was incubated at 370C and 5% CO2. After the confluence of the cells reached >80%, digestion and passage were performed. Thirdgeneration cells were digested with trypsin for the experiment.
Effect of Drug-Carrying Serum on the Proliferation of FLSs In Vitro
The third-generation osteoblasts were adjusted to a concentration of 2×103 cells/mL and seeded in a 96-well culture plate at 100μl/well. After culture with medium containing 2% fetal bovine serum for 24 hours, the cells were divided into 4 groups:
a) The 20% blank serum group;
b) The 20% high-dose total flavonoids of Rhizoma Drynariae medicated serum group;
c) The 20% low-dose total flavonoids of Rhizoma Drynariae medicated serum group and
d) The 20% tripterygium glucosides medicated serum group, with 10 wells for each group.
After culture in an incubator at 370C and 5% CO2 for 24h, 100μl of the culture solution was discarded and 20μl of thiazolyl tetrazolium (MTT) solution (5mg/mL) was added to each well, followed by incubation at 370C for 4h. The supernatant was discarded, and the crystals were dissolved in 150μl of dimethyl sulfoxide (DMSO) per well. The absorbance (wavelength at 490nm) was measured using an enzyme-linked immunosorbent assay (ELISA) analyzer. The calculation performed was as follows: cell viability % = (OD of the tested well / OD of the control well) × 100%.
Effect of Drug-Carrying Serum on the Expression of Proliferating Cell Nuclear Antigen (PCNA) mRNA and Bcl-2 mRNA in FLSs in vitro
a) The third-generation osteoblasts were digested with 0.25% trypsin to prepare a single cell suspension by repeated pipetting; the cell concentration was adjusted to 2×10 4/mL, and the cells were seeded onto a 12-well cell culture plate in 1mL per well. The cells were cultured in an incubator at 370C and 5% CO2 for 24 h. The culture medium in the well was removed completely and replaced with culture medium containing 1% fetal bovine serum for 12h; then, the culture medium in the well plate was aspirated and replaced with 20% blank control serum, 20% high-dose total flavonoids of Rhizoma Drynariae drug-carrying serum, 20% lowdose total flavonoids of Rhizoma Drynariae drug-carrying serum or 20% of tripterygium glycosides drug-carrying serum, followed by culture for another 24h.
b) The total RNA of the FLSs was extracted following the instructions of the Trizol one-step reagent, and 1μg of total RNA was used as the template to synthesize cDNA by reverse transcription.
c) The primers were synthesized by Shanghai Shenggong Bioengineering Technology Service Co., Ltd.; the primer sequences are provided in Table 1.
d) Real-time quantitative PCR: the fluorescent dye SYBR Green and the primers for the target gene, Bcl-2, and the internal reference, actin, were mixed in 5μl of SYBR Green+0.15μl of primers. After sealing, the reaction mixture was put into a real-time PCR analyzer (ABI 7900) for PCR to determine the CT values for PCNA and Bcl-2 mRNA expression. The expression level was calculated based on the 2-ΔCT equation (Table 1).
Statistical Analysis: Statistical analysis was performed on the experimental data using SPSS 18.0 software. The normally distributed data are presented as means ± standard deviations. If the variance of each treatment group was homogeneous, the LSD-t test was used for the comparison between the two groups. When the mean values of the two groups of samples were compared, if the variance was homogeneous, the t-test was used; if the variance was heterogeneous, the t’ test was used. Differences with P<0.05 were considered statistically significant.
Results
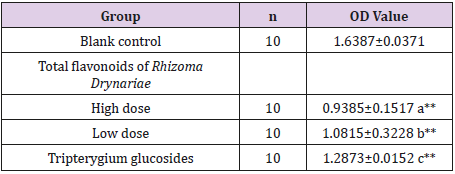
Effects of the Serum Carrying Total Flavonoids of Rhizoma Drynariae on the in vitro Cultured FLSs
Table 2: in vitro stimulation of RA-FLSs by treatment with 20% drug-carrying serum for 48h.
Note: Compared with the blank control group, **P<0.0001; at=11.325; bt = 6.125; ct = 6.335.
As shown in Table 2, compared with the blank control group, treatment with sera containing 20% high-dose and low-dose total flavonoids of Rhizoma Drynariae and tripterygium glycosides could significantly inhibit the proliferation of FLSs (P<0.0001), with no significant differences between the three groups (Table 2).
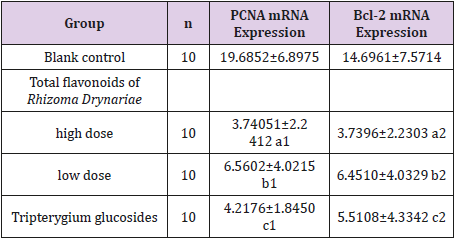
Effect of Drug-Carrying Serum on the Expression of PCNA mRNA in the in vitro Cultured FLSs
As shown in Table 3, compared with the blank control group, treatment with sera containing 20% high-dose and low-dose total flavonoids of Rhizoma Drynariae and tripterygium glycosides could significantly inhibit the expression of PCNA mRNA in FLS (P<0.0001), in which the inhibitory effect of the high-dose total flavonoids of Rhizoma Drynariae was more significant than that of the low-dose treatment (P=0.037).
Table 3: PCNA mRNA expression and BCL-2 mRNA expression.
Note: Compared with the blank control group, a1t=6.053, P<0.0001; b1t=4.879, P=0.0004; c1t=4.745, P=0.0004. a2t=5.785, P<0.0001; b2t=4.754, P=0.0005; c2t=4.753, P=0.0005.
Effect of Drug-Carrying Serum on the Expression of Bcl-2 mRNA in the in vitro Cultured FLS
As shown in Table 3, compared with the blank control group, treatment with sera containing 20% high-dose and low-dose total flavonoids of Rhizoma Drynariae and tripterygium glycosides could significantly inhibit the expression of Bcl-2 mRNA in FLS (P<0.0001), with no statistically significant differences between the three groups (Table 3).
Conclusion
Total flavonoids of Rhizoma Drynariae may inhibit the proliferation of FLSs by regulating the expression of the PCNA and Bcl-2 genes, thereby inhibiting inflammation and reducing bone loss.
Discussion
FLSs in patients with RA can play an important role in inflammation and joint destruction by secreting various proteases, arachidonic acid metabolites and their cytokines [4-6]. They are the ultimate target cells for the pathological changes of RA, and their excessive proliferation may result in synovial hyperplasia, which is one of the main pathological features of RA [7,8]. Currently, the hypothesis of insufficient apoptosis of FLS received the most attention. When the number of cells in inflammatory synovial tissue is multiplied, under normal conditions, the body can eliminate these excess cells through apoptosis to maintain a balance, but insufficient cell apoptosis may lead to hyperplasia of the synovial tissue and even a series of pathological changes in arthritis. Therefore, the excess hyperplasia and insufficient apoptosis of FLSs are the main mechanisms of RA and inhibiting the proliferation of FLSs and inducing their apoptosis are considered to be effective methods for treating RA [2,9]. Traditional plant extracts, such as celastrol [10] and curcumin [11], can inhibit the proliferation of FLSs by inducing DNA damage, cell cycle arrest and apoptosis; thus, they are considered to have potential in treating RA. However, their application is limited due to their high toxicity and lack of identification of the active ingredients.
The Chinese traditional drugs used in RA treatment have multiple targets and multiple pathways, with a wide range of pharmacological activities, showing the importance of their development and their research value. The continuous efficacy and safety of chemical synthetic drugs and kinase inhibitors in the treatment of RA were confirmed by a clinical study, but their longterm safety remains to be further investigated [12]. Proliferating cell nuclear antigen (PCNA) is an accessory protein of DNA polymerase 8, also known as cyclin. As a peptide synthesized and expressed only in proliferating cells, PCNA plays an important role in the initiation of cell proliferation. The change in its content and expression level is consistent with the activity of DNA synthesis and replication, which can be used as an indicator to evaluate the state of cell proliferation. Our study showed that the high-dose and lowdose total flavonoids of Rhizoma Drynariae medicated sera could significantly inhibit the expression of PCNA mRNA in FLSs and that the effect of the high-dose treatment was more significant than that of the low-dose treatment. The Bcl-2 gene family, which is the apoptosis-regulating gene family that receives the most attention, and its related protein Bcl-2 are the earliest studied genes related to apoptosis.
Bcl-2 is an important apoptosis-regulating gene that can prolong cell life by inhibiting various forms of cell death, leading to an increase in the number of cells and an increase in the mutation rate of genetic material. The high-dose and low-dose total flavonoids of Rhizoma Drynariae medicated sera could significantly inhibit the expression of Bcl-2 mRNA in FLSs. In this study, the serum pharmacological research method was used to observe the effect of serum carrying total flavonoids of Rhizoma Drynariae on the proliferation and apoptosis of FLSs in vitro. The results showed that 20% serum carrying total flavonoids of Rhizoma Drynariae could promote the proliferation of FLSs cultured in vitro and that the effect of the high-dose treatment was more significant than that of the low-dose treatment. It was further confirmed by the results of real-time quantitative PCR that the expression levels of PCNA and Bcl-2 were decreased significantly after treatment with the drug-carrying sera, indicating that the proliferation of FLSs was significantly inhibited, which is consistent with the results of the MTT assay. This may also be one of the mechanisms by which the serum carrying total flavonoids of Rhizoma Drynariae can downregulate the proliferation of FLSs. This study provides a powerful theoretical basis for the clinical treatment of RA using total flavonoids of Rhizoma Drynariae at the cellular and molecular levels.
Declarations
Ethics Approval and Consent to Participate
Ethical approval was given by the Ethics Committee of Guanghua Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai, China. All patients gave their written information consent.
Consent for Publication
Not applicable.
Availability of Data and Material
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Competing Interests
All the authors declare that they have no conflict of interest.
Funding
This work was supported by the Project of Shanghai Science and Technology Commission, grant number 134119b1500 and the Natural Science Project of Shanghai University of Traditional Chinese Medicine, grant number 18LK077.
Author’s Contributions
LS and XL contributed to the study design. All authors collected the data and performed the data analysis. All authors prepared the manuscript.
Acknowledgements
None.
References
- Mercante JW, Winchell JM (2015) Current and emerging Legionella diagnostics for laboratory and
- outbreak investigations. Clin microbiol rev 28(1): 95-133.
- Yamamoto H, Sugiura M, Kusunoki S, Ezaki T, Ikedo M, et al. (1992) Factors stimulating propagation of legionellae in cooling tower water. Appl environ microbiol 58(4): 1394-1397.
- Mouchtouri VA, Goutziana G, Kremastinou J, Hadjichristodoulou C (2010) Legionella species colonization in cooling towers: risk factors and assessment of control measures. Am J infect control 38(1): 50-55.
- Legionella Treatment.
- Hilborn ED, Wade TJ, Hicks L, Garrison L, Carpenter J, et al. (2013) Surveillance for waterborne disease outbreaks associated with drinking water and other nonrecreational water-United States, 2009–2010. MMWR 62(35): 714-720.
- Adams D, Fullerton K, Jajosky R, Sharp P, Onweh D, et al. (2015) Summary of notifiable infectious diseases and conditions-United States, 2013. MMWR Morb mortal wkly rep 62(53): 1-122.
- CDC (2015) Notice to readers: Final 2014 reports of nationally notifiable infectious diseases. MMWR
- Morb mortal wkly rep 64(36): 1019-1033.
- Burillo A, Pedro-Botet ML, Bouza E (2017) Microbiology and epidemiology of legionnaire's disease.
- Infect dis clin North Am 31(1): 7-27.
- Brady MF, Sundareshan V (2018) Legionnaire’s disease (Legionella Infection) [Updated 2018 Oct 27].
- In: StatPearls [Internet]. Treasure Island (FL): Stat Pearls Publishing.
- Palmer A, Painter J, Hassler H, Richards VP, Bruce T, et al. (2016) Legionella clemsonensis sp. nov.: A green fluorescing Legionella strain from a patient with pneumonia. Microbiol immunol 60(10): 694-701.
- Leung MH, Lee PK (2016) The roles of the outdoors and occupants in contributing to a potential
- pan-microbiome of the built environment: A review. Microbiome 4(1): 21.
- Baron JL, Vikram A, Duda S, Stout JE, Bibby K (2014) Shift in the microbial ecology of a hospital hot water system following the introduction of an on-site monochloramine disinfection system. Plos one 9(7): e102679.
- Feazel LM, Baumgartner LK, Peterson KL, Frank DN, Harris JK, et al. (2009) Opportunistic pathogens enriched in showerhead biofilms. Proce National Academy of Sci United State Am106(38): 16393-16399.
- Wery N, Bru-Adan V, Minervini C, Delgenes JP, Garrelly L, et al. (2008) Dynamics of Legionella spp. and bacterial populations during the proliferation of pneumophila in a cooling tower facility. App environ microbiol 74(10): 3030-3037.
- Sharmin F, Wakelin S, Huygens F, Hargreaves M (2013) Firmicutes dominate the bacterial taxa within
- sugar-cane processing plants. Scientific reports 3: 3107.
- Pereira RPA, Peplies J, Hofle MG, Brettar I (2017) Bacterial community dynamics in a cooling tower with emphasis on pathogenic bacteria and Legionella species using universal and genus-specific deep sequencing. Water Res 122: 363-376.
- Buchbinder S, Trebesius K, Heesemann J (2002) Evaluation of detection of Legionella spp. in water samples by fluorescence in situ hybridization, PCR amplification and bacterial culture. Int J Med Microbiol 292: 241-245.
- (2011) Centers for Disease Control and Prevention. Legionellosis-United States, 2000-2009. Morb Mortal Wkly Rep 60: 1083-1086.
- (2013) European Centre for Disease Prevention and Control. Legionnaire’s Disease Surveillance in Europe, 2011. Stockholm: ECDC.
- Adams DA, Gallagher KM, Jajosky RA, Kriseman J, Sharp P, et al. (2013) Centers for Disease Control and Prevention. Summary of notifiable diseases-United States, 2011. MMWR Morb Mortal Wkly Rep 60(53): 1-117.
- (2014) Department of Health. Australia’s National Notifiable Diseases Surveillance System (NNDSS). Canberra, ACT: Australian Government.
- Harriet W, Alexandra K, Howard F, Kirstin R (2004) Uncertainties associated with assessing the public health risk from Legionella. Front Microbiol 5.
- Casini B, Aquino F, Totaro M, Miccoli M, Galli I, et al. (2017) Application of hydrogen peroxide as an innovative method of treatment for Legionella control in a hospital water network. Pathogens 6(2):pii: 15.
- Moosavian M, Dashti A (2011) Isolation and identification of Legionellosis agents from fishponds,swimming pools and cooling towers in Khuzestan province, Iran. Jundishapur J Microbiol 4: 209-216.
- Weiss D, Boyd C, Rakeman JL, Greene SK, Fitzhenry R, et al. (2017) a large community outbreak of legionnaires' disease associated with a cooling tower in New York City, 2015. Public Health Rep 132(2): 241-250.
- (2015) NYC Health. Fact sheet: Legionnaires’ disease in the South Bronx.
- (2016) NSW Health. Public Health investigation into the legionella outbreaks in Sydney CBD.
- (2016) NSW Health. Legionella outbreaks2016- A detailed overview.

 Research Article
Research Article